Abstract
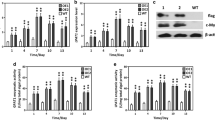
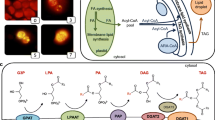
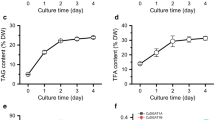
The oleaginous green microalga Lobosphaera (formerly Parietochloris) incisa accumulates high amounts of arachidonic-acid-rich triacylglycerols (TAG), in particular under conditions of nitrogen starvation. This photosynthetic organism is of great interest for studying the mechanisms responsible for storage lipid biosynthesis and the deposition of long-chain polyunsaturated fatty acids (LC-PUFA) in TAG. In this work, we report on cloning a complementary DNA (cDNA) for the putative L. incisa glycerol-3-phosphate acyltransferase (GPAT), whose deduced amino acid sequence features distinctive motives found in those mammalian and Arabidopsis GPAT isoforms that have been implicated in TAG biosynthesis. Temporal analysis of LiGPAT expression in the course of nitrogen starvation showed a positive relationship between changes in the transcript level and patterns of fatty acid production. When expressed in Arabidopsis leaf mesophyll protoplasts, the green fluorescent protein (GFP) fused to the C-terminus of LiGPAT localized outside the chloroplasts in agreement with its predicted extraplastidial localization. Based on an in silico analysis of a deduced amino acid sequence and on similarity to other GPATs participating in TAG biosynthesis, LiGPAT was expressed in the green model microalga Chlamydomonas reinhardtii in order to confirm the predicted function in a heterologous microalgal system. Overexpression of LiGPAT resulted in an up to 50 % increase in the content of TAG on a cell dry weight basis as compared to the control in the stationary phase culture without negative impact on growth parameters. Total fatty acids and TAG of the transformant lines featured an elevated level of oleic acid (18:1 n-9) and a concurrent decrease in C18 PUFA. Additional studies on the acyl substrate preference of LiGPAT are required.








Similar content being viewed by others
References
Beigneux AP, Vergnes L, Qiao X, Quatela S, Davis R, Watkins SM, Coleman RA, Walzem RL, Philips M, Reue K, Young SG (2006) Agpat6—a novel lipid biosynthetic gene required for triacylglycerol production in mammary epithelium. J Lipid Res 47:734–744
Bertolesi GE, Iannattone S, Johnston J, Zaremberg V, McFarlane S (2012) Identification and expression analysis of GPAT family genes during early development of Xenopus laevis. Gene Expr Patterns 12:219–227
Bigogno C, Khozin-Goldberg I, Boussiba S, Vonshak A, Cohen Z (2002) Lipid and fatty acid composition of the green oleaginous alga Parietochloris incisa, the richest plant source of arachidonic acid. Phytochemistry 60:497–503
Bligh EG, Dyer WJ (1959) A rapid method of total lipid extraction and purification. Can J Biochem Physiol 37:911–991
Boyle NR, Page MD, Liu B, Blaby IK, Casero D, Kropat J, Cokus SJ, Hong-Hermesdorf A, Shaw J, Karpowicz SJ, Gallaher SD, Johnson S, Benning C, Pellegrini M, Grossman A, Merchant SS (2012) Three acyltransferases and nitrogen-responsive regulator are implicated in nitrogen starvation-induced triacylglycerol accumulation in Chlamydomonas. J Biol Chem 287:15811–15825
Bustin SA, Benes V, Garson JA, Hellemans J, Huggett J, Kubista M, Mueller R, Nolan T, Pfaffl MW, Shipley GL, Vandesompele J, Wittwer CT (2009) The MIQE guidelines: minimum information for publication of quantitative real-time PCR experiments. Clin Chem 55:611–622
Cao J, Li JL, Li D, Tobin JF, Gimeno RE (2006) Molecular identification of microsomal acyl-CoA:glycerol-3-phosphate acyltransferase, a key enzyme in de novo triacylglycerol synthesis. Proc Natl Acad Sci U S A 103:19695–19700
Cattaneo ER, Pellon-Maison M, Rabassa ME, Lacunza E, Coleman RA, Gonzalez-Baro MR (2012) Glycerol-3-phosphate acyltransferase-2 is expressed in spermatic germ cells and incorporates arachidonic acid into triacylglycerols. PLoS One 7(8):e42986. doi:10.1371/journal.pone.0042986
Chen X, Truksa M, Snyder CL, El-Mezawy A, Shah S, Weselake RJ (2011) Three homologous genes encoding sn-glycerol-3-phosphate acyltransferase 4 exhibit different expression patterns and functional divergence in Brassica napus. Plant Physiol 155:851–865
Christie WW, Nikolova-Damyanova B, Laakso P, Herslof B (1991) Stereospecific analysis of triacyl-sn-glycerols via resolution of diastereomeric diacylglycerol derivatives by high-performance liquid-chromatography on silica. J Am Oil Chem Soc 68:695–701
Coleman RA, Lee DP (2004) Enzymes of triacylglycerol synthesis and their regulation. Prog Lipid Res 43:134–176
Coleman RA, Mashek DG (2011) Mammalian triacylglycerol metabolism: synthesis, lipolysis, and signaling. Chem Rev 111:6359–6386
Gidda SK, Shockey JM, Rothstein SJ, Dyer JM, Mullen RT (2009) Arabidopsis thaliana GPAT8 and GPAT9 are localized to the ER and possess distinct ER retrieval signals: functional divergence of the dilysine ER retrieval motif in plant cells. Plant Physiol Biochem 47:867–879
Gimeno RE, Cao J (2008) Thematic review series: glycerolipids. Mammalian glycerol-3-phosphate acyltransferases: new genes for an old activity. J Lipid Res 49:2079–2088
Goldberg T, Hamp T, Rost B (2012) LocTree2 predicts localization for all domains of life. Bioinformatics 28:i458–i465
Guihéneuf F, Leu S, Zarka A, Khozin-Goldberg I, Khalilov I, Boussiba S (2011) Cloning and molecular characterization of a novel acyl‐CoA: diacylglycerol acyltransferase 1‐like gene (PtDGAT1) from the diatom Phaeodactylum tricornutum. FEBS J 278:3651–3666
Gulvady AA, Murphy EJ, Ciolino HP, Cabrera RM, Jolly CA (2013) Glycerol-3-phosphate acyltransferase-1 gene ablation results in altered thymocyte lipid content and reduces thymic T cell production in mice. Lipids 48:3–12
Harwood JL, Guschina IA (2013) Regulation of lipid synthesis in oil crops. FEBS Lett 587:2079–2081
Horton P, Park KJ, Obayashi T, Fujita N, Harada H, Adams-Collier CJ, Nakai K (2007) WoLF PSORT: protein localization predictor. Nucleic Acids Res 35:W585–W587
Hu Q, Sommerfeld M, Jarvis E, Ghirardi M, Posewitz M, Seibert M, Darzins A (2008) Microalgal triacylglycerols as feedstocks for biofuel production: perspectives and advances. Plant J 54:621–639
Iskandarov U, Khozin-Goldberg I, Cohen Z (2009) Cloning and characterization of the ∆6 polyunsaturated fatty acid elongase from the green microalga Parietochloris incisa. Lipids 44:545–554
Iskandarov U, Khozin-Goldberg I, Cohen Z (2010) Identification and characterization of Delta12, Delta6, and Delta5 desaturases from the green microalga Parietochloris incisa. Lipids 45:519–530
Iskandarov U, Khozin-Goldberg I, Cohen Z (2011) Selection of a DGLA-producing mutant of the microalga Parietochloris incisa: I. Identification of mutation site and expression of VLC-PUFA biosynthesis genes. Appl Microbiol Biotechnol 90:249–256
Iwai M, Ikeda K, Shimojima M, Ohta H (2014) Enhancement of extraplastidic oil synthesis in Chlamydomonas reinhardtii using a type‐2 diacylglycerol acyltransferase with a phosphorus starvation-inducible promoter. Plant Biotechnol J 12:808–819
Jackson MR, Nilsson T, Peterson PA (1990) Identification of a consensus motif for retention of transmembrane proteins in the endoplasmic reticulum. EMBO J 9:3153–3162
Jain RK, Coffey M, Lai K, Kumar A, MacKenzie SL (2000) Enhancement of seed oil content by expression of glycerol-3-phosphate acyltransferase genes. Biochem Soc Trans 28:958–961
Kennedy EP (1961) Biosynthesis of complex lipids. Fed Proc 20:934–940
Khozin-Goldberg I, Iskandarov U, Cohen Z (2011) LC-PUFA from photosynthetic microalgae: occurrence, biosynthesis, and prospects in biotechnology. Appl Microbiol Biotechnol 91:905–915
Kindle KL (1990) High-frequency nuclear transformation of Chlamydomonas reinhardtii. Proc Natl Acad Sci U S A 87:1228–1232
La Russa M, Bogen C, Uhmeyer A, Doebbe A, Filippone E, Kruse O, Mussgnug JH (2012) Functional analysis of three type-2 DGAT homologue genes for triacylglycerol production in the green microalga Chlamydomonas reinhardtii. J Biotechnol 162:13–20
Lewin TM, Wang P, Coleman RA (1999) Analysis of amino acid motifs diagnostic for the sn-glycerol-3-phosphate acyltransferase reaction. Biochemistry 38:5764–5771
Li X, Moellering ER, Liu B, Johnny C, Fedewa M, Sears BB, Kuo MH, Benning C (2012) A galactoglycerolipid lipase is required for triacylglycerol accumulation and survival following nitrogen deprivation in Chlamydomonas reinhardtii. Plant Cell 24:4670–4686
Li J, Han D, Wang D, Ning K, Jia J, Wei L, Jing X, Huang S, Chen J, Li Y, Hu Q, Xu J (2014) Choreography of transcriptomes and lipidomes of Nannochloropsis reveals the mechanisms of oil synthesis in microalgae. Plant Cell 26:1645–1665
Liu B, Benning C (2013) Lipid metabolism in microalgae distinguishes itself. Curr Opin Biotechnol 24:300–309
Maisonneuve S, Bessoule JJ, Lessire R, Delseny M, Roscoe TJ (2010) Expression of rapeseed microsomal lysophosphatidic acid acyltransferase isozymes enhances seed oil content in Arabidopsis. Plant Physiol 152:670–684
Marr N, Foglia J, Terebiznik M, Athenstaedt K, Zaremberg V (2012) Controlling lipid fluxes at glycerol-3-phosphate acyltransferase step in yeast: unique contribution of Gat1p to oleic acid-induced lipid particle formation. J Biol Chem 287:10251–10264
Molnar A, Bassett A, Thuenemann E, Schwach F, Karkare S, Ossowski S, Weigel D, Baulcombe D (2009) Highly specific gene silencing by artificial microRNAs in the unicellular alga Chlamydomonas reinhardtii. Plant J 58:165–174
Murata N, Tasaka Y (1997) Glycerol-3-phosphate acyltransferase in plants. Biochim Biophys Acta 1348:10–16
Nagle CA, Vergnes L, Dejong H, Wang S, Lewin TM, Reue K, Coleman RA (2008) Identification of a novel sn-glycerol-3-phosphate acyltransferase isoform, GPAT4, as the enzyme deficient in Agpat6−/− mice. J Lipid Res 49:823–831
Nakagawa T, Harada N, Miyamoto A, Kawanishi Y, Yoshida M et al (2012) Membrane topology of murine glycerol-3-phosphate acyltransferase 2. Biochem Biophys Res Commun 418:506–511
Neupert J, Karcher D, Bock R (2009) Generation of Chlamydomonas strains that efficiently express nuclear transgenes. Plant J 57:1140–1150
Nguyen HM, Baudet M, Cuiné S, Adriano JM, Barthe D et al (2011) Proteomic profiling of oil bodies isolated from the unicellular green microalga Chlamydomonas reinhardtii: with focus on proteins involved in lipid metabolism. Proteomics 11:4266–4273
Nilsson T, Jackson M, Peterson PA (1989) Short cytoplasmic sequences serve as retention signals for transmembrane proteins in the endoplasmic reticulum. Cell 58:707–718
Niu YF, Zhang MH, Li DW, Yang WD, Liu JS, Bai WB, Li HY (2013) Improvement of neutral lipid and polyunsaturated fatty acid biosynthesis by overexpressing a type 2 diacylglycerol acyltransferase in marine diatom Phaeodactylum tricornutum. Mar Drugs 11:4558–4569
Rismani-Yazdi H, Haznedaroglu BZ, Hsin C, Peccia J (2012) Transcriptomic analysis of the oleaginous microalga Neochloris oleoabundans reveals metabolic insights into triacylglyceride accumulation. Biotechnol Biofuels 5:1–16
Sanjaya MR, Durrett TP, Kosma DK, Lydic TA et al (2013) Altered lipid composition and enhanced nutritional value of Arabidopsis leaves following introduction of an algal diacylglycerol acyltransferase 2. Plant Cell 25:677–693
Shan D, Li JL, Wu L, Li D, Hurov J, Tobin JF, Gimeno RE, Cao J (2010) GPAT3 and GPAT4 are regulated by insulin-stimulated phosphorylation and play distinct roles in adipogenesis. J Lipid Res 51:1971–1981
Shrestha P (2005) Biosynthesis and mobilization of arachidonic-acid-rich triacylglycerols in the green microalga Parietochloris incisa. PhD Thesis. Ben Gurion University of the Negev
Shtaida N, Khozin-Goldberg I, Solovchenko A, Chekanov K, Didi-Cohen S, Leu S, Cohen Z, Boussiba S (2014) Downregulation of a putative plastid PDC E1α subunit impairs photosynthetic activity and triacylglycerol accumulation in nitrogen-starved photoautotrophic Chlamydomonas reinhardtii. J Exp Bot 65:6563–6576
Solovchenko AE, Khozin-Goldberg I, Didi-Cohen S, Cohen Z, Merzlyak MN (2008) Effects of light intensity and nitrogen starvation on growth, total fatty acids and arachidonic acid in the green microalga Parietochloris incisa. J Appl Phycol 20:245–251
Solovchenko AE, Khozin-Goldberg I, Cohen Z, Merzlyak MN (2009) Carotenoid-to-chlorophyll ratio as a proxy for assay of total fatty acids and arachidonic acid content in the green microalga Parietochloris incisa. J Appl Phycol 21:361–366
Suh MC, Schultz DJ, Ohlrogge JB (2002) What limits production of unusual monoenoic fatty acids in transgenic plants? Planta 215:584–595
Tang W, Yuan J, Chen X, Gu X, Luo K, Li J, Wan B, Wang Y, Yu L (2006) Identification of a novel human lysophosphatidic acid acyltransferase, LPAAT-theta, which activates mTOR pathway. J Biochem Mol Biol 39:626–635
Taylor DC, Katavic V, Zou J, MacKenzie SL, Keller WA et al (2002) Field testing of transgenic rapeseed cv. Hero transformed with a yeast sn-2 acyltransferase results in increased oil content, erucic acid content and seed yield. Mol Breed 8:317–322
Wagner M, Hoppe K, Czabany T, Heilmann M, Daum G, Feussner I, Fulda M (2010) Identification and characterization of an acyl-CoA:diacylglycerol acyltransferase 2 (DGAT2) gene from the microalga O. tauri. Plant Physiol Biochem 48:407–416
Wendel AA, Lewin TM, Coleman RA (2009) Glycerol-3-phosphate acyltransferases: rate limiting enzymes of triacylglycerol biosynthesis. Biochim Biophys Acta 1791:501–506
Wiberg E, Edwards P, Byrne J, Stymne S, Dehesh K (2000) The distribution of caprylate, caprate and laurate in lipids from developing and mature seeds of transgenic Brassica napus L. Planta 212:33–40
Wilfling F, Wang H, Haas JT, Krahmer N, Gould TJ et al (2013) Triacylglycerol synthesis enzymes mediate lipid droplet growth by relocalizing from the ER to lipid droplets. Dev Cell 24:384–399
Xu J, Zheng Z, Zou J (2009) A membrane-bound glycerol-3-phosphate acyltransferase from Thalassiosira pseudonana regulates acyl composition of glycerolipids. Botany 87:544–551
Yachdav G, Kloppmann E, Kajan L, Hecht M, Goldberg T, et al (2014) PredictProtein—an open resource for online prediction of protein structural and functional features. Nucleic Acids Res W337-43. doi: 10.1093/nar/gku366
Yoo SD, Cho YH, Sheen J (2007) Arabidopsis mesophyll protoplasts: a versatile cell system for transient gene expression analysis. Nat Protoc 2:1565–1572
Zaremberg V, McMaster CR (2002) Differential partitioning of lipids metabolized by separate yeast glycerol-3-phosphate acyltransferases reveals that phospholipase D generation of phosphatidic acid mediates sensitivity to choline-containing lysolipids and drugs. J Biol Chem 277:39035–39044
Zheng Z, Zou J (2001) The initial step of the glycerolipid pathway: identification of glycerol 3-phosphate/dihydroxyacetone phosphate dual substrate acyltransferases in Saccharomyces cerevisiae. J Biol Chem 276:41710–41716
Zorin B, Grundman O, Khozin-Goldberg I, Leu S, Shapira M, Kaye Y, Tourasse N, Vallon O, Boussiba S (2014) Development of a nuclear transformation system for oleaginous green alga Lobosphaera (Parietochloris) incisa and genetic complementation of a mutant strain, deficient in arachidonic acid biosynthesis. Plos One 9(8), e105223
Zou J, Katavic V, Giblin EM, Barton DL, MacKenzie SL, Keller WA, Hu X, Taylor DC (1997) Modification of seed oil content and acyl composition in the brassicaceae by expression of a yeast sn-2 acyltransferase gene. Plant Cell 9:909–923
Zou J, Zheng Z, Xu J (2009) Algal glycerol-3 phosphate acyltransferase, U.S. Patent Application 12/736,170
Acknowledgments
This research was financially supported by the European Commission’s Seventh Framework Program for Research and Technology Development (FP7), project GIAVAP, grant no. 266401. The support of the Kreitman School of Advanced Graduate Studies and the Albert Katz International School for Desert Studies at the Ben-Gurion University of the Negev is gratefully acknowledged. The authors would like to thank Dr. Neeru Jain and Dr. Ilkhom Khalilov for their valuable contributions at the early stages of the research, and Prof. Gideon Grafi, Dr. Simon Barak, and Dr. Assif Khan for their helpful advice in the experiments with Arabidopsis protoplasts.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 4875 kb)
Rights and permissions
About this article
Cite this article
Iskandarov, U., Sitnik, S., Shtaida, N. et al. Cloning and characterization of a GPAT-like gene from the microalga Lobosphaera incisa (Trebouxiophyceae): overexpression in Chlamydomonas reinhardtii enhances TAG production. J Appl Phycol 28, 907–919 (2016). https://doi.org/10.1007/s10811-015-0634-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10811-015-0634-1