Abstract
The fast Fourier transformation has been the gold standard for transforming data from time to frequency domain in many spectroscopic methods, including NMR. While reliable, it has as a drawback that it requires a grid of uniformly sampled data points. This needs very long measuring times for sampling in multidimensional experiments in all indirect dimensions uniformly and even does not allow reaching optimal evolution times that would match the resolution power of modern high-field instruments. Thus, many alternative sampling and transformation schemes have been proposed. Their common challenges are the suppression of the artifacts due to the non-uniformity of the sampling schedules, the preservation of the relative signal amplitudes, and the computing time needed for spectra reconstruction. Here we present a fast implementation of the Iterative Soft Thresholding approach (istHMS) that can reconstruct high-resolution non-uniformly sampled NMR data up to four dimensions within a few hours and make routine reconstruction of high-resolution NUS 3D and 4D spectra convenient. We include a graphical user interface for generating sampling schedules with the Poisson-Gap method and an estimation of optimal evolution times based on molecular properties. The performance of the approach is demonstrated with the reconstruction of non-uniformly sampled medium and high-resolution 3D and 4D protein spectra acquired with sampling densities as low as 0.8%. The method presented here facilitates acquisition, reconstruction and use of multidimensional NMR spectra at otherwise unreachable spectral resolution in indirect dimensions.







Similar content being viewed by others
Abbreviations
- NMR:
-
Nuclear magnetic resonance
- IST:
-
Iterative soft thresholding
- istHMS:
-
Implementation of IST at Harvard Medical School
- FM reconstruction:
-
Forward maximum entropy reconstruction
- MDD:
-
Multi-dimensional decomposition
- FDM:
-
Filter diagonalization method
- FFT:
-
Fast Fourier transformation
- DFT:
-
Discrete Fourier transformation
- NOE:
-
Nuclear Overhauser enhancement
- NOESY:
-
NOE spectroscopy
- GUI:
-
Graphical user interface
References
Barna JCJ, Laue ED, Mayger MR, Skilling J, Worrall SJP (1987) Exponential sampling, an alternative method for sampling in two-dimensional NMR experiments. J Magn Reson 73:69–77
Candes EJ, Romberg J, Tao T (2006) Robust uncertainty principles: exact signal reconstruction from highly incomplete frequency information. Information Theory, IEEE Transactions on 52(2):489–509
Cavanagh J, Fairbrother WJ, Palmer AG III, Rance M, Skelton NJ (2007) Protein NMR spectroscopy: principles and practice, 2nd edn. Academic Press, New York
Chen J, Nietlispach D, Shaka AJ, Mandelshtam VA (2004) Ultra-high resolution 3D NMR spectra from limited-size data sets. J Magn Reson 169(2):215–224. doi:10.1016/j.jmr.2004.04.017
Coggins BE, Zhou P (2008) High resolution 4-D spectroscopy with sparse concentric shell sampling and FFT-CLEAN. J Biomol NMR 42(4):225–239. doi:10.1007/s10858-008-9275-x
Denk W, Baumann R, Wagner G (1986) Quantitative evaluation of cross peak intensities by projection of two-dimensional NOE spectra on a linear space spanned by a set of reference resonance lines. J Magn Reson 67:386–390
Donoho DL (1995) De-noising by soft-thresholding. Inform Theory IEEE Trans 41:613–627
Donoho DL (2006) Compressed sensing. Information Theory, IEEE Transactions on 52(4):1289–1306
Drori I (2007) Fast l1 minimizatio by iterative thresholding for multidimensional NMR spectroscopy. Eurasip J Adv Signal Process 1–10
Gautier A, Mott HR, Bostock MJ, Kirkpatrick JP, Nietlispach D (2010) Structure determination of the seven-helix transmembrane receptor sensory rhodopsin II by solution NMR spectroscopy. Nat Struct Mol Biol 17(6):768–774. doi:10.1038/nsmb.1807
Gronenborn AM, Filpula DR, Essig NZ, Achari A, Whitlow M, Wingfield PT, Clore GM (1991) A novel, highly stable fold of the immunoglobulin binding domain of streptococcal protein G. Science 253(5020):657–661
Hiller S, Ibraghimov I, Wagner G, Orekhov VY (2009) Coupled decomposition of four-dimensional NOESY spectra. J Am Chem Soc 131(36):12970–12978. doi:10.1021/ja902012x
Hoch JC (1989) Modern spectrum analysis in nuclear magnetic resonance: alternatives to the Fourier transform. Methods Enzymol 176:216–241
Högbom (1974) Aperture synthesis with a non-regular distribution of interferometer baselines. Astron Astrophys Suppl 15:417–426
Holland DJ, Bostock MJ, Gladden LF, Nietlispach D (2011) Fast multidimensional NMR spectroscopy using compressed sensing. Angew Chem 50(29):6548–6551. doi:10.1002/anie.201100440
Hyberts SG, Heffron GJ, Tarragona NG, Solanky K, Edmonds KA, Luithardt H, Fejzo J, Chorev M, Aktas H, Colson K, Falchuk KH, Halperin JA, Wagner G (2007) Ultrahigh-resolution (1)H-(13)C HSQC spectra of metabolite mixtures using nonlinear sampling and forward maximum entropy reconstruction. J Am Chem Soc 129(16):5108–5116
Hyberts SG, Frueh DP, Arthanari H, Wagner G (2009) FM reconstruction of non-uniformly sampled protein NMR data at higher dimensions and optimization by distillation. J Biomol NMR 45(3):283–294. doi:10.1007/s10858-009-9368-1
Hyberts SG, Takeuchi K, Wagner G (2010) Poisson-gap sampling and forward maximum entropy reconstruction for enhancing the resolution and sensitivity of protein NMR data. J Am Chem Soc 132(7):2145–2147. doi:10.1021/ja908004w
Hyberts SG, Arthanari H, Wagner G (2011) Applications of non-uniform sampling and processing. Top Curr Chem. doi:10.1007/128_2011_187
Kazimierczuk K, Orekhov VY (2011) Accelerated NMR spectroscopy by using compressed sensing. Angew Chem 50(24):5556–5559. doi:10.1002/anie.201100370
Kazimierczuk K, Zawadzka A, Kozminski W, Zhukov I (2007) Lineshapes and artifacts in multidimensional Fourier transform of arbitrary sampled NMR data sets. J Magn Reson 188(2):344–356. doi:10.1016/j.jmr.2007.08.005
Kazimierczuk K, Zawadzka A, Kozminski W (2008) Optimization of random time domain sampling in multidimensional NMR. J Magn Reson 192(1):123–130. doi:10.1016/j.jmr.2008.02.003
Kupce E, Freeman R (2003) Projection-reconstruction of three-dimensional NMR spectra. J Am Chem Soc 125(46):13958–13959
Kupce E, Freeman R (2005) Fast multidimensional NMR: radial sampling of evolution space. J Magn Reson 173(2):317–321. doi:10.1016/j.jmr.2004.12.004
LeMaster DM, Kushlan DM (1996) Dynamical mapping of E. coli thioredoxin via 13C NMR relaxation analysis. J Am Chem Soc 118(39):9255–9264
Lustig M, Donoho D, Pauly JM (2007) Sparse MRI: the application of compressed sensing for rapid MR imaging. Magnetic resonance in medicine. Official J Soci Magn Reson Med/Soci of Magn Reson Med 58(6):1182–1195. doi:10.1002/mrm.21391
Mandelshtam VA, Taylor HS, Shaka AJ (1998) Application of the filter diagonalization method to one- and two-dimensional NMR spectra. J Magn Reson 133(2):304–312
Matsuki Y, Eddy MT, Herzfeld J (2009) Spectroscopy by integration of frequency and time domain information for fast acquisition of high-resolution dark spectra. J Am Chem Soc 131(13):4648–4656. doi:10.1021/ja807893k
Milbradt AG, Kulkarni M, Yi T, Takeuchi K, Sun ZY, Luna RE, Selenko P, Naar AM, Wagner G (2011) Structure of the VP16 transactivator target in the Mediator. Nat Struct Mol Biol 18(4):410–415. doi:10.1038/nsmb.1999
Mobli M, Stern AS, Hoch JC (2006) Spectral reconstruction methods in fast NMR: reduced dimensionality, random sampling and maximum entropy. J Magn Reson 182(1):96–105. doi:10.1016/j.jmr.2006.06.007
Peng JW, Wagner G (1992) Mapping of spectral density functions using heteronuclear NMR relaxation measurements. J Magn Res 98:308–332
Peng JW, Wagner G (1994) Investigation of protein motions via relaxation measurements. Methods Enzymol 239:563–596
Pervushin K, Riek R, Wider G, Wuthrich K (1997) Attenuated T2 relaxation by mutual cancellation of dipole–dipole coupling and chemical shift anisotropy indicates an avenue to NMR structures of very large biological macromolecules in solution. Proc Natl Acad Sci USA 94(23):12366–12371
Rovnyak D, Frueh DP, Sastry M, Sun ZY, Stern AS, Hoch JC, Wagner G (2004a) Accelerated acquisition of high resolution triple-resonance spectra using non-uniform sampling and maximum entropy reconstruction. J Magn Reson 170(1):15–21
Rovnyak D, Hoch JC, Stern AS, Wagner G (2004b) Resolution and sensitivity of high field nuclear magnetic resonance spectroscopy. J Biomol NMR 30(1):1–10
Stanek J, Kozminski W (2010) Iterative algorithm of discrete Fourier transform for processing randomly sampled NMR data sets. J Biomol NMR 47(1):65–77. doi:10.1007/s10858-010-9411-2
Stanek J, Augustyniak R, Kozminski W (2011) Suppression of sampling artefacts in high-resolution four-dimensional NMR spectra using signal separation algorithm. J Magn Reson. doi:10.1016/j.jmr.2011.10.009
Stern AS, Donoho DL, Hoch JC (2007) NMR data processing using iterative thresholding and minimum l(1)-norm reconstruction. J Magn Reson 188(2):295–300. doi:10.1016/j.jmr.2007.07.008
Suzuki H, Toriwaki J (1991) Automatic segmentation of head MRI images by knowledge guided thresholding. Comput Med Imaging Graph 15(4):233–240
Ting M, Raich R, Hero AO 3rd (2009) Sparse image reconstruction for molecular imaging. IEEE Trans Image Process 18(6):1215–1227. doi:10.1109/TIP.2009.2017156
Tugarinov V, Kay LE (2004) An isotope labeling strategy for methyl TROSY spectroscopy. J Biomol NMR 28(2):165–172. doi:10.1023/B:JNMR.0000013824.93994.1f
Tugarinov V, Hwang PM, Ollerenshaw JE, Kay LE (2003) Cross-correlated relaxation enhanced 1H[bond]13C NMR spectroscopy of methyl groups in very high molecular weight proteins and protein complexes. J Am Chem Soc 125(34):10420–10428. doi:10.1021/ja030153x
Tugarinov V, Kay LE, Ibraghimov I, Orekhov VY (2005) High-resolution four-dimensional 1H–13C NOE spectroscopy using methyl-TROSY, sparse data acquisition, and multidimensional decomposition. J Am Chem Soc 127(8):2767–2775
Wagner G (1993) NMR relaxation and protein mobility. Curr Opin Struct Biol 3:748–754
Wagner G (1997) An account of NMR in structural biology. Nat Struct Biol 4(Suppl):841–844
Wen J, Wu J, Zhou P (2011) Sparsely sampled high-resolution 4-D experiments for efficient backbone resonance assignment of disordered proteins. J Magn Reson 209(1):94–100. doi:10.1016/j.jmr.2010.12.012
Wilton DJ, Tunnicliffe RB, Kamatari YO, Akasaka K, Williamson MP (2008) Pressure-induced changes in the solution structure of the GB1 domain of protein G. Proteins 71(3):1432–1440. doi:10.1002/prot.21832
Zhou P, Lugovskoy AA, Wagner G (2001) A solubility-enhancement tag (SET) for NMR studies of poorly behaving proteins. J Biomol NMR 20(1):11–14
Acknowledgment
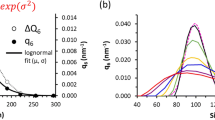
This research was supported by the National Institutes of Health (Grants GM047467, CA127990, GM094608 and EB002026). We thank Dr. Koh Takeuchi for providing the data used in Fig. 7B.
Author information
Authors and Affiliations
Corresponding author
Additional information
Contribution from Harvard Medical School, Boston, MA 02115, USA.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Hyberts, S.G., Milbradt, A.G., Wagner, A.B. et al. Application of iterative soft thresholding for fast reconstruction of NMR data non-uniformly sampled with multidimensional Poisson Gap scheduling. J Biomol NMR 52, 315–327 (2012). https://doi.org/10.1007/s10858-012-9611-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10858-012-9611-z