Abstract
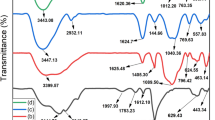
An imidazolium salt-based hydrogel with hyperbranched topology (hbGel-PEG-TTIm+Br −6 ) was proposed for efficient adsorption towards anionic hexavalent chromium ions (Cr(VI)). For the purpose, a bromo-terminated A3 type monomer and a imidazole-terminated B3 type monomer were firstly synthesized via O-esterification and Michael addition, respectively. Then, hbGel-PEG-TTIm+Br −6 was obtained by the one-pot step polymerization of “A3 + B3” approach via N-alkylation reaction. The chemical structure and morphology of the as-prepared hydrogel were confirmed by Fourier transform infrared and scanning electron microscope. The cross-linking network structure was calculated according to the Flory–Rehner equilibrium swelling equation. The effects of solution pH, adsorbent dosage, contact time and initial Cr(VI) concentration on the adsorption performance were also investigated, and the maximum adsorption capacity could reach ~ 238 mg g−1. In addition, the adsorption process of Cr(VI) on hbGel-PEG-TTIm+Br −6 was well described by the pseudo-second-order kinetic model, and the adsorption equilibrium data fitted with Langmuir isotherm model better than Freundlich isotherm model. This novel hydrogel with hyperbranched topology can potentially be applied in the treatment of wastewater containing Cr(VI).











Similar content being viewed by others
References
Saha B, Orvig C (2010) Biosorbents for hexavalent chromium elimination from industrial and municipal effluents. Coord Chem Rev 254(23–24):2959–2972
Salmani Abyaneh A, Fazaelipoor MH (2016) Evaluation of rhamnolipid (RL) as a biosurfactant for the removal of chromium from aqueous solutions by precipitate flotation. J Environ Manag 165:184–187
Aoudj S, Khelifa A, Drouiche N, Belkada R, Miroud D (2015) Simultaneous removal of chromium(VI) and fluoride by electrocoagulation–electroflotation: application of a hybrid Fe–Al anode. Chem Eng J 267:153–162
Nancharaiah YV, Venkata Mohan S, Lens PNL (2015) Metals removal and recovery in bioelectrochemical systems: a review. Bioresour Technol 195:102–114
Athanasekou C, Romanos GE, Papageorgiou SK, Manolis GK, Katsaros F, Falaras P (2017) Photocatalytic degradation of hexavalent chromium emerging contaminant via advanced titanium dioxide nanostructures. Chem Eng J 318:171–180
Chug R, Gour VS, Mathur S, Kothari SL (2016) Optimization of extracellular polymeric substances production using azotobacter beijreinckii and bacillus subtilis and its application in chromium (VI) removal. Bioresour Technol 214:604–608
Bao Y, Yan X, Du W, Xie X, Pan Z, Zhou J, Li L (2015) Application of amine-functionalized MCM-41 modified ultrafiltration membrane to remove chromium (VI) and copper (II). Chem Eng J 281:460–467
Song W, Gao B, Zhang T, Xu X, Huang X, Yu H, Yue Q (2015) High-capacity adsorption of dissolved hexavalent chromium using amine-functionalized magnetic corn stalk composites. Bioresour Technol 190:550–557
Liang X, Fan X, Li R, Li S, Shen S, Hu D (2018) Efficient removal of Cr(VI) from water by quaternized chitin/branched polyethylenimine biosorbent with hierarchical pore structure. Bioresour Technol 250:178–184
Wang S, Ma H, Peng J, Zhang Y, Chen J, Wang L, Xu L, Li J, Zhai M (2015) Facile synthesis of a novel polymeric ionic liquid gel and its excellent performance for hexavalent chromium removal. Dalton Trans 44(16):7618–7625
Jiang Y, Li F, Ding G, Chen Y, Liu Y, Hong Y, Liu P, Qi X, Ni L (2015) Synthesis of a novel ionic liquid modified copolymer hydrogel and its rapid removal of Cr(VI) from aqueous solution. J Colloid Interface Sci 455:125–133
Lee S, Eom Y, Park J, Lee J, Kim SY (2017) Micro-hydrogel particles consisting of hyperbranched polyamidoamine for the removal of heavy metal Ions from water. Sci Rep 7(1):1–9
Khiratkar AG, Senthil Kumar S, Bhagat PR (2016) Designing a sulphonic acid functionalized benzimidazolium based poly(ionic liquid) for efficient adsorption of hexavalent chromium. RSC Adv 6(44):37757–37764
Mi H, Jiang Z, Kong J (2013) Hydrophobic poly(ionic liquid) for highly effective separation of methyl blue and chromium ions from water. Polymers 5(4):1203–1214
Yeganeh H, Jamshidi H, Jamshidi S (2007) Synthesis and properties of novel biodegradable poly(ɛ-caprolactone)/poly(ethylene glycol)-based polyurethane elastomers. Polym Int 56(1):41–49
Merrill EW, Dennison KA, Sung C (1993) Partitioning and diffbsion of solutes in hydrogels of poly(ethylene oxide). Biomaterials 14(15):1117–1126
Zhuang Y, Yu F, Chen H, Zheng J, Ma J, Chen J (2016) Alginate/graphene double-network nanocomposite hydrogel beads with low-swelling, enhanced mechanical properties, and enhanced adsorption capacity. J Mater Chem A 4(28):10885–10892
Dinda D, Gupta A, Saha SK (2013) Removal of toxic Cr(VI) by UV-active functionalized graphene oxide for water purification. J Mater Chem A 1(37):11221–11228
Zhu L, Liu Y, Chen J (2009) Synthesis of N-methylimidazolium functionalized strongly basic anion exchange resins for adsorption of Cr(VI). Ind Eng Chem Res 48(7):3261–3267
Park D, Yun Y, Jo JH, Park JM (2006) Biosorption process for treatment of electroplating wastewater containing Cr(VI): laboratory-scale feasibility test. Ind Eng Chem Res 45(14):5059–5065
Zhang Y, Xu L, Zhao L, Peng J, Li C, Li J, Zhai M (2012) Radiation synthesis and Cr(VI) removal of cellulose microsphere adsorbent. Carbohyd Polym 88(3):931–938
Qian L, Yang M, Zhang S, Hou C, Song W, Yang J, Tang R (2018) Preparation of a sustainable bioadsorbent by modifying filter paper with sodium alginate, with enhanced mechanical properties and good adsorption of methylene blue from wastewaters. Cellulose 25:2021–2036
Shi Y, Zhang T, Ren H, Kruse A, Cui R (2018) Polyethylene imine modified hydrochar adsorption for chromium (VI) and nickel (II) removal from aqueous solution. Bioresour Technol 247:370–379
Zhang X, Zhang L, Li A (2018) Eucalyptus sawdust derived biochar generated by combining the hydrothermal carbonization and low concentration KOH modification for hexavalent chromium removal. J Environ Manag 206:989–998
Rahman MT, Barikbin Z, Badruddoza AZM, Doyle PS, Khan SA (2013) Monodisperse polymeric ionic liquid microgel beads with multiple chemically switchable functionalities. Langmuir 29(30):9535–9543
Li C, Zhang Y, Peng J, Wu H, Li J, Zhai M (2012) Adsorption of Cr(VI) using cellulose microsphere-based adsorbent prepared by radiation-induced grafting. Radiat Phys Chem 81(8):967–970
Zhu L, Zhang C, Liu Y, Wang D, Chen J (2010) Direct synthesis of ordered N-methylimidazolium functionalized mesoporous silica as highly efficient anion exchanger of Cr(VI). J Mater Chem 20:1553–1559
Deng Y, Long T, Zhao H, Zhu L, Chen J (2012) Application of porous N-methylimidazolium strongly basic anion exchange resins on Cr(VI) adsorption from electroplating wastewater. Sep Sci Technol 47:256–263
Dong Z, Zhao L (2018) Covalently bonded ionic liquid onto cellulose for fast adsorption and efficient separation of Cr(VI): Batch, column and mechanism investigation. Carbohyd Polym 189:190–197
Yan J, Huang Y, Miao Y, Tjiu WW, Liu T (2015) Polydopamine-coated electrospun poly(vinyl alcohol)/poly(acrylic acid) membranes as efficient dye adsorbent with good recyclability. J Hazard Mater 283:730–739
Dong Z, Zhao J, Du J, Li C, Zhao L (2016) Radiation synthesis of spherical cellulose-based adsorbent for efficient adsorption and detoxification of Cr(VI). Radiat Phys Chem 126:68–74
Acknowledgements
This work was supported by the Science Research Foundation of Xijing University (XJ18T03 and XJ16T01), the Natural Science Foundation of Shaanxi Province (CN) (No: 2017JQ2019) and the National Natural Science Foundation of China (No: 51673157).
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Li, K., Qian, L., Song, W. et al. Preparation of an ionic liquid-based hydrogel with hyperbranched topology for efficient removal of Cr(VI). J Mater Sci 53, 14821–14833 (2018). https://doi.org/10.1007/s10853-018-2609-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10853-018-2609-5