Abstract
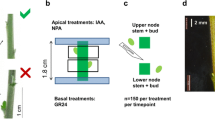
Axillary bud outgrowth is regulated by both environmental cues and internal plant hormone signaling. Central to this regulation is the balance between auxins, cytokinins, and strigolactones. Auxins are transported basipetally and inhibit the axillary bud outgrowth indirectly by either restricting auxin export from the axillary buds to the stem (canalization model) or inducing strigolactone biosynthesis and limiting cytokinin levels (second messenger model). Both models have supporting evidence and are not mutually exclusive. In this study, we used a modified split-plate bioassay to apply different plant growth regulators to isolated stem segments of chrysanthemum and measure their effect on axillary bud growth. Results showed axillary bud outgrowth in the bioassay within 5 days after nodal stem excision. Treatments with apical auxin (IAA) inhibited bud outgrowth which was counteracted by treatments with basal cytokinins (TDZ, zeatin, 2-ip). Treatments with basal strigolactone (GR24) could inhibit axillary bud growth without an apical auxin treatment. GR24 inhibition of axillary buds could be counteracted with auxin transport inhibitors (TIBA and NPA). Treatments with sucrose in the medium resulted in stronger axillary bud growth, which could be inhibited with apical auxin treatment but not with basal strigolactone treatment. These observations provide support for both the canalization model and the second messenger model with, on the one hand, the influence of auxin transport on strigolactone inhibition of axillary buds and, on the other hand, the inhibition of axillary bud growth by strigolactone without an apical auxin source. The inability of GR24 to inhibit bud growth in a sucrose treatment raises an interesting question about the role of strigolactone and sucrose in axillary bud outgrowth and calls for further investigation.






Similar content being viewed by others
References
Barbier F, Peron T, Lecerf M et al (2015) Sucrose is an early modulator of the key hormonal mechanisms controlling bud outgrowth in Rosa hybrida. J Exp Bot 66:2569–2582. doi:10.1093/jxb/erv047
Bennett T, Leyser O (2006) Something on the side: axillary meristems and plant development. Plant Mol Biol 60:843–854. doi:10.1007/s11103-005-2763-4
Beveridge CA, Symons GM, Turnbull CG (2000) Auxin inhibition of decapitation-induced branching is dependent on graft-transmissible signals regulated by genes Rms1 and Rms2. Plant Physiol 123:689–698
Blakeslee JJ, Bandyopadhyay A, Lee OR et al (2007) Interactions among PIN-FORMED and P-glycoprotein auxin transporters in Arabidopsis. Plant Cell 19:131–147. doi:10.1105/tpc.106.040782
Brewer PB, Dun EA, Ferguson BJ et al (2009) Strigolactone acts downstream of auxin to regulate bud outgrowth in pea and Arabidopsis. Plant Physiol 150:482–493. doi:10.1104/pp.108.134783
Brewer PB, Dun EA, Gui R et al (2015) Strigolactone inhibition of branching independent of polar auxin transport. Plant Physiol. doi:10.1104/pp.15.00014
Chatfield SP, Stirnberg P, Forde BG, Leyser O (2000) The hormonal regulation of axillary bud growth in Arabidopsis. Plant J 24:159–169
Chen X, Zhou X, Xi L et al (2013) Roles of DgBRC1 in regulation of lateral branching in chrysanthemum (Dendranthema × grandiflora cv. Jinba). PLoS One 8:e61717. doi:10.1371/journal.pone.0061717
Cline MG (1991) Apical dominance. Bot Rev 57:318–358. doi:10.1007/BF02858771
Crawford S, Shinohara N, Sieberer T et al (2010) Strigolactones enhance competition between shoot branches by dampening auxin transport. Development 137:2905–2913. doi:10.1242/dev.051987
Domagalska MA, Leyser O (2011) Signal integration in the control of shoot branching. Nat Rev Mol Cell Biol 12:211–221. doi:10.1038/nrm3088
Dun EA, de Saint Germain A, Rameau C, Beveridge CA (2013) Dynamics of strigolactone function and shoot branching responses in Pisum sativum. Mol Plant 6:128–140. doi:10.1093/mp/sss131
Foo E, Bullier E, Goussot M et al (2005) The branching gene RAMOSUS1 mediates interactions among two novel signals and auxin in pea. Plant Cell 17:464–474. doi:10.1105/tpc.104.026716.1
Foo E, Morris SE, Parmenter K et al (2007) Feedback regulation of xylem cytokinin content is conserved in pea and Arabidopsis. Plant Physiol 143:1418–1428. doi:10.1104/pp.106.093708
Friml J, Vieten A, Sauer M et al (2003) Efflux-dependent auxin gradients establish the apical-basal axis of Arabidopsis. Nature 426:147–153. doi:10.1038/nature02085
Gomez-Roldan V, Fermas S, Brewer PB et al (2008) Strigolactone inhibition of shoot branching. Nature 455:189–194. doi:10.1038/nature07271
Katekar GF, Geissler AE (1977) Auxin transport inhibitors. Plant Physiol 60:826–829
Kebrom TH, Mullet JE (2015) Photosynthetic leaf area modulates tiller bud outgrowth in sorghum. Plant Cell Environ n/a n/a. doi:10.1111/pce.12500
Kebrom TH, Brutnell TP, Finlayson SA (2010) Suppression of sorghum axillary bud outgrowth by shade, phyB and defoliation signalling pathways. Plant, Cell Environ 33:48–58. doi:10.1111/j.1365-3040.2009.02050.x
Kushwah S, Laxmi A (2014) The interaction between glucose and cytokinin signal transduction pathway in Arabidopsis thaliana. Plant Cell Environ 37:235–253. doi:10.1111/pce.12149
Liang J, Zhao L, Challis R, Leyser O (2010) Strigolactone regulation of shoot branching in chrysanthemum (Dendranthema grandiflorum). J Exp Bot 61:3069–3078. doi:10.1093/jxb/erq133
Ljung K, Bhalerao RP, Sandberg G (2001) Sites and homeostatic control of auxin biosynthesis in Arabidopsis during vegetative growth. Plant J 28:465–474. doi:10.1046/j.1365-313X.2001.01173.x
Mashiguchi K, Sasaki E, Shimada Y et al (2009) Feedback-regulation of strigolactone biosynthetic genes and strigolactone-regulated genes in Arabidopsis. Biosci Biotechnol Biochem 73:2460–2465. doi:10.1271/bbb.90443
Mason MG, Ross JJ, Babst BA et al (2014) Sugar demand, not auxin, is the initial regulator of apical dominance. Proc Natl Acad Sci. doi:10.1073/pnas.1322045111
Mishra BS, Singh M, Aggrawal P, Laxmi A (2009) Glucose and auxin signaling interaction in controlling Arabidopsis thaliana seedlings root growth and development. PLoS One 4:e4502. doi:10.1371/journal.pone.0004502
Morris SE, Cox MCH, Ross JJ et al (2005) Auxin dynamics after decapitation are not correlated with the initial growth of axillary buds. Plant Physiol 138:1665–1672. doi:10.1104/pp.104.058743
Ongaro V, Leyser O (2008) Hormonal control of shoot branching. J Exp Bot 59:67–74. doi:10.1093/jxb/erm134
Petrásek J, Friml J (2009) Auxin transport routes in plant development. Development 136:2675–2688. doi:10.1242/dev.030353
Petrásek J, Mravec J, Bouchard R et al (2006) PIN proteins perform a rate-limiting function in cellular auxin efflux. Science 312:914–917
Rabot A, Henry C, Ben Baaziz K et al (2012) Insight into the Role of Sugars in Bud Burst Under Light in the Rose. Plant Cell Physiol 53:1068–1082. doi:10.1093/pcp/pcs051
Rameau C, Bertheloot J, Leduc N et al (2015) Multiple pathways regulate shoot branching. Front Plant Sci 5:1–15. doi:10.3389/fpls.2014.00741
Sachs T, Thimann KV (1967) The role of auxins and cytokinins in the release of buds from dominance. Am J Bot 54:136–144
Shinohara N, Taylor C, Leyser O (2013) Strigolactone can promote or inhibit shoot branching by triggering rapid depletion of the auxin efflux protein PIN1 from the plasma membrane. PLoS Biol 11:e1001474. doi:10.1371/journal.pbio.1001474
Tanaka M, Takei K, Kojima M et al (2006) Auxin controls local cytokinin biosynthesis in the nodal stem in apical dominance. Plant J 45:1028–1036. doi:10.1111/j.1365-313X.2006.02656.x
Van Minnebruggen A, Roldan-Ruiz I, Van Dingenen J et al (2013) Morphological and molecular characterization of branching in red clover (Trifolium pratense). In: Barth S, Milbourne D (eds) Breeding strategies for sustainable forage and turf grass improvement SE—20. Springer, Netherlands, pp 161–167
Waldie T, McCulloch H, Leyser O (2014) Strigolactones and the control of plant development: lessons from shoot branching. Plant J 79:607–622. doi:10.1111/tpj.12488
Young NF, Ferguson BJ, Antoniadi I et al (2014) Conditional auxin response and differential cytokinin profiles in shoot branching mutants. Plant Physiol 165:1723–1736. doi:10.1104/pp.114.239996
Acknowledgements
This research was funded by the Institute for the Promotion of Innovation through Science and Technology in Flanders (IWT-Vlaanderen Grant No. 110771). The authors wish to thank Annemie Van Minnebruggen and Gerda Cnops for demonstrating the split-plate assay in red clover. DVDS acknowledges Ghent University for financial support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by H. Li.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Dierck, R., Dhooghe, E., Van Huylenbroeck, J. et al. Response to strigolactone treatment in chrysanthemum axillary buds is influenced by auxin transport inhibition and sucrose availability. Acta Physiol Plant 38, 271 (2016). https://doi.org/10.1007/s11738-016-2292-6
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11738-016-2292-6