Abstract
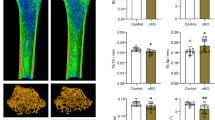
Osteoporosis (OP) is a systemic skeletal disease that promotes bone fragility and the risk of fractures. Recent studies have shown the relevance of microRNAs (miRNAs) in the development of OP. This study aimed to evaluate the possible mechanisms of action underlying miR-27a loaded by mesenchymal stem cell (MSC)–derived extracellular vesicles (MSC-EVs) in OP. Serum samples from OP patients and normal controls were collected for miRNA microarray analysis. The expression of filtered miRNA was upregulated in osteoblasts (OB) and osteoclasts (OCs) for biological activity assessment. After developing OP mice using ovariectomy (OVX) and confirming OP, the miR-27a expression level was upregulated in mice by MSC-EV application. Dual-luciferase assays were conducted to validate the relationship between miR-27a and DKK2 expression. The poor expression of miR-27a was observed in patients with OP. miR-27a increased the expression of OB markers, the number of ALP-positive cells, and the number of calcium nodules in OCs. In OVX mice, miR-27a increased bone density, improved bone structure damage recovery, decreased the levels of bone resorption markers, and decreased OC number. miR-27a transmitted by MSC-EVs interacted with DKK2. MSC-EVs exerted the same protective effects as miR-27a on OP, whereas miR-27a inhibitor abolished the attenuating effects of MSC-EVs. In contrast, DKK2 depletion reversed the stimulatory effects of the miR-27a inhibitor on OP. The Wnt/β-catenin pathway was activated upon MSC-EV application and DKK2 silencing and was impaired upon the downregulation of the expression of miR-27a. MSC-EVs are effective in preventing mouse OP. This mechanism is mediated by the miR-27a/DKK2/Wnt/β-catenin signaling pathway.











Similar content being viewed by others
DATA AVAILABILITY
The analyzed data sets generated during the study are available from the corresponding author on reasonable request.
References
Noh. J.Y., Y. Yang, and H. Jung. 2020. Molecular mechanisms and emerging therapeutics for osteoporosis. International Journal of Molecular Sciences 21 (20). https://doi.org/10.3390/ijms21207623
Sozen, T., L. Ozisik, and N.C. Basaran. 2017. An overview and management of osteoporosis. Eur J Rheumatol 4 (1): 46–56. https://doi.org/10.5152/eurjrheum.2016.048.
Feng, X., and S.L. Teitelbaum. 2013. Osteoclasts: New insights. Bone Res 1 (1): 11–26. https://doi.org/10.4248/BR201301003.
Yang, D.H., and M.Y. Yang. 2019. The role of macrophage in the pathogenesis of osteoporosis. International Journal of Molecular Sciences 20 (9). https://doi.org/10.3390/ijms20092093
van Rooij, E., and S. Kauppinen. 2014. Development of microRNA therapeutics is coming of age. EMBO Molecular Medicine 6 (7): 851–864. https://doi.org/10.15252/emmm.201100899.
Gao, Y., S. Patil, and A. Qian. 2020. The role of MicroRNAs in bone metabolism and disease. International Journal of Molecular Sciences 21 (17). https://doi.org/10.3390/ijms21176081
Liu, S., X. Xu, S. Liang, Z. Chen, Y. Zhang, A. Qian, and L. Hu. 2020. The application of MSCs-derived extracellular vesicles in bone disorders: Novel cell-free therapeutic strategy. Front Cell Dev Biol 8: 619. https://doi.org/10.3389/fcell.2020.00619.
Ni, Z., S. Zhou, S. Li, L. Kuang, H. Chen, X. Luo, J. Ouyang, M. He, X. Du, and L. Chen. 2020. Exosomes: Roles and therapeutic potential in osteoarthritis. Bone Res 8: 25. https://doi.org/10.1038/s41413-020-0100-9.
Yang, B.C., M.J. Kuang, J.Y. Kang, J. Zhao, J.X. Ma, and X.L. Ma. 2020. Human umbilical cord mesenchymal stem cell-derived exosomes act via the miR-1263/Mob1/Hippo signaling pathway to prevent apoptosis in disuse osteoporosis. Biochemical and Biophysical Research Communications 524 (4): 883–889. https://doi.org/10.1016/j.bbrc.2020.02.001.
Guo, L., K. Chen, J. Yuan, P. Huang, X. Xu, C. Li, N. Qian, J. Qi, Z. Shao, L. Deng, C. He, and J. Xu. 2018. Estrogen inhibits osteoclasts formation and bone resorption via microRNA-27a targeting PPARgamma and APC. Journal of Cellular Physiology 234 (1): 581–594. https://doi.org/10.1002/jcp.26788.
Wang, T., and Z. Xu. 2010. miR-27 promotes osteoblast differentiation by modulating Wnt signaling. Biochemical and Biophysical Research Communications 402 (2): 186–189. https://doi.org/10.1016/j.bbrc.2010.08.031.
Wang, J., R. Huang, Q. Xu, G. Zheng, G. Qiu, M. Ge, Q. Shu, and J. Xu. 2020. Mesenchymal stem cell-derived extracellular vesicles alleviate acute lung injury via transfer of miR-27a-3p. Critical Care Medicine 48 (7): e599–e610. https://doi.org/10.1097/CCM.0000000000004315.
Xu, Q., Y. Cui, J. Luan, X. Zhou, H. Li, and J. Han. 2018. Exosomes from C2C12 myoblasts enhance osteogenic differentiation of MC3T3-E1 pre-osteoblasts by delivering miR-27a-3p. Biochemical and Biophysical Research Communications 498 (1): 32–37. https://doi.org/10.1016/j.bbrc.2018.02.144.
Wu, X., Q. Gu, X. Chen, W. Mi, T. Wu, and H. Huang. 2019. MiR-27a targets DKK2 and SFRP1 to promote reosseointegration in the regenerative treatment of peri-implantitis. Journal of Bone and Mineral Research 34 (1): 123–134. https://doi.org/10.1002/jbmr.3575.
Xu, R., X. Shen, Y. Si, Y. Fu, W. Zhu, T. Xiao, Z. Fu, P. Zhang, J. Cheng, and H. Jiang. 2018. MicroRNA-31a-5p from aging BMSCs links bone formation and resorption in the aged bone marrow microenvironment. Aging Cell 17 (4): e12794. https://doi.org/10.1111/acel.12794.
Alliston, T., and R. Derynck. 2002. Medicine: Interfering with bone remodelling. Nature 416 (6882): 686–687. https://doi.org/10.1038/416686a.
Das, S., and J.C. Crockett. 2013. Osteoporosis - a current view of pharmacological prevention and treatment. Drug Des Devel Ther 7: 435–448. https://doi.org/10.2147/DDDT.S31504.
Qin, Y., L. Wang, Z. Gao, G. Chen, and C. Zhang. 2016. Bone marrow stromal/stem cell-derived extracellular vesicles regulate osteoblast activity and differentiation in vitro and promote bone regeneration in vivo. Science and Reports 6: 21961. https://doi.org/10.1038/srep21961.
Hu, R., H. Li, W. Liu, L. Yang, Y.F. Tan, and X.H. Luo. 2010. Targeting miRNAs in osteoblast differentiation and bone formation. Expert Opinion on Therapeutic Targets 14 (10): 1109–1120. https://doi.org/10.1517/14728222.2010.512916.
Taipaleenmaki, H., L. Bjerre Hokland, L. Chen, S. Kauppinen, and M. Kassem. 2012. Mechanisms in endocrinology: Micro-RNAs: Targets for enhancing osteoblast differentiation and bone formation. European Journal of Endocrinology 166 (3): 359–371. https://doi.org/10.1530/EJE-11-0646.
Ismail, S.M., N.A. El Boghdady, H.S. Hamoud, and M.I. Shabayek. 2020. Evaluation of circulating miRNA-208a-3p, miRNA-155-5p and miRNA-637 as potential non-invasive biomarkers and the possible mechanistic insights into pre- and postmenopausal osteoporotic females. Archives of Biochemistry and Biophysics 684: 108331. https://doi.org/10.1016/j.abb.2020.108331.
Long, F. 2011. Building strong bones: Molecular regulation of the osteoblast lineage. Nature Reviews Molecular Cell Biology 13 (1): 27–38. https://doi.org/10.1038/nrm3254.
Bai, Y., Y. Liu, S. Jin, K. Su, H. Zhang, and S. Ma. 2019. Expression of microRNA27a in a rat model of osteonecrosis of the femoral head and its association with TGFbeta/Smad7 signalling in osteoblasts. International Journal of Molecular Medicine 43 (2): 850–860. https://doi.org/10.3892/ijmm.2018.4007.
Guo, D., Q. Li, Q. Lv, Q. Wei, S. Cao, and J. Gu. 2014. MiR-27a targets sFRP1 in hFOB cells to regulate proliferation, apoptosis and differentiation. PLoS ONE 9 (3): e91354. https://doi.org/10.1371/journal.pone.0091354.
Del Fattore, A., A. Teti, and N. Rucci. 2012. Bone cells and the mechanisms of bone remodelling. Frontiers in Bioscience (Elite Edition) 4: 2302–2321. https://doi.org/10.2741/543.
Matsumoto, T., and I. Endo. 2021. RANKL as a target for the treatment of osteoporosis. Journal of Bone and Mineral Metabolism 39 (1): 91–105. https://doi.org/10.1007/s00774-020-01153-7.
Tao, S.C., and S.C. Guo. 2019. Extracellular vesicles in bone: “dogrobbers” in the “eternal battle field.” Cell Communication and Signaling: CCS 17 (1): 6. https://doi.org/10.1186/s12964-019-0319-5.
You, L., L. Pan, L. Chen, W. Gu, and J. Chen. 2016. MiR-27a is essential for the shift from osteogenic differentiation to adipogenic differentiation of mesenchymal stem cells in postmenopausal osteoporosis. Cellular Physiology and Biochemistry 39 (1): 253–265. https://doi.org/10.1159/000445621.
Tan, W., Y. Zhang, M. Li, X. Zhu, X. Yang, J. Wang, S. Zhang, W. Zhu, J. Cao, H. Yang, and L. Zhang. 2019. miR-27a-containing exosomes secreted by irradiated skin keratinocytes delayed the migration of unirradiated skin fibroblasts. International Journal of Biological Sciences 15 (10): 2240–2255. https://doi.org/10.7150/ijbs.35356.
Hu, X., Y. Zhong, Y. Kong, Y. Chen, J. Feng, and J. Zheng. 2019. Lineage-specific exosomes promote the odontogenic differentiation of human dental pulp stem cells (DPSCs) through TGFbeta1/smads signaling pathway via transfer of microRNAs. Stem Cell Research & Therapy 10 (1): 170. https://doi.org/10.1186/s13287-019-1278-x.
Kawao, N., H. Morita, S. Iemura, M. Ishida, and H. Kaji 2020. Roles of Dkk2 in the linkage from muscle to bone during mechanical unloading in mice. International Journal of Molecular Sciences 21 (7). https://doi.org/10.3390/ijms21072547
Chan, T.F., D. Couchourel, E. Abed, A. Delalandre, N. Duval, and D. Lajeunesse. 2011. Elevated Dickkopf-2 levels contribute to the abnormal phenotype of human osteoarthritic osteoblasts. Journal of Bone and Mineral Research 26 (7): 1399–1410. https://doi.org/10.1002/jbmr.358.
Olivares-Navarrete, R., S. Hyzy, M. Wieland, B.D. Boyan, and Z. Schwartz. 2010. The roles of Wnt signaling modulators Dickkopf-1 (Dkk1) and Dickkopf-2 (Dkk2) and cell maturation state in osteogenesis on microstructured titanium surfaces. Biomaterials 31 (8): 2015–2024. https://doi.org/10.1016/j.biomaterials.2009.11.071.
Marie, P.J., and M. Kassem. 2011. Osteoblasts in osteoporosis: Past, emerging, and future anabolic targets. European Journal of Endocrinology 165 (1): 1–10. https://doi.org/10.1530/EJE-11-0132.
Hiramitsu, S., M. Terauchi, and T. Kubota. 2013. The effects of Dickkopf-4 on the proliferation, differentiation, and apoptosis of osteoblasts. Endocrinology 154 (12): 4618–4626. https://doi.org/10.1210/en.2013-1387.
Zhang, W.B., W.J. Zhong, and L. Wang. 2014. A signal-amplification circuit between miR-218 and Wnt/beta-catenin signal promotes human adipose tissue-derived stem cells osteogenic differentiation. Bone 58: 59–66. https://doi.org/10.1016/j.bone.2013.09.015.
Mu, Y., L. Zhang, X. Chen, S. Chen, Y. Shi, and J. Li. 2019. Silencing microRNA-27a inhibits proliferation and invasion of human osteosarcoma cells through the SFRP1-dependent Wnt/beta-catenin signaling pathway. Bioscience Reports 39 (6). https://doi.org/10.1042/BSR20182366.
Author information
Authors and Affiliations
Contributions
YW: conception and design of the research, analysis, and interpretation of data; XQZ: acquisition of data and statistical analysis; DLW: drafting the manuscript, revision of manuscript for important intellectual content; all authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethical Approval
All human material was acquired with signed agreements from patients or normal controls following the Ethical Committee guidelines of Tibet Corps Hospital, Chinese People’s Armed Police Forces. All animal care and experimental procedures were approved by the Animal Study Committee of Tibet Corps Hospital, Chinese People’s Armed Police Forces and were performed in accordance with the guide for the use of laboratory animals.
Consent for Publication
Not applicable.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wang, Y., Zhou, X. & Wang, D. Mesenchymal Stem Cell–Derived Extracellular Vesicles Inhibit Osteoporosis via MicroRNA-27a-Induced Inhibition of DKK2-Mediated Wnt/β-Catenin Pathway. Inflammation 45, 780–799 (2022). https://doi.org/10.1007/s10753-021-01583-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10753-021-01583-z