Abstract
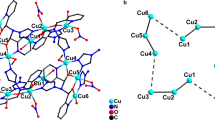
Methyliminodiacetic acid (H2Mida) and imidazole react with copper(II) to form crystals of the square pyramidal complex [Cu(Mida)Im]. One N and two O atoms of the Mida ligand (Cu-N 2.010(1) Å, Cu-O 1.955(1) Å, and 1.978(1) Å) and the imidazole N atom (1.950(1) Å) lie at the base of the pyramid. The carboxyl O atom of the neighboring complex lies at the apical position (2.411(1) Å); in this way the individual complexes are linked into infinite zigzag chains. Substitution of imidazole by 1,10-phenanthroline gave [Cu2(Mida)2(Phen)H2O]·2H2O crystals with two nonequivalent centrosymmetric octahedral anions [Cu(Mida)2]2− of face type (Cu-N 2.023 Å and 2.028(2) Å, Cu-Oax 2.579 Å and 2.530(2) Å, Cu-Obas 1.952 Å and 1.936(2) Å). The anions serve as bridges in chains between the [Cu(Phen)H2O]2+ cation fragments to which they are bonded by their axial carboxyl groups. The Cu atom of the cation has a [4+1] environment (with the H2O molecule lying on the axis of the pyramid, and with two N atoms of the ligand and two O atoms of the anions lying at the base).
Similar content being viewed by others
References
G. Nardin, L. Randaccio, R. P. Bonomo, and E. Rizzarelli, J. Chem. Soc., Dalton Trans., No. 3 369–375 (1980).
A. Castineiras, M. E. Abarca, L. De la Cueva, et al., J. Coord. Chem., 30, Nos. 3/4, 273–281 (1993).
M. Tribet, B. Covelo, D. Choquesillo-Lazarte, et al., Inorg. Chem. Commun., 6, No. 4 343–345 (2003).
Xu Duanjun, Cheng Chaorong, Xu Yuanzhi, and Hu Shengzhi, Jiegou Huaxue, 8, No. 2 81–85 (1989).
N. Bresciani-Pahor, G. Nardin, R. P. Bonomo, and E. Rizzarelli, J. Chem. Soc., Dalton Trans., No. 12, 2625–2630 (1984).
Yu. Xu, C. Cheng, D. Xu, and D. Chen, Huaxue Wuli Xuebae, No. 2 480–486 (1989).
I. S. De la Cueva, A. G. Sicilia, J. M. Gonzalez, et al., React. Funct. Polym., 36, No. 3 211–216 (1998).
I. F. Burshtein, A. L. Poznyak, and L. V. Stopolyanskaya, Zh. Neorg. Khim., 44, No. 8, 1251–1257 (1999).
A. Castineiras, J. M. Tercero, A. Matilla, et al., J. Coord. Chem., 35, Nos. 1/2, 61–72 (1995).
N.-H. Dung, B. Viossat, A. Busnot, et al., Inorg. Chim. Acta, 174, No. 2 145–148 (1990).
N.-H. Dung, B. Viossat, A. Busnot, et al., ibid., 169, No. 1 9–12.
D. Chen, Z. Liu, W. Tang, et al., Acta Crystallogr., C46, No. 8, 1426–1429 (1990).
A. Uehara, E. Kyuno, and R. Tsuchiya, Bull. Chem. Soc. Jpn., 43, No. 5, 1394–1400 (1970).
A. Altomare, M. C. Burla, M. Carnalli, et al., J. Appl. Crystallogr., 32, 115 (1999).
G. M. Shedrick, SHELXL-97, Program for Crystal Structure Refinement, Univ. Göttingen, Germany (1997).
D. Cremer and J. A. Pople, J. Am. Chem. Soc., 97, No. 6, 1354–1358 (1975).
P. Sivy, B. Koren, F. Valach, and I. Lukes, Acta Crystallogr., C45, No. 1 23–25 (1989).
I. N. Polyakova, A. L. Poznyak, and V. S. Sergienko, Zh. Neorg. Khim., 46, No. 4 633–640 (2001).
Author information
Authors and Affiliations
Additional information
Original Russian Text Copyright © 2005 by M. Zabel, A. L. Poznyak, and V. I. Pawlowski
__________
Translated from Zhurnal Strukturnoi Khimii, Vol. 46, No. 3, pp. 564–568, May–June, 2005.
Rights and permissions
About this article
Cite this article
Zabel, M., Poznyak, A.L. & Pawlowski, V.I. Crystal structures of copper(II) complexes with methyliminodiacetate ions and 1,10-phenanthroline or imidazole [Cu(Phen)H2O][Cu(Mida)2]·2H2O and [Cu(Mida)Im]. J Struct Chem 46, 551–555 (2005). https://doi.org/10.1007/s10947-006-0138-6
Received:
Issue Date:
DOI: https://doi.org/10.1007/s10947-006-0138-6