Abstract
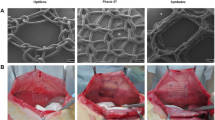
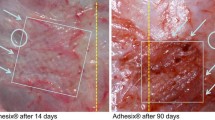
The increasing use of mesh insertion for groin hernia repair is dashed by a worrying prevalence of chronic pain frequently related as a reaction to the biomaterial implantation. Thus, new biocompatible prosthesis, designed as a composite material associating polypropylene (PP) and long-term absorbable material, are now under development. In the present study, the typical commercially available Prolene® mesh has been compared to two new meshes designed with 3-fold less PP, either alone (light PP) or associated with poly-l-lactic acid (PP-PLA) accounting for 90% of the mesh weight. These PP-mesh variants were implanted in an extraperitoneal position within the abdominal wall of 90 rats. Mesh adhesion and size were determined at autopsy 2, 4 and 8 weeks after implantation (10 animals per group) and morphometric parameters of the host tissues by light microscopy. Prolene® and light PP-meshes presented intra-corporeal shrinkage and tissue adhesion, both more pronounced with light-PP, whilst PP-PLA meshes were not affected in spite of a strongest fibrosis. In contrast to Prolene® and even more with light PP meshes, inflammation and cell-mediated immune responses were reduced without occurrence of angiogenesis or oedema. All these findings advocate together for a better tolerance of this new composite biomaterial, more likely due to a low macrophage response that appeared statistically correlated to the absence of mesh shrinkage and to a decreased adhesion to the tissue. On the basis of these experimental observations, it could be expected that the better tolerance of this composite biomaterial may avoid both long-term pain and recurrence when used as plug in groin hernia repair.



Similar content being viewed by others
References
T. FASHID, T. J. HEIKKINEN, S. WOLLERT, J. OSTERBERG, S. SMEDBERG, H. GRANLUND, S. RAMEL, G. FELLANDER and B. ANDERBERG, Ann. R. Coll. Surg. Engl. 82 (2000) 396.
J. M. HAY, M. J. BOUDET, A. FINGERHUT, J. POUCHER, H. HENNET, E. HABIB, M. VEYRIERES and Y. FLAMANT, Ann. Surg. 222 (1995) 719.
I. L. LICHTENSTEIN, A. G. SHULMAN, P. K. AMID and M. M. MONTLLOR, Am. J. Surg. 157 (1989) 188.
J. E. MCGILLICUDDY, Arch. Surg. 133 (1998) 974.
R. C. READ, Hernia 8 (2004) 8.
H. S. GOLDSTEIN, Hernia 3 (1999) 23.
J. M. BELLON, J. BUJAN, L. CONTRERAS and A. HERNANDO, Biomaterials 16 (1995) 381.
B. KLOSTERHALFEN, K. JUNGE, B. HERMANNS and U. KLINGE, Br. J. Surg. 89 (2002) 1043.
J. KÖNINGER, J. REDECKE and M. BUTTERS, Langenbeck Arch. Surg. 389 (2004) 361.
M. Bay-Nielsen, F. M. Perkins, H. Kehlet and Danish Hernia Database, Ann. Surg. 233 (2001) 1.
C. A. COURTENEY, K. DUFFY, M. G. SERPELL and P. J. O’DWYER, Br. J. Surg. 89 (2002) 1310.
E. P. PÉLISSIER, D. BLUM, J. M. DAMAS and P. MARRE, Hernia 4 (1999) 201.
J. P. LAMB, T. VITALE and D. L. KAMINSKI, Surgery 93 (1983) 643.
J. TYRELL, H. SIBERMAN, P. CHANDRASOMA, J. NILAND and J. SHULL, Surg. Gynecol. Obstet. 168 (1989) 227.
U. KLINGE, V. SCHUMPELICK and B. KLOSTERHALFEN, Biomaterials 22 (2001) 1415.
B. KLOSTERHALFEN, U. KLINGE and V. SCHUMPELICK, Biomaterials 19 (1998) 2235.
P. K. AMID, Hernia 1 (1997) 15.
A. S. LOWHAM, C. J. FILIPI, R. J. Jr FITZGIBBONS, R. STOPPA, G. E. WANTZ, E. L. FELIX and W. B. CRAFTON, Ann. Surg. 225 (1997) 422.
U. KLINGE, B. KLOSTERHALFEN, M. MULLER, A. P. OTTINGER and V. SCHUMPELICK, Eur. J. Surg. 164 (1998) 965.
B. KLOSTERHALFEN, U. KLINGE, B. HERMANNS and V. SCHUMPELICK, Chirurg 71 (2000) 43.
K. JUNGE, U. KLINGE, R. ROSCH, B. KLOSTERHALFEN and V. SCHUMPELICK, World J. Surg. 26 (2002) 1472.
S. BRINGMAN, T. J. HEIKKINEN, S. WOLLERT, J. OSTERBERG, S. SMEDBERG, H. GRANLUND, S. RAMEL, G. FELLANDER and B. ANDERBERG, Hernia 8 (2004) 127.
M. KELLOMAKI, H. NIIRANEN, K. PUUMANEN, N. ASHAMMAKHI, T. WARIS and P. TORMALA, Biomaterials 21 (2000) 2495.
K. S. FURUKAWA, T. USHIDA, K. TOITA, Y. SAKAI and T. TATEISHI, Cell Transplant. 11 (2002) 475.
G. R. EVANS, K. BRANDT, S. KATZ, P. CHAUVIN, L. OTTO, M. BOGLE, B. WANG, R. K. MESZLENYI, L. LU, A. G. MIKOS and C. W. Jr PATRICK, Biomaterials 23 (2002) 841.
J. R. FUCHS, I. POMERANTSEVA, E. R. OCHOA, J. P. VACANTI and D. O. FAUZA, J. Pediatr. Surg. 38 (2003) 1348.
S. J. LEIBOVICH and R. ROSS, Am. J. Pathol. 78 (1975) 71.
U. KLINGE, B. KLOSTERHALFEN, M. MÜLLER and V. SCHUMPELICK, Eur. J. Surg. 165 (1999) 665.
F. C. USHER, J. OCHSNER and L. L. Jr TUTTLE, Am. Surg. 24 (1958) 969.
J. RIVES, Int. Surg. 47 (1967) 360.
R. STOPPA, J. PETIT and X. HENRY, Int. Surg. 60 (1975) 411.
J. P. ARNAUD, R. ELOY, M. ADLOFF and J. F. GRENIER, Am. J. Surg. 3 (1977) 338.
J. C. MAYAGOITIA, Hernia 8 (2004) 64.
S. DABROWIECKI, K. SVANES, J. LEKVEN and K. GRONG, Eur. Surg. Res. 23 (1991) 240.
R. ROSCH, K. JUNGE, R. QUESTER, U. KLINGE, B. KLOSTERHALFEN and V. SCHUMPELICK, Eur. Surg. Res. 35 (2003) 445.
R. G. HOLZHEIMER, Eur. J. Med. Res. 9 (2004) 323.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tanaka, K., Mutter, D., Inoue, H. et al. In vivo evaluation of a new composite mesh (10% polypropylene/90% poly-l-lactic acid) for hernia repair. J Mater Sci: Mater Med 18, 991–999 (2007). https://doi.org/10.1007/s10856-006-0090-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10856-006-0090-1