Abstract
Purpose
To investigate the effects of male aging on sperm quality and sperm DNA fragmentation.
Methods
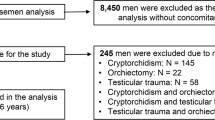
The ejaculates of 320 unselected men attending a fertility clinic and, as a control, 84 normozoospermic men without any history of ART were analyzed according to WHO guidelines. Sperm DNA fragmentation was measured by flow cytometry after staining with propidiumiodide.
Results
The patients were divided into four groups: <30 years, 30–35 years, 36–39 years and ≥40 years. Sperm motility decreased with increasing age whereas sperm concentration, morphology, and DNA fragmentation fluctuated throughout the four groups both among patients and among controls. However, we could not detect any significant correlation between male age and conventional semen parameters or sperm DNA fragmentation, respectively, neither in the patients’ group nor among the controls. This also applies to a classification of patients and controls into only two age groups with a cut-off point at 35 years.
Conclusions
Our findings suggest that neither the routinely assessed semen parameters nor the amount of spermatozoa with fragmented DNA are affected by male age.

Similar content being viewed by others
References
Engel W, Sancken U, Laccone F. Paternal age from a genetic point of view. J Reproduktionsmed Endokrinol. 2004;1:263–7. German.
Hook EB, Schreinemachers DM, Willey AM, Cross PK. Rates of mutant structural chromosome rearrangements in human foetuses: data from prenatal cytogenetic studies and associations with maternal age and parental mutagen exposure. Am J Hum Genet. 1983;35:96–109.
Hecht F, Hecht BK. Aneuploidy in humans: Dimensions, demography, and dangers of abnormal numbers of chromosomes. In: Vig BK, Sandberg AA, editors. Aneuploidy part II: Incidence and etiology. New York: Alan R. Liss; 1987. p. 9–49.
Hassold T, Hunt PA, Sherman S. Trisomy in humans: incidence, origin and etiology. Curr Opin Genet Dev. 1993;3:398–403. doi:10.1016/0959-437X(93)90111-2.
Wyrobek AJ, Aardema M, Eichenlaub-Ritter U, Ferguson L, Marchetti F. Mechanisms and targets involved in maternal and paternal age effects on numerical aneuploidy. Environ Mol Mutagen. 1996;28:254–64. doi:10.1002/(SICI)1098-2280(1996)28:3<254::AID-EM9>3.0.CO;2-D.
Hassold T, Hunt P. To err (meiotically) is human: the genesis of human aneuploidy. Nat Rev Genet. 2001;2:280–91. doi:10.1038/35066065.
Nicolaidis P, Petersen MB. Origin and mechanisms of non-disjunction in human autosomal trisomies. Hum Reprod. 1998;13:313–9. doi:10.1093/humrep/13.2.313.
Sloter ED, Marchetti F, Eskenazi B, Weldon RH, Nath J, Cabreros D, et al. Frequency of human sperm carrying structural aberrations of chromosome 1 increases with advancing age. Fertil Steril. 2007;87:1077–86. doi:10.1016/j.fertnstert.2006.08.112.
Luetjens CM, Rolf C, Gassner P, Werny JE, Nieschlag E. Sperm aneuploidy rates in younger and older men. Hum Reprod. 2002;17:1826–32. doi:10.1093/humrep/17.7.1826.
Donnelly ET, O’Connell M, McClure N, Lewis SE. Differences in nuclear DNA fragmentation and mitochondrial integrity of semen and prepared human spermatozoa. Hum Reprod. 2000;15:1552–61. doi:10.1093/humrep/15.7.1552.
Sakkas D, Mariethoz E, Manicardi G, Bizzaro D, Bianchi PG, Bianchi U. Origin of DNA damage in ejaculated human spermatozoa. Rev Reprod. 1999;4:31–7. doi:10.1530/ror.0.0040031.
Nicoletti I, Migliorati G, Pagliacci MC, Grignani F, Riccardi C. A rapid and simple method for measuring thymocyte apoptosis by propidium iodide staining and flow cytometry. J Immunol Methods. 1991;139(2):271–9. doi:10.1016/0022-1759(91)90198-O.
World Health Organization. WHO laboratory manual for the examination of human semen and sperm-cervical mucus interaction, 4th ed. Cambridge, UK: Published on behalf of the World Health Organization by Cambridge University Press; 1999.
Zini A, Libman J. Sperm DNA damage: clinical significance in the era of assisted reproduction. Curr Opin Urol. 2006;16:428–34. doi:10.1097/01.mou.0000250283.75484.dd.
Lopes S, Jurisicova A, Casper RF. Gamete-specific DNA fragmentation in unfertilized human oocytes after intracytoplasmic sperm injection. Hum Reprod. 1998;13(3):703–8. doi:10.1093/humrep/13.3.703.
Lopes S, Jurisicova A, Sun JG, Casper RF. Reactive oxygen species: potential cause for DNA fragmentation in human spermatozoa. Hum Reprod. 1998;13:896–900. doi:10.1093/humrep/13.4.896.
Potts RJ, Norarianni LJ, Jefferies TM. Seminal plasma reduces exogenous damage to human sperm, determined by the measurement of DNA strand breaks and lipid peroxidation. Mutat Res. 2000;2:249–56. doi:10.1016/S0027-5107(99)00215-8.
Agarwal A, Ramadan AS, Bedaiwy MA. Role of reactive oxygen species in the pathophysiology of human reproduction. Fertil Steril. 2003;79:829–43. doi:10.1016/S0015-0282(02)04948-8.
Baumber J, Ball BA, Linfor JJ, Meyers SA. Reactive oxygen species and cryopreservation promote DNA fragmentation in equine spermatozoa. J Androl. 2003;4:621–8.
Agarwal A, Prabakaran SA. Oxidative stress and antioxidants in male infertility: a difficult balance. Iranian J Reprod Med. 2005;3:1–8.
Gandini L, Lombardo F, Paoli D, Caponecchia L, Familiari G, Verlengia C, et al. Study of apoptotic DNA- fragmentation in human spermatozoa. Hum Reprod. 2000;15:830–9. doi:10.1093/humrep/15.4.830.
Sina Hikim AP, Swerdloff RS. Hormonal and genetic control of germ cell apoptosis in testis. Rev Reprod. 1999;4:38–47. doi:10.1530/ror.0.0040038.
Singh NP, Muller CH, Berger RE. Effects of age on DNA double strand breaks and apoptosis in human sperm. Fertil Steril. 2003;80:1420–30. doi:10.1016/j.fertnstert.2003.04.002.
Schmid TE, Eskenazi B, Baumgartner A, Marchetti F, Young S, Weldon R, et al. The effects of male age on sperm DNA damage in healthy non-smokers. Hum Reprod. 2007;22:180–7. doi:10.1093/humrep/del338.
Vagnini L, Baruffi RL, Mauri AL, Petersen CG, Massaro FC, Pontes A, et al. The effects of male age on sperm DNA damage in an infertile population. Reprod Biomed Online. 2007;15:514–9.
Plastira K, Msaouel P, Angelopoulou R, Zanioti K, Plastiras A, Pothos A, et al. The effects of age on DNA fragmentation, chromatin packaging and conventional semen parameters in spermatozoa of oligoasthenoteratozoospermic patients. J Assist Reprod Genet. 2007;24:437–43. doi:10.1007/s10815-007-9162-5.
Auger J, Jouannet P. Age and male fertility: biological factors. Rev Epidemiol Sante Publique. 2005;53:2S25–35. Spec No 2.
Sun Y, Vestergaard M, Zhu JL, Madsen KM, Olsen J. Paternal age and Apgar scores of newborn infants. Epidemiology. 2006;17:473–4. doi:10.1097/01.ede.0000220690.43455.22.
Zhu JL, Madsen KM, Vestergaard M, Olesen AV, Basso O, Olsen J. Paternal age and congenital malformations. Hum Reprod. 2005;20:3173–7. doi:10.1093/humrep/dei186.
Sloter E, Schmid TE, Marchetti F, Eskenazi B, Nath J, Wyrobek AJ. Quantitative effects of male age on sperm motion. Hum Reprod. 2006;21:2868–75. doi:10.1093/humrep/del250.
Levitas E, Lunenfeld E, Weisz N, Friger M, Potashnik G. Relationship between age and semen parameters in men with normal sperm concentration: analysis of 6,022 semen samples. Andrologia. 2007;39:45–50. doi:10.1111/j.1439-0272.2007.00761.x.
Hellstrom WJ, Overstreet JW, Sikka SC, Denne J, Ahuja S, Hoover AM, et al. Semen and sperm reference ranges for men 45 years of age and older. J Androl. 2006;27:421–8. doi:10.2164/jandrol.05156.
Tiemann-Boege I, Navisi W, Greewal R, Cohn D, Eskenaz B, Wyrobek AJ, et al. The observed human sperm mutation frequency cannot explain the achondroplasia paternal age effect. Proc Natl Acad Sci USA. 2002;99:14952–7. doi:10.1073/pnas.232568699.
Acknowledgements
This work was gratefully supported by a grant from Merck Serono.
Author information
Authors and Affiliations
Corresponding author
Additional information
Capsule Neither conventional parameters of semen quality nor sperm DNA fragmentation were significantly correlated with paternal age in 320 infertility patients and 84 control subjects.
Rights and permissions
About this article
Cite this article
Winkle, T., Rosenbusch, B., Gagsteiger, F. et al. The correlation between male age, sperm quality and sperm DNA fragmentation in 320 men attending a fertility center. J Assist Reprod Genet 26, 41–46 (2009). https://doi.org/10.1007/s10815-008-9277-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10815-008-9277-3