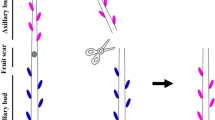
Abstract
Shoot branching is essential in ornamental chrysanthemum production and determines final plant shape and quality. Auxin is associated with apical dominance to indirectly inhibit bud outgrowth. Two non-mutually exclusive models exist for indirect auxin inhibition. Basipetal auxin transport inhibits axillary bud outgrowth by limiting auxin export from buds to stem (canalization model) or by increasing strigolactone levels (second messenger model). Here we analyzed bud outgrowth in treatments with auxin (IAA), strigolactone (GR24) and auxin transport inhibitor (NPA) using a split-plate bioassay with isolated chrysanthemum stem segments. Besides measuring bud length, dividing cell percentage was measured with flow cytometry and RT-qPCR was used to monitor expression levels of genes involved in auxin transport (CmPIN1) and signaling (CmAXR2), bud dormancy (CmBRC1, CmDRM1) and strigolactone biosynthesis (CmMAX1, CmMAX3). Treatments over a 5-day period showed bud outgrowth in the control and inhibition with IAA and IAA + GR24. Bud outgrowth in the control coincided with high dividing cell percentage, decreased expression of CmBRC1 and CmDRM1 and increased CmPIN1 expression. Inhibition by IAA and IAA + GR24 coincided with low dividing cell percentage and unchanged or increased expressions of CmBRC1, CmDRM1 and CmPIN1. Treatment with GR24 showed restricted bud outgrowth that was counteracted by NPA. This restricted bud outgrowth was still concomitant with a high dividing cell percentage and coincided with decreased expression of dormancy genes. These results indicate incomplete inhibition of bud outgrowth by GR24 treatment and suggest involvement of auxin transport in the mechanism of bud inhibition by strigolactones, supporting the canalization model.






Similar content being viewed by others
References
Abel S, Theologis A (1996) Early genes and auxin action. Plant Physiol 111:9–17. https://doi.org/10.1104/pp.111.1.9
Aguilar-Martínez JA, Poza-Carrión C, Cubas P (2007) Arabidopsis BRANCHED1 acts as an integrator of branching signals within axillary buds. Plant Cell 19:458–472. https://doi.org/10.1105/tpc.106.048934
Anderson NO (2007) Flower breeding and genetics issues, challenges and opportunities for the 21st century. Springer, Dordrecht
Arite T, Iwata H, Ohshima K, Maekawa M, Nakajima M, Kojima M, Sakakibara H, Kyozuka J (2007) DWARF10, an RMS1/MAX4/DAD1 ortholog, controls lateral bud outgrowth in rice. Plant J 51:1019–1029. https://doi.org/10.1111/j.1365-313X.2007.03210.x
Balla J, Kalousek P, Reinohl V, Friml J, Prochazka S (2011) Competitive canalization of PIN-dependent auxin flow from axillary buds controls pea bud outgrowth. Plant J 65:571–577. https://doi.org/10.1111/j.1365-313X.2010.04443.x
Barbier F, Peron T, Lecerf M, Perez-Garcia M-D, Barriere Q, Rolcik J, Boutet-Mercey S, Citerne S, Lemoine R, Porcheron B, Roman H, Leduc N, Le Gourrierec J, Bertheloot J, Sakr S (2015) Sucrose is an early modulator of the key hormonal mechanisms controlling bud outgrowth in Rosa hybrida. J Exp Bot 66:2569–2582. https://doi.org/10.1093/jxb/erv047
Bennett T, Leyser O (2006) Something on the side: axillary meristems and plant development. Plant Mol Biol 60:843–854. https://doi.org/10.1007/s11103-005-2763-4
Bennett T, Sieberer T, Willett B, Booker J, Luschnig C, Leyser O (2006) The Arabidopsis MAX pathway controls shoot branching by regulating auxin transport. Curr Biol 16:553–563. https://doi.org/10.1016/j.cub.2006.01.058
Bennett T, Hines G, van Rongen M, Waldie T, Sawchuk MG, Scarpella E, Ljung K, Leyser O (2016) Connective auxin transport in the shoot facilitates communication between shoot apices. PLoS Biol 14:e1002446. https://doi.org/10.1371/journal.pbio.1002446
Booker J, Auldridge M, Willis S, McCarty D, Klee H, Leyser O (2004) MAX3/CCD7 is a carotenoid cleavage dioxygenase required for the synthesis of a novel plant signaling molecule. Curr Biol 14:1232–1238. https://doi.org/10.1016/j.cub.2004.06.061
Brewer PB, Dun E, Ferguson BJ, Rameau C, Beveridge C (2009) Strigolactone acts downstream of auxin to regulate bud outgrowth in pea and Arabidopsis. Plant Physiol 150:482–493. https://doi.org/10.1104/pp.108.134783
Brewer PB, Dun EA, Gui R, Mason M, Beveridge CA (2015) Strigolactone inhibition of branching independent of polar auxin transport. Plant Physiol. https://doi.org/10.1104/pp.15.00014
Chen X, Zhou X, Xi L, Li J, Zhao R, Ma N, Zhao L (2013) Roles of DgBRC1 in regulation of lateral branching in chrysanthemum (Dendranthema × grandiflora cv. Jinba). PLoS ONE 8:e61717. https://doi.org/10.1371/journal.pone.0061717
Crawford S, Shinohara N, Sieberer T, Williamson L, George G, Hepworth J, Müller D, Domagalska M, Leyser O (2010) Strigolactones enhance competition between shoot branches by dampening auxin transport. Development 137:2905–2913. https://doi.org/10.1242/dev.051987
De Keyser E, Desmet L, Van Bockstaele E, De Riek J (2013) How to perform RT-qPCR accurately in plant species? A case study on flower colour gene expression in an azalea (Rhododendron simsii hybrids) mapping population. BMC Mol Biol 14:13. https://doi.org/10.1186/1471-2199-14-13
Devitt ML, Stafstrom JP (1995) Cell cycle regulation during growth-dormancy cycles in pea axillary buds. Plant Mol Biol 29:255–265. https://doi.org/10.1007/BF00043650
Dierck R, De Keyser E, De Riek J, Dhooghe E, Van Huylenbroeck J, Prinsen E, Van Der Straeten D (2016a) Change in auxin and cytokinin levels coincides with altered expression of branching genes during axillary bud outgrowth in chrysanthemum. PLoS ONE 11:e0161732. https://doi.org/10.1371/journal.pone.0161732
Dierck R, Dhooghe E, Van Huylenbroeck J, De Riek J, De Keyser E, Van Der Straeten D (2016b) Response to strigolactone treatment in chrysanthemum axillary buds is influenced by auxin transport inhibition and sucrose availability. Acta Physiol Plant 38:271. https://doi.org/10.1007/s11738-016-2292-6
Domagalska M, Leyser O (2011) Signal integration in the control of shoot branching. Nat Rev Mol Cell Biol 12:211–221. https://doi.org/10.1038/nrm3088
Dong L, Ishak A, Yu J, Zhao R, Zhao L (2013) Identification and functional analysis of three MAX2 orthologs in chrysanthemum. J Integr Plant Biol 55:434–442. https://doi.org/10.1111/jipb.12028
Dun E, de Saint Germain A, Rameau C, Beveridge C (2012) Antagonistic action of strigolactone and cytokinin in bud outgrowth control. Plant Physiol 158:487–498. https://doi.org/10.1104/pp.111.186783
Dun E, de Saint Germain A, Rameau C, Beveridge CA (2013) Dynamics of strigolactone function and shoot branching responses in Pisum sativum. Mol Plant 6:128–140. https://doi.org/10.1093/mp/sss131
Ferguson BJ, Beveridge C (2009) Roles for auxin, cytokinin, and strigolactone in regulating shoot branching. Plant Physiol 149:1929–1944. https://doi.org/10.1104/pp.109.135475
Finlayson S, Krishnareddy SR, Kebrom TH, Casal JJ (2010) Phytochrome regulation of branching in Arabidopsis. Plant Physiol 152:1914–1927. https://doi.org/10.1104/pp.109.148833
Friml J, Vieten A, Sauer M, Weijers D, Schwarz H, Hamann T, Offringa R, Jürgens G (2003) Efflux-dependent auxin gradients establish the apical-basal axis of Arabidopsis. Nature 426:147–153. https://doi.org/10.1038/nature02085
Galbraith DW, Harkins KR, Maddox JM, Ayres NM, Sharma DP, Firoozabady E (1983) Rapid flow cytometric analysis of the cell cycle in intact plant tissues. Science 220:1049–1051. https://doi.org/10.1126/science.220.4601.1049
Gälweiler L, Guan C, Müller A, Wisman E, Mendgen K, Yephremov A, Palme K (1998) Regulation of polar auxin transport by AtPIN1 in Arabidopsis vascular tissue. Science 282:2226–2230. https://doi.org/10.1126/science.282.5397.2226
Gomez-Roldan V, Fermas S, Brewer PB, Puech-Pagès V, Dun E, Pillot J-P, Letisse F, Matusova R, Danoun S, Portais J-C, Bouwmeester H, Bécard G, Beveridge C, Rameau C, Rochange SF (2008) Strigolactone inhibition of shoot branching. Nature 455:189–194. https://doi.org/10.1038/nature07271
González-Grandío E, Poza-Carrión C, Sorzano COS, Cubas P (2013) BRANCHED1 promotes axillary bud dormancy in response to shade in Arabidopsis. Plant Cell 25:834–850. https://doi.org/10.1105/tpc.112.108480
Hayward A, Stirnberg P, Beveridge C, Leyser O (2009) Interactions between auxin and strigolactone in shoot branching control. Plant Physiol 151:400–412. https://doi.org/10.1104/pp.109.137646
Hellemans J, Mortier G, De Paepe A, Speleman F, Vandesompele J (2007) qBase relative quantification framework and software for management and automated analysis of real-time quantitative PCR data. Genome Biol 8:R19. https://doi.org/10.1186/gb-2007-8-2-r19
Kohlen W, Charnikhova T, Liu Q, Bours R, Domagalska M, Beguerie S, Verstappen F, Leyser O, Bouwmeester H, Ruyter-Spira C (2011) Strigolactones are transported through the xylem and play a key role in shoot architectural response to phosphate deficiency in nonarbuscular mycorrhizal host Arabidopsis. Plant Physiol 155:974–987. https://doi.org/10.1104/pp.110.164640
Kretzschmar T, Kohlen W, Sasse J, Borghi L, Schlegel M, Bachelier JB, Reinhardt D, Bours R, Bouwmeester HJ, Martinoia E (2012) A petunia ABC protein controls strigolactone-dependent symbiotic signalling and branching. Nature 483:341–344. https://doi.org/10.1038/nature10873
Leyser O (2009) The control of shoot branching: an example of plant information processing. Plant Cell Environ 32:694–703. https://doi.org/10.1111/j.1365-3040.2009.01930.x
Liang J, Zhao L, Challis R, Leyser O (2010) Strigolactone regulation of shoot branching in chrysanthemum (Dendranthema grandiflorum). J Exp Bot 61:3069–3078. https://doi.org/10.1093/jxb/erq133
Luypaert G, Witters J, Van Huylenbroeck J, De Clercq P, De Riek J, De Keyser E (2017) Induced expression of selected plant defence related genes in pot azalea, Rhododendron simsii hybrid. Euphytica 213:227. https://doi.org/10.1007/s10681-017-2010-5
Mashiguchi K, Sasaki E, Shimada Y, Nagae M, Ueno K, Nakano T, Yoneyama K, Suzuki Y, Asami T (2009) Feedback-regulation of strigolactone biosynthetic genes and strigolactone-regulated genes in Arabidopsis. Biosci Biotechnol Biochem 73:2460–2465. https://doi.org/10.1271/bbb.90443
Mason MG, Ross JJ, Babst B, Wienclaw BN, Beveridge C (2014) Sugar demand, not auxin, is the initial regulator of apical dominance. Proc Natl Acad Sci. https://doi.org/10.1073/pnas.1322045111
McSteen P, Leyser O (2005) Shoot branching. Annu Rev Plant Biol 56:353–374. https://doi.org/10.1146/annurev.arplant.56.032604.144122
Minakuchi K, Kameoka H, Yasuno N, Umehara M, Luo L, Kobayashi K, Hanada A, Ueno K, Asami T, Yamaguchi S, Kyozuka J (2010) FINE CULM1 (FC1) works downstream of strigolactones to inhibit the outgrowth of axillary buds in rice. Plant Cell Physiol 51:1127–1135. https://doi.org/10.1093/pcp/pcq083
Nagpal P, Walker LM, Young JC, Sonawala A, Timpte C, Estelle M, Reed JW (2000) AXR2 encodes a member of the Aux/IAA protein family. Plant Physiol 123:563–574. https://doi.org/10.1104/pp.123.2.563
Ongaro V, Leyser O (2008) Hormonal control of shoot branching. J Exp Bot 59:67–74. https://doi.org/10.1093/jxb/erm134
Otto F (1990) DAPI staining of fixed cells for high-resolution flow cytometry of nuclear DNA. Methods Cell Biol 33:105–110
Prusinkiewicz P, Crawford S, Smith RS, Ljung K, Bennett T, Ongaro V, Leyser O (2009) Control of bud activation by an auxin transport switch. Proc Natl Acad Sci USA 106:17431–17436. https://doi.org/10.1073/pnas.0906696106
Rae GM, David K, Wood M (2013) The dormancy marker DRM1/ARP associated with dormancy but a broader role in planta. Dev Biol J 2013:12. https://doi.org/10.1155/2013/632524
Ramakers C, Ruijter JM, Lekanne Deprez RH, Moorman AFM (2003) Assumption-free analysis of quantitative real-time polymerase chain reaction (PCR) data. Neurosci Lett 339:62–66. https://doi.org/10.1016/S0304-3940(02)01423-4
Rameau C, Bertheloot J, Leduc N, Andrieu B, Foucher F, Sakr S (2015) Multiple pathways regulate shoot branching. Front Plant Sci 5:1–15. https://doi.org/10.3389/fpls.2014.00741
Ruijter JM, Ramakers C, Hoogaars WMH, Karlen Y, Bakker O, van den Hoff MJB, Moorman AFM (2009) Amplification efficiency: linking baseline and bias in the analysis of quantitative PCR data. Nucleic Acids Res 37:e45. https://doi.org/10.1093/nar/gkp045
Sauer M, Balla J, Luschnig C, Wis J, Reinöhl V, Benková E (2006) Canalization of auxin flow by Aux/IAA-ARF-dependent feedback regulation of PIN polarity. Genes Dev 20:2902–2911. https://doi.org/10.1101/gad.390806
Shinohara N, Taylor C, Leyser O (2013) Strigolactone can promote or inhibit shoot branching by triggering rapid depletion of the auxin efflux protein PIN1 from the plasma membrane. PLoS Biol 11:e1001474. https://doi.org/10.1371/journal.pbio.1001474
Shinoyama H, Anderson N, Furuta H, Mochizuki A, Nomura Y, Singh R, Datta SK, Wang B, da Silva J (2006) Chrysanthemum biotechnology. In: Teixeira da Silva JA (ed) Floriculture, ornamental and plant biotechnology: advances and topical issues, vol II. Global Science Books
Umehara M, Hanada A, Yoshida S, Akiyama K, Arite T, Takeda-Kamiya N, Magome H, Kamiya Y, Shirasu K, Yoneyama K, Kyozuka J, Yamaguchi S (2008) Inhibition of shoot branching by new terpenoid plant hormones. Nature 455:195–200. https://doi.org/10.1038/nature07272
Vandesompele J, De Preter K, Pattyn F, Poppe B, Van Roy N, De Paepe A, Speleman F (2002) Accurate normalization of real-time quantitative RT-PCR data by geometric averaging of multiple internal control genes. Genome Biol 3:RESEARCH0034
Vieten A, Vanneste S, Wisniewska J, Benková E, Benjamins R, Beeckman T, Luschnig C, Friml J (2005) Functional redundancy of PIN proteins is accompanied by auxin-dependent cross-regulation of PIN expression. Development 132:4521–4531. https://doi.org/10.1242/dev.02027
Waldie T, McCulloch H, Leyser O (2014) Strigolactones and the control of plant development: lessons from shoot branching. Plant J. https://doi.org/10.1111/tpj.12488
Wen C, Xi L, Gao B, Wang K, Lv S, Kou Y, Ma N, Zhao L (2015) Roles of DgD14 in regulation of shoot branching in chrysanthemum (Dendranthema grandiflorum “Jinba”). Plant Physiol Biochem 96:241–253. https://doi.org/10.1016/j.plaphy.2015.07.030
Wood M, Rae GM, Wu RM, Walton EF, Xue B, Hellens RP, Uversky VN (2013) Actinidia DRM1—an intrinsically disordered protein whose mRNA expression is inversely correlated with spring budbreak in kiwifruit. PLoS ONE. https://doi.org/10.1371/journal.pone.0057354
Xi L, Wen C, Fang S, Chen X, Nie J, Chu J, Yuan C, Yan C, Ma N, Zhao L (2015) Impacts of strigolactone on shoot branching under phosphate starvation in chrysanthemum (Dendranthema grandiflorum cv. Jinba). Front Plant Sci 6:1–15. https://doi.org/10.3389/fpls.2015.00694
Xie X, Wang G, Yang L, Cheng T, Gao J, Wu Y, Xia Q (2015a) Cloning and characterization of a novel Nicotiana tabacum ABC transporter involved in shoot branching. Physiol Plant 153:299–306. https://doi.org/10.1111/ppl.12267
Xie X, Yoneyama K, Kisugi T, Nomura T, Akiyama K, Asami T, Yoneyama K (2015b) Strigolactones are transported from roots to shoots, although not through the xylem. J Pestic Sci 40:214–216. https://doi.org/10.1584/jpestics.D15-045
Zwanenburg B, Pospísil T (2013) Structure and activity of strigolactones: new plant hormones with a rich future. Mol Plant 6:38–62. https://doi.org/10.1093/mp/sss141
Acknowledgements
This research was funded by the Institute for the Promotion of Innovation through Science and Technology in Flanders (IWT-Vlaanderen Grant No. 110771). The authors would like to thank Magali Losschaert for technical assistance with gene expression analysis and Laurence Desmet, Jorien Oomen, Veerle Buysens, Katrijn Van Laere and Evelien Calsyn for their help in preparing the split-plate assays and sampling of plant tissue. Frederik Delbeke, Roger Dobbelaere and Jo De Groote are acknowledged for maintenance of the plant material in the greenhouse.
Author information
Authors and Affiliations
Contributions
RD conceptualized and performed the experiments and wrote the manuscript. LL performed flow cytometry analysis. EDK, LL, DVDS, ED, JVH and JDR assisted in conceptualizing the experiment and proofread and revised the manuscript.
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Appendix
Appendix
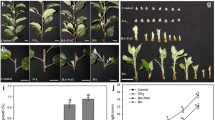
The mitotic index (percentage of dividing cells) is represented by the sum of the amount of cells in the S and G2 phase divided by the total number of nuclei analyzed (G1 + S + G2), as was delineated on a flow-cytometrical histogram. a leaf; b shoot apex; c, d control axillary buds at day 0 (c) and day 2 (d); e–h hormone treated (e IAA, f GR24, g GR24+IAA, h GR24+NPA) axillary buds at D2
Rights and permissions
About this article
Cite this article
Dierck, R., Leus, L., Dhooghe, E. et al. Branching gene expression during chrysanthemum axillary bud outgrowth regulated by strigolactone and auxin transport. Plant Growth Regul 86, 23–36 (2018). https://doi.org/10.1007/s10725-018-0408-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10725-018-0408-2