Abstract
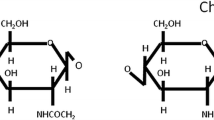
The swim bladder of fish is an internal gas-filled organ that allows fish to control their buoyancy and swimming depth. Fish maws (the dried swim bladders of fish) have been used over many centuries as traditional medicines, tonics and a luxurious gourmet food in China and Southeast Asia. Little is known about the structural information of polysaccharides comprising this important functional material of fish tissue. In the present study, the total glycosaminoglycan (GAG) from fish maw was characterized. Two GAGs were identified, chondroitin sulfate (CS, having a molecular weight of 18–40 kDa) and heparan sulfate (HS), corresponding to 95% and 5% of the total GAG, respectively. Chondroitinase digestion showed that the major CS GAG was composed of ΔUA-1 → 3-GalNAc4S (59.7%), ΔUA-1 → 3-GalNAc4,6S (36.5%), ΔUA-1 → 3-GalNAc6S (2.2%) and ΔUA-1 → 3-GalNAc (1.6%) disaccharide units. 1H–NMR analysis and degradation with specific chondroitinases, both CS-type A/C and CS-type B were present in a ratio of 1.4:1. Analysis using surface plasmon resonance showed that fibroblast growth factor (FGF)-2 bound to the CS fraction (KD = 136 nM). These results suggest that this CS may be involved in FGF-signal pathway, mediating tissue repair, regeneration and wound healing. The CS, as the major GAG in fish maw, may have potential pharmacological activity in accelerating wound healing.







Similar content being viewed by others
Abbreviations
- Ac:
-
acetyl
- AMAC:
-
2-aminoacridone
- CS:
-
chondroitin sulfate
- ΔUA:
-
4-deoxy-β-L-threo-hex-4-enopyranosiduronic acid
- DMMB:
-
1,9-dimethylmethylene blue
- ESI:
-
electrospray ionization;
- FGF:
-
fibroblast growth factor
- GAG:
-
glycosaminoglycan
- GalN:
-
galactosamine
- GlcA:
-
glucuronic acid
- GlcN:
-
glucosamine
- GPC:
-
gel permeation chromatography
- HPLC:
-
high performance liquid chromatography
- HS:
-
heparan sulfate
- IdoA:
-
iduronic acid
- KS:
-
keratan sulfate
- MS:
-
mass spectrometry
- MWCO:
-
molecular weight cut-off
- NMR:
-
nuclear magnetic resonance
- PAGE:
-
polyacrylamide gel electrophoresis
- RU:
-
resonance units
- S:
-
sulfo
- SEC:
-
size exclusion chromatography
- SPR:
-
surface plasmon resonance
- USP:
-
United States Pharmacopeia
References
Clarke, S.C.: Understanding pressures onfishery resources through trade statistics: a pilot study of four products in the Chinese dried seafood market. Fish Fish. 5, 53–74 (2004)
Lin, S.Y.: Fish air-bladders of commercial value in China. The Hong Kong Naturalist. 9, 108–118 (1939)
Sadovy, Y., Cheung, W.L.: Near extinction of a highly fecundfish: the one that nearly got away. Fish Fish. 4, 86–99 (2003)
Jian, J.C., ZH, W.: Effects of traditional Chinese medicine on nonspecific immunity and disease resistance of large yellow croaker, Pseudosciaena Crocea (Richardson). Aquaculture. 218, 1–9 (2003)
Li, C., Yao, C.L.: Molecular and expression characterizations of interleukin-8 gene in large yellow croaker (Larimichthys crocea). Fish. Shellfish. Immunol. 34, 799–809 (2013)
Li, G.J., et al.: Preventive effect of polysaccharide of Larimichthys Crocea swimming bladder on activated carbon-induced constipation in mice. J Korean Soc Appl Biol Chem. 57(2), 167–172 (2013)
Jiang, X., et al.: Therapeutic effect of polysaccharide of large yellow croaker swim bladder on lupus nephritis of mice. Nutrients. 6(3), 1223–1235 (2014)
Chen, S., et al.: Preventive effect of polysaccharides from the large yellow croaker swim bladder on HCl/ethanol induced gastric injury in mice. Exp. Ther. Med. 8(1), 316–322 (2014)
Capila, I., Linhardt, R.J.: Heparin-Protein Interactions. AngewandteChemie Int. Ed. 41, 390–412 (2002)
Pomin, V.H., Mulloy, B.: Current structural biology of the heparin interactome. Curr. Opin. Struct. Biol. 34, 17–25 (2015)
Munoz, E.M., Linhardt, R.J.: Heparin-binding domains in vascular biology. Arterioscler. Thromb. Vasc. Biol. 24, 1549–1557 (2004)
Belting, M.: Glycosaminoglycans in cancer treatment. Thromb. Res. 133(Suppl2), 95–101 (2014)
Sterner, E., et al.: FGF-FGFR signaling mediated through glycosaminoglycans in microfile plate and cell-based microarray assays. Biochemistry. 52, 9–19 (2013)
Kamhi, E., Joo, E.J., Dordick, J.S., Linhardt, R.J.: Glycosaminoglycans in infectious disease. Biol. Rev. 88, 928–943 (2013)
Shute, J.: Glycosaminoglycan and chemokine/growth factor interactions. Handb. Exp. Pharmacol. 207, 307–324 (2012)
Linhardt, R.J.: Claude S Hudson award address in carbohydrate chemistry. Heparin: structure and activity. J. Med. Chem. 46(13), 2551–2564 (2003)
Yamada, S., Sugahara, K.: Potential therapeutic application of chondroitin sulfate/ dermatan sulfate. Curr. Drug Disc. Technol. 5, 289–301 (2008)
Goldberg, V.M., Buckwalter, J.A.: Hyaluronans in the treatment of osteoarthritis of the knee: evidence for disease-modifying activity. Osteoarthr. Cartil. 13, 216–224 (2005)
Vázquez, J.A., Rodríguez-Amado, I., Montemayor, M., Fraguas, J., González, M.P., Murado, M.A.: Chondroitin sulfate, hyaluronic acid and chitin/chitosan production using marine waste sources: characteristics, applications and eco-friendly processes: a review. Mar. Drugs. 11, 747–774 (2013)
Sasisekharan, R., Bulmer, M., Moremen, K.W., Cooney, C.L., Langer, R.: Cloning and expression of heparinase I gene from Flavobacterium heparinum. Proc. Natl. Acad. Sci. U. S. A. 1993(90), 3660–3664 (1993)
Williams, A., He, W., Cress, B.F., Liu, X., Alexandria, J., Yoshizawa, H., Nishimura, K., Toida, T., Koffas, M., Linhardt, R.J.: Cloning and expression of recombinant chondroitinase AC II and its comparison to the Arthrobacter aurescens enzyme. Biotechnol. J. (2017). https://doi.org/10.1002/biot.201700239
Bitter, T., Muir, H.M.: A modiWed uronic acid carbazole reaction. Anal. Biochem. 4, 330–334 (1962)
Yang, B., Chang, Y., Weyers, A.M., Sterner, E., Linhardt, R.J.: Disaccharide analysis of glycosaminoglycan mixtures by ultra-high-performance liquid chromatography-mass spectrometry. J Chromatogr A. 1225, 91–98 (2012)
Liu, Z., Masuko, S., Solakyildirim, K., Pu, D., Linhardt, R.J., Zhang, F.: Glycosaminoglycans of the porcine central nervous system. Biochemistry. 49, 9839–9847 (2010)
Im, A.R., Kim, J.Y., Kim, H.S., Cho, S., Park, Y., Kim, Y.S.: Wound healing and antibacterial activities of chondroitin sulfate- and acharan sulfate-reduced silver nanoparticles. Nanotechnology. (2013). https://doi.org/10.1088/0957-4484/24/39/395102
Zou, X.H., Foong, W.C., Cao, T., Bay, B.H., Ouyang, H.W., Yip, G.W.: Chondroitin sulfate in palatal wound healing. J. Dent. Res. 83, 880–885 (2004)
Shi, H.X., Lin, C., Lin, B.B., Wang, Z.G., Zhang, H.Y., FZ, W., Cheng, Y., Xiang, L.J., Guo, D.J., Luo, X., Zhang, G.Y., XB, F., Bellusci, S., Li, X.K., Xiao, J.: The anti-scar effects of basic fibroblast growth factor on the wound repair in vitro and in vivo. PLoS One. 8, e59966 (2013)
Acknowledgments
This work was supported by National Institutes of Health Grants HL125371, GM38060, HL096972, HL062244 and HL136271.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflicts of interest
The authors declare that they have no conflicts of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Rights and permissions
About this article
Cite this article
Pan, Y., Wang, P., Zhang, F. et al. Glycosaminoglycans from fish swim bladder: isolation, structural characterization and bioactive potential. Glycoconj J 35, 87–94 (2018). https://doi.org/10.1007/s10719-017-9804-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10719-017-9804-5