Abstract
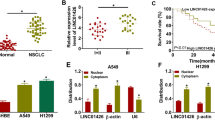
Accumulating evidence suggested that many long noncoding RNAs (lncRNAs) were widely involved in the development and progression of non-small cell lung cancer (NSCLC). However, the roles of lncRNA homeobox A11 antisense (HOXA11-AS) and its underlying mechanism in NSCLC remains largely unknown. The expression levels of HOXA11-AS, miR-3619-5p and sal-like protein 4 (SALL4) were detected by quantitative real-time polymerase chain reaction (qRT-PCR). Western blot analysis was used to measure the protein levels of hexokinase II (HK2) and SALL4. Cell proliferation, apoptosis, migration and invasion were determined by 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide assay, flow cytometry and transwell assay, respectively. The glucose consumption and lactate production were measured using glucose assay kit and lactate assay kit, respectively. The potential binding sites between miR-3619-5p and HOXA11-AS or SALL4 were predicted by online software and verified by luciferase report assay. A xenograft tumor model was established to confirm the function of HOXA11-AS in NSCLC in vivo. HOXA11-AS and SALL4 were upregulated while miR-3619-5p was downregulated in NSCLC tissues and cells. HOXA11-AS knockdown suppressed cell proliferation, migration, invasion, and glycolysis but promoted apoptosis in NSCLC cells. Moreover, miR-3619-5p could directly bind to HOXA11-AS and its inhibition attenuated the inhibitory effect of HOXA11-AS knockdown on progression of NSCLC cells. Furthermore, SALL4 was a direct target of miR-3619-5p and its overexpression reversed the anti-tumor role of miR-3619-5p in NSCLC cells. Besides, HOXA11-AS modulated SALL4 expression via sponging miR-3619-5p. Additionally, silencing HOXA11-AS inhibited tumor growth though upregulating miR-3619-5p and downregulating SALL4. Collectively, HOXA11-AS knockdown inhibited the progression of NSCLC by regulating miR-3619-5p/SALL4 axis, which might offer a novel avenue for interpreting the mechanism of NSCLC development.







Similar content being viewed by others
References
Al-Baradie R, Yamada K, Hilaire CS, Chan W-M, Andrews C, McIntosh N, Nakano M, Martonyi EJ, Raymond WR, Okumura S (2002) Duane radial ray syndrome (Okihiro syndrome) maps to 20q13 and results from mutations in SALL4, a new member of the SAL family. Am J Hum Genet 71:1195–1199
Bai Y, Lang L, Zhao W, Niu R (2019) Long Non-Coding RNA HOXA11-AS Promotes Non-Small Cell Lung Cancer Tumorigenesis Through microRNA-148a-3p/DNMT1 Regulatory Axis. Onco Targets Ther 12:11195–11206
Chen J-H, Zhou L-Y, Xu S, Zheng Y-L, Wan Y-F, Hu C-P (2017) Overexpression of lncRNA HOXA11-AS promotes cell epithelial–mesenchymal transition by repressing miR-200b in non-small cell lung cancer. Cancer Cell Int 17:64
Gao M, Li H, Bi Y, Zhang Z, Wang S, Li J, Yang Z, Lv X, Zhou B, Yin Z (2020) The polymorphisms of lncRNA HOXA11-AS and the risk of lung cancer in Northeastern Chinese population. J Cancer 11:592–598
Hsu PP, Sabatini DM (2008) Cancer cell metabolism: Warburg and beyond. Cell 134:703–707
Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D (2011) Global cancer statistics. CA Cancer J Clin 61:69–90
Jemal A, Siegel R, Xu J, Ward E (2010) Cancer statistics, 2010. CA Cancer J Clin 60:277–300
Chen JH, Zhou LY, Xu S, Zhen YL, Wan YF, Hu CP (2017) Overexpression of lncRNA HOXA11-AS promotes cell epithelial-mesenchymal transition by repressing miR-200b in non-small cell lung cancer. Cancer Cell Int 17:64
Kobayashi D, Kuribayashi K, Tanaka M, Watanabe N (2011a) Overexpression of SALL4 in lung cancer and its importance in cell proliferation. Oncol Rep 26:965–970
Kobayashi D, Kuribayshi K, Tanaka M, Watanabe N (2011b) SALL4 is essential for cancer cell proliferation and is overexpressed at early clinical stages in breast cancer. Int J Oncol 38:933–939
Kumar MS, Armenteros-Monterroso E, East P, Chakravorty P, Matthews N, Winslow MM, Downward J (2014) HMGA2 functions as a competing endogenous RNA to promote lung cancer progression. Nature 505:212
Li J, Xuan Z, Liu C (2013) Long non-coding RNAs and complex human diseases. Int J Mol Sci 14:18790–18808
Li S, Wang C, Yu X, Wu H, Hu J, Wang S, Ye Z (2017a) miR-3619-5p inhibits prostate cancer cell growth by activating CDKN1A expression. Oncol Rep 37:241–248
Li T, Xu C, Cai B, Zhang M, Gao F, Gan J (2016) Expression and clinicopathological significance of the lncRNA HOXA11-AS in colorectal cancer. Oncol Lett 12:4155–4160
Li W, Jia G, Qu Y, Du Q, Liu B (2017b) Long Non-coding RNA (LncRNA) HOXA11-AS promotes breast cancer invasion and metastasis by regulating epithelial-mesenchymal transition. Med Sci Monitor Int Med J Exp Clin Res 23:3393–3403
Liu W, Peng X, Han W, Li W, Demiao K, Fenglei Y (2017a) MicroRNA-98 plays a suppressive role in non-small cell lung cancer through inhibition of SALL4 protein expression. Oncol Res 25:975
Liu W, Xiao P, Wu H, Wang L, Kong D, Yu F (2017b) MicroRNA-98 plays a suppressive role in non-small cell lung cancer through inhibition of SALL4 protein expression. Oncol Res Featur Preclin Clin Cancer Therap 25:975–988
Lu CW, Zhou DD, Xie T, Hao JL, Pant OP, Lu CB, Liu XF (2018) HOXA 11 antisense long noncoding RNA (HOXA 11-AS): a promising lnc RNA in human cancers. Cancer Med 7:3792–3799
Lu KH, Wei L, Liu XH, Ming S, Zhang ML, Wu WQ, Xie WP, Hou YY (2013) Long non-coding RNA MEG3 inhibits NSCLC cells proliferation and induces apoptosis by affecting p53 expression. BMC Cancer 13:461–461
Nie W, Ge H-j, Yang X-q, Sun X, Huang H, Tao X, Chen W-s, Li B (2016) LncRNA-UCA1 exerts oncogenic functions in non-small cell lung cancer by targeting miR-193a-3p. Cancer Lett 371:99–106
Niu X, Liu S, Jia L, Chen J (2015) Role of MiR-3619-5p in β-catenin-mediated non-small cell lung cancer growth and invasion. Cell Physiol Biochem 37:1527–1536
Prensner JR, Chinnaiyan AM (2011) The emergence of lncRNAs in cancer biology. Cancer Discov 1:391
Qiu M, Xu Y, Yang X, Wang J, Hu J, Xu L, Yin R (2014) CCAT2 is a lung adenocarcinoma-specific long non-coding RNA and promotes invasion of non-small cell lung cancer. Tumor Biology 35:5375–5380
Salmena L, Poliseno L, Tay Y, Kats L, Pandolfi PP (2011) A ceRNA hypothesis: the Rosetta stone of a hidden RNA language? Cell 146:353–358
Siegel R, DeSantis C, Virgo K, Stein K, Mariotto A, Smith T, Cooper D, Gansler T, Lerro C, Fedewa S (2014) Cancer treatment and survivorship statistics, 2012. CA Cancer J Clin 64:252–271
Sun M, Nie F, Wang Y, Zhang Z, Hou J, He D, Xie M, Xu L, De W, Wang Z (2016) LncRNA HOXA11-AS promotes proliferation and invasion of gastric cancer by scaffolding the chromatin modification factors PRC2, LSD1, and DNMT1. Cancer Res 76:6299–6310
Tan A, Li Q, Chen L (2019) CircZFR promotes hepatocellular carcinoma progression through regulating miR-3619–5p/CTNNB1 axis and activating Wnt/β-catenin pathway. Arch Biochem Biophys 661:196–202
Tay Y, Rinn J, Pandolfi PP (2014) The multilayered complexity of ceRNA crosstalk and competition. Nature 505:344–352
Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A (2015) Global cancer statistics, 2012. CA Cancer J Clin 65:87–108
Wan L, Sun M, Liu GJ, Wei CC, Zhang EB, Kong R, Xu TP, Huang MD, Wang ZX (2016) Long noncoding RNA PVT1 promotes non-small cell lung cancer cell proliferation through epigenetically regulating LATS2 expression. Mol Cancer Ther 15:1082–1094
Wang Q, Zhang J, Liu Y, Zhang W, Zhou J, Duan R, Pu P, Kang C, Han L (2016) A novel cell cycle-associated lncRNA, HOXA11-AS, is transcribed from the 5-prime end of the HOXA transcript and is a biomarker of progression in glioma. Cancer Lett 373:251–259
Yan G, Su Y, Ma Z, Yu L, Chen N (2019) Long noncoding RNA LINC00202 promotes tumor progression by sponging miR-3619-5p in retinoblastoma. Cell Struct Funct 44:51–60
Yang J, Lin J, Liu T, Chen T, Pan S, Huang W, Li S (2014) Analysis of lncRNA expression profiles in non-small cell lung cancers (NSCLC) and their clinical subtypes. Lung Cancer 85:110–115
Yuan X, Zhang X, Zhang W, Liang W, Xu W (2016) SALL4 promotes gastric cancer progression through activating CD44 expression. Oncogenesis 5:e268
Zhang Q, Miao S, Han X, Li C, Zhang M, Cui K, Xiong T, Chen Z, Wang C, Xu H (2018) MicroRNA-3619-5p suppresses bladder carcinoma progression by directly targeting β-catenin and CDK2 and activating p21. Cell Death Dis 9:960
Zhang Y, He R-q, Dang Y-w, Zhang X-l, Wang X, Huang S-n, Huang W-t, Jiang M-t, Gan X-n, Xie Y (2016) Comprehensive analysis of the long noncoding RNA HOXA11-AS gene interaction regulatory network in NSCLC cells. Cancer Cell Int 16:89
Zhang Y, Yuan Y, Li Y, Zhang P, Chen P, Sun S (2019) An inverse interaction between HOXA11 and HOXA11-AS is associated with cisplatin resistance in lung adenocarcinoma. Epigenetics 14:949–960
Funding
The study was completed by the author at his own expense.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interests
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Xia, H., Niu, Q., Ding, Y. et al. Long noncoding HOXA11-AS knockdown suppresses the progression of non-small cell lung cancer by regulating miR-3619-5p/SALL4 axis. J Mol Histol 52, 729–740 (2021). https://doi.org/10.1007/s10735-021-09981-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10735-021-09981-1