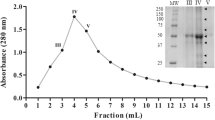
Abstract
A new proteolytic enzyme, named hieronymain III, has been purified by ion-exchange chromatography from unripe fruits of Bromelia hieronymi Mez. The new peptidase belongs to the cysteine catalytic type, as well as hieronymain I and II, the other two peptidases previously isolated from this species. Hieronymain III showed optimum alkaline pH range (8.6–9.3) and the molecular mass (MALDI-TOF) was 23713 Da. The N-terminal sequence (AVPQSIDWRRYGAVTTSRNQG) exhibited a higher percentage identity with hieronymain II (93%) than with hieronymain I (71%). The three peptidases showed notable differences on synthetic substrates degradation: whereas hieronymain III was the only one able to hidrolyze Z-Arg-Arg-p-nitroanilide, hieronymain I and II could degrade Z-Phe-Arg-p-nitroanilide; on the other hand, PFLNA was only split by hieronymain I. Finally, the three proteases showed different preferences on N-α-CBZ-p-nitrophenyl aminoacid ester substrates. From a biotechnological point of view, cleavage specificity differences are significant enough to use these enzymes as potential tools in that area.







Similar content being viewed by others
Abbreviations
- AMPSO:
-
3-[(1, 1-dimethyl-2-hydroxyethyl)amino]-2-hydroxy-propanesulfonic acid
- BLAST:
-
Basic local alignment search tool
- CAPS:
-
3-(ciclohexylamino)-1-propanesulfonic acid
- CBZ:
-
Carbobenzoxy
- DMSO:
-
Dimethyl sulfoxide
- DTT:
-
Dithiothreitol
- E-64:
-
trans-epoxysuccinyl-L-leucylamido-(4-guanidino)butane
- EDTA:
-
Ethylendiaminetetraacetic acid
- IEF:
-
Isoelectric focusing
- FPLC:
-
Fast protein liquid chromatographay
- MALDI-MS TOF:
-
Matrix-assisted laser desorption/ionization mass spectrometry time of flying
- MES:
-
2-(N-morpholino)ethanesulfonic acid
- MOPS:
-
3-(N-morpholino)propanesulfonic acid
- PFLNA:
-
p-Glu-Phe-Leu-p-nitro-anilide
- Q-Sepharose HP:
-
Quaternary sepharose high performance
- SDS-PAGE:
-
Sodium dodecyl sulfate polyacrylamide gel electrophoresis
- SP-Sepharose HP:
-
Sulphopropyl sepharose high performance
- TAPS:
-
N-tris (hydroxymethyl) methyl-3-aminopropanesulfonic acid
- TFA:
-
Trifluoroacetic acid
- Z:
-
Benzyloxycarbonyl
References
Barrett AJ, Rawlings ND, Woessner JF (2004) In: Handbook of proteolytic enzymes, 2nd edn. Academic Press, London, UK, pp 1051–1204
Murachi T, Yamazaki M (1970) Biochemistry 9:1935–1938
Rowan AD, Buttle DJ (1994) Method Enzymol 244:555–568
Vallés D, Furtado S, Cantera AMB (2007) Enzyme Microb Tech 40:409–413
Pardo MF, López LMI, Caffini NO, Natalucci CL (2001) Biol Chem Hoppe Seyler 382:871–874
Cabral H, Leopoldino AM, Tajara EH, Greene LJ, Faça VM, Mateus RP, Ceron CR, Judice WAS, Julianod L, Bonilla-Rodriguez GO (2006) Protein Pept Lett 13:83–89
Bruno MA, Pardo MF, Caffini NO, López LMI (2003) J Protein Chem 22:127–134
Bruno MA, Trejo SA, Avilés FX, Caffini NO, López LMI (2006) Protein J 25:224–231
Abreu Payrol J, Obregón WD, Natalucci CL, Caffini NO (2005) Fitoterapia 6:540–548
Abreu Payrol J, Obregón WD, Trejo SA, Caffini NO (2007) Protein J 27:88–96
López LMI, Sequeiros C, Natalucci CL, Brullo A, Maras B, Barra D, Caffini NO (2000) Protein Expr Purif 18:133–140
López LMI, Sequeiros C, Trejo SA, Pardo MF, Caffini NO, Natalucci CL (2001) Biol Chem Hoppe-Seyler 382:875–878
Harrach T, Eckert K, Schulze-Forster K, Nuck R, Grunow D, Maurer HR (1995) J Protein Chem 14:41–52
Napper AD, Bennett SP, Borowski M, Holdridge MB, Leonard MJ, Rogers EE, Duan Y, Laursen RA, Reinhold B, Shames SL (1994) Biochem J 301:727–735
Rowan AD, Buttle DJ, Barrett AJ (1988) Archiv Biochem Biophys 267:262–270
Ota S, Muta E, Katanita Y, Okamoto Y (1985) J Biochem 98:219–228
Rowan AD, Buttle DJ, Barrett AJ (1990) Biochem J 266:869–875
Silverstein RM (1974) Anal Biochem 62:478–484
Carter CE, Marriage H, Goodenough PW (2000) Biochemistry 39:11005–11013
Lee KL, Albee KL, Bernasconi RJ, Edmunds T (1997) Biochem J 327:199–202
Wiederanders B (2003) Acta Biochim Pol 50:691–713
Grudkowska M, Zagdańska B (2004) Acta Biochim Pol 51:609–624
Walsh G (2002) Proteins Biochemistry and Biotechnology. John Wiley and Sons Ltd, West Sussex, England, p 420
Haileselassie SS, Lee BH, Gibbs BF (1999) J Dairy Sci 82:1612–1617
Silva SV, Malcata FX (2005) Int Dairy J 15:1–15
Perpetuo EA, Juliano L, Lebrun I (2003) J Prot Chem 22:601–606
Torres-Llanez M, Vallejo-Cordova B, González-Córdova A (2005) Arch Latinoam Nutr 55:111–117
Bruno MA, Pardo MF, Caffini NO, López LMI (2002) Acta Farm Bonaerense 21:51–56
Natalucci CL, Priolo NS, Buttazzoni MS, Caffini NO (1985) Acta Farm Bonaerense 4:93–98
Anderson JW (1968) Phytochemistry 7:1973–1988
Bradford MB (1976) Anal Biochem 72:248–254
Natalucci CL, Brullo A, López LMI, Hilal RM, Caffini NO (1996) J Food Biochem 19:443–454
Salvesen GS, Nagase H (2001) In: Beynon R, Bond JS (eds) Proteolytic Enzymes, 2nd edn. Oxford University Press, Oxford, pp 116–119
Filippova IY, Lysogorskaya EN, Oksenoit ES, Rudenskaya GN, Stepanov VM (1984) Anal Biochem 143:293–297
Laemmli UK (1970) Nature 227:680–685
Barrett AJ, Kirschke H (1981) Method Enzymol 80:535–561
Altschul SF, Madden TL, Schaffer AA, Zhang J, Zhang Z, Miller W, Lipman DJ (1997) Nucleic Acids Res 25:3389–3402
Rawlings ND, Barrett AJ (1994) Method Enzymol 244:461–486
Ritonja A, Buttle DJ, Rawlings ND, Turk V, Barrett AJ (1989) FEBS Lett 258:109–112
Muta E, Okamoto Y, Ota S (1994) DDBJ, Accession D38531, (Direct Submission)
Taylor MA, Al-Sheikh M, Revell DF, Sumner IG, Connerton IF (1999) Plant Sci 145:41–47
Kyndt T, Van Damme EJ, Van Beeumen J, Gheysen G (2007) FEBS J 274:451–462
Ohyanagi H, Tanaka T, Sakai H, Shigemoto Y, Yamaguchi K, Habara T, Fujii Y, Antonio BA, Nagamura Y, Imanishi T, Ikeo K, Itoh T, Gojobori T, Sasak T (2006) Nucleic Acids Res 34:D741–D744
Walreavens V, Jaziri M, van Beeumen J, Schnek AG, Kleinschmidt T, Looze Y (1993) Biol Chem 374:501–506
Ling JQ, Kojima T, Shiraiwa M, Takahara H (2003) Biochim Biophys Acta 1627:129–139
Funk V, Kositsup B, Zhao C, Beers EP (2002) Plant Physiol 128:84–94
Sakuta C, Oda A, Konishi M, Yamakawa S, Kamada H, Satoh S (2001) Biosci Biotechnol Biochem 65:2243–2248
Jones ML, Chaffin GS, Eason JR, Clark DG (2005) J Exp Bot 56:2733–2744
Acknowledgments
N. O. Caffini is member of the CICPBA Researcher Career; L. M. I. López is member of the CONICET Researcher Career; M. A. Bruno is UNLP fellow. The present work was supported by grants from ANPCyT (PICT 38088), CONICET (PIP 5931), CYTED (Project IV.22), CICPBA (Res. 527/06), and University of La Plata, Argentina (Project X-445). Authors wish thank to Elisa F. Villagra and Adrián Ojeda for very helpful technical assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bruno, M.A., Trejo, S.A., Caffini, N.O. et al. Purification and Characterization of Hieronymain III. Comparison with Other Proteases Previously Isolated from Bromelia hieronymi Mez. Protein J 27, 426–433 (2008). https://doi.org/10.1007/s10930-008-9152-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10930-008-9152-1