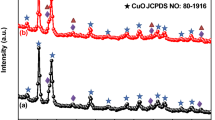
Abstract
Herein, clusters of Cu with size of ˂10 nm were successfully synthesized via galvanic replacement method using various copper salts as precursors. The clusters self-assembled into various morphologies of Cu nanostructures with different apparent colors. Then, Ag nanostructures were deposited on dendritic nanostructures of Cu via same route for preparing Cu–Ag bimetallic alloy, for the first time. Effect of precursor and surfactant type on morphology of products was investigated. As-synthesized products were characterized by techniques such as SEM, TEM, DRS and XRD. Moreover, the prepared Cu–Ag bimetallic alloy was utilized as photocatalyst for decolorization of avid violet 7 as an azo dye under visible light irradiation. As a result, enhanced photocatalytic efficiency was obtained for Cu–Ag nanostructures compared to bare Cu and Ag nanostructures (98% degradation compared to 59 and 78% in 90 min). Effect of deposition time of Ag and thus, effect of Ag percentage in alloy on photocatalytic efficiency were studied and the results showed that 30 s is optimum time for achieving highest photocatalytic efficiency.








Similar content being viewed by others
References
D.P. Huang, Y.Y. Qi, X.T. Bai, L.J. Shi, H. Jia, D.J. Zhang, L.Q. Zheng, ACS Appl. Mater. Interfaces 4, 4665 (2012)
W. Zhang, F.T. Tan, W. Wang, X.L. Qiu, X.L. Qiao, J.G. Chen, J. Hazard. Mater. 36, 217 (2012)
A. Gutés, C. Carraro, R. Maboudian, J. Am. Chem. Soc. 132, 1476 (2010)
H.P. Wu, J.F. Liu, X.J. Wu, M.Y. Ge, Y.W. Wang, G.Q. Zhang, J.Z. Jiang, Int. J. Adhes. Adhes. 26, 617 (2006)
Y. Zhang, C. Lu, G. Zhao, Z. Wang, RSC Adv. 6, 51569 (2016)
J. Fu, W. Ye, C. Wang, Mater. Chem. Phys. 141, 107 (2013)
X. Wen, Y.-T. Xie, M.W.C. Mak, K.Y. Cheung, X.Y. Li, R. Renneberg, S. Yang. Langmuir 22, 4836 (2006)
M.A. El-Sayed, Acc. Chem. Res. 34, 257 (2001)
J.B. Jackson, N.J. Halas, J. Phys. Chem. B 105, 2473 (2001)
P. Zhou, Z.H. Dai, M. Fang, X.H. Huang, J.C. Bao, J.F. Gong, J. Phys. Chem. C 111, 12609 (2007)
X.L. Xu, J.B. Jia, X.R. Yang, S.J. Dong, Langmuir 26, 7627 (2010)
Q.M. Shen, L.P. Jiang, H. Zhang, Q.H. Min, W.H. Hou, J.J. Zhu, J. Phys. Chem. C 112, 16385 (2008)
R. Qiu, H.G. Cha, H.B. Noh, Y.B. Shim, X.L. Zhang, R. Qiao, D. Zhang, Y.I. Kim, U. Pal, Y.S. Kang, J. Phys. Chem. C 113, 15891 (2009)
X.J. Zhang, G.F. Wang, X.W. Liu, H.Q. Wu, B. Fang, Cryst. Growth Des. 8, 1430 (2008)
Y. Chen, Q. Xu, B. Hu, J. Xu, J. Wenga, RSC Adv. 6, 2464 (2016)
H. Zhang, Y. Ni, Y. Zhong, H. Wua, M. Zhaia, RSC Adv. 5, 96639 (2015)
Y. Wang, S. Liang, Q. Yang, X. Wang, Appl. Surf. Sci. 387, 805 (2016)
H.H. Nersisyan, Y.J. Lee, S.H. Joo, S.K. Han, T.H. Lee, J.S. Lee, Y.S. An, J.H. Lee, Cryst. Eng. Commun. 17, 7535 (2015)
Y. Asano, K. Nakaoka, K. Murashiro, T. Komatsu, K. Hoshino, Mater. Lett. 81, 162 (2012)
S. Mansour, N. Boutarek, S.E. Amar, Adv. Mat. Lett. 5, 356 (2014)
N. Toshima, T. Yonezawa, New J. Chem. 22, 1179 (1998)
L.-u. Rahman, A. Shah, S.K. Lunsford, C. Han, M.N. Nadagouda, E. Sahle-Demessie, R. Qureshi, M.S. Khan, H.B. Kraatz, D.D. Dionysiou, RSC Adv. 5, 44427 (2015)
L.-u. Rahman, A. Shah, S.B. Khan, A.M. Asiri, H. Hussain, C. Han, R. Qureshi, M.N. Ashiq, M.A. Zia, M. Ishaq, H.-B. Kraatz, J. Appl. Electrochem. 45, 463 (2015)
L.-u. Rahman, A. Shah, R. Qureshi, S.B. Khan, A. M. Asiri, A.-u.-H.A. Shah, M. Ishaq, M.S. Khan, S.K. Lunsford, M.A. Zia, Adv. Mater. Sci. Eng. 10, 1155 (2015)
M.S. Khan, M. Zaka, B.H. Abbasi, L. Rahman, A. Shah, IET Nanobiotech. 10, 359 (2016)
L.-u. Rahman, R. Qureshi, M.M. Yasinzai, A. Shah, C.R. Chim. 15, 533 (2012)
M. Zaka, B.H. Abbasi, L.-u. Rahman, A. Shah, M. Zia, IET Nanobiotech. 10, 134 (2016)
M.A. Al-Jalali, Adv. Mater. Lett. 5, 14 (2014)
L. Fu, M.M. Sokiransky, J. Wang, G. Lai, A. Yu, Physica. E. 83, 146 (2016)
A.M. Vora, Adv. Mater. Lett. 4, 146 (2013)
Z. Zarghami, M. Ramezani, M. Maddahfar, J. Mater. Sci. -Mater. Electron. 26, 5884 (2015)
Z. Zarghami, A. Akbari, A.M. Latifi, M.A. Amani, Bioresour. Technol. 205, 230 (2106)
M. Mousavi-Kamazani, Z. Zarghami, M. Salavati-Niasari, J. Phys. Chem. C 120, 2096 (2016)
N. Toshima, Y. Wang, Adv. Mater. 6, 240 (1994)
N. Toshima, P. Lu, Chem. Lett. 25, 725 (1996)
M.K. Kumar, K. Bhavani, G. Naresh, B. Srinivas, A. Venugopal, Appl. Catal. B Environ. 199, 282 (2016)
Acknowledgements
This work was supported by the Chemistry Research Center at Islamic Azad University, Birjand branch.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rahmatolahzadeh, R., Ebadi, M. & Motevalli, K. Preparation and characterization of Cu clusters and Cu–Ag alloy via galvanic replacement method for azo dyes degradation. J Mater Sci: Mater Electron 28, 6056–6063 (2017). https://doi.org/10.1007/s10854-016-6281-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10854-016-6281-8