Abstract
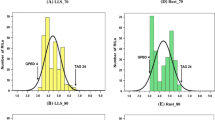
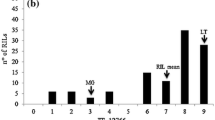
Forage sorghum cultivars grown in India are susceptible to various foliar diseases, of which anthracnose, rust, zonate leaf spot, drechslera leaf blight and target leaf spot cause severe damage. We report here the quantitative trait loci (QTLs) conferring resistance to these foliar diseases. QTL analysis was undertaken using 168 F7 recombinant inbred lines (RILs) of a cross between a female parental line 296B (resistant) and a germplasm accession IS18551 (susceptible). RILs and parents were evaluated in replicated field trials in two environments. A total of twelve QTLs for five foliar diseases on three sorghum linkage groups (SBI-03, SBI-04 and SBI-06) were detected, accounting for 6.9–44.9% phenotypic variance. The morphological marker Plant color (Plcor) was associated with most of the QTL across years and locations. The QTL information generated in this study will aid in the transfer of foliar disease resistance into elite susceptible sorghum breeding lines through marker-assisted selection.


Similar content being viewed by others
References
Afanasenko OS, Makarova IG, Zubkovich AA (1999) The number of genes controlling resistance to Pyrenophora teres Drechs. Strains in barley. Russ J Genet 35:274–283
Agrama HA, Houssin SF, Tarek MA (2002) Cloning of AFLP markers linked to resistance to Peronosclerospora sorghi in maize. Mol Genet Genomics 267:814–819
Anahosur KH (1992) Sorghum diseases in India: knowledge and research needs. In: De Milliano WAJ, Frederiksen RA, Bengston GD (eds) Sorghum and millet diseases: a second world review. ICRISAT, Patancheru, pp 45–56
Bolwell GP (1999) Role of active oxygen species and NO in plant defense responses. Curr Opin Plant Biol 2:287–294
Boora KS, Frederiksen RA, Magill CW (1999) A molecular marker that segregates with sorghum leaf blight resistance in one cross is maternally inherited in another. Mol Gen Genet 261:317–322
Brader G, Djamei A, Teige M, Palva T, Hirt H (2007) The MAP Kinase MKK2 affects disease resistance in Arabidopsis. Mol Plant Microbe Interact 20:589–596
Bres-Patry C, Lorieux M, Clement G, Bangratz M, Ghesquiere A (2001) Heredity and genetic mapping of domestication-related traits in a temperate japonica weedy rice. Theor Appl Genet 102:118–126
Browder LE (1985) Parasite: host: environment specificity in the cereal rusts. Annu Rev Phytopathol 23:201–222
Bunker RN, Mathur K (2006) Pathogenic variations in sorghum leaf blight pathogen Exserohilum turcicum in India. Intl Sorghum Millets Newsl 47:58–59
Cai HW, Morishima H (2002) QTL clusters reflect character associations in wild and cultivated rice. Theor Appl Genet 104:1217–1228
Cakir M, Gupta S, Platz GJ, Ablett GA, Loughman R, Embiri LC, Poulsen D, Li CD, Lance RCM, Galwey NW, Jones MGK, Appels R (2003) Mapping and validation of the genes for resistance to Pyrenophora teres f. teres in barley (Hordeum vulgare L.). Aust J Agric Res 54:1369–1377
Chang SW, Hwang BK (2003) Effects of plant age, leaf position, inoculum density, and wetness period on Bipolaris coicis infection on adults differing for resistance. Plant Dis 87:821–826
Dalmacio SC (2000) Target leaf spot. In: Frederiksen RA, Odvody GN (eds) Compendium of sorghum diseases, 2nd edn. The American Phytopathological Society/APS Press, St Pauls, pp 16–17
De Vicente C, Metz T, Alercia A (2004) Descriptors for genetic markers technologies. IPGRI, Rome
Doggett H (1988) Sorghum, 2nd edn. Wiley, New York
Ellis J, Dodds P, Pryor T (2000) Structure, function and evolution of plant disease resistance genes. Curr Opin Plant Biol 3:278–284
Frederiksen RA (2000) Diseases and disease management in sorghum. In: Smith WC, Frederiksen RA (eds) Sorghum: origin, history, technology and production. Wiley, New York, pp 497–533
Fritz AK, Reddy AS, Pammi S, Ayres NM (1995) Silver staining as a low-cost, non-radioactive method of detecting PCR products. In: Agronomy abstracts. ASA, Madison, p 184
Gebhardt C, Valkonen JPT (2001) Organization of genes controlling disease resistance in the potato genome. Annu Rev Phytopathol 39:79–102
Geiger HH, Heun M (1989) Genetics of quantitative resistance to fungal diseases. Annu Rev Phytopathol 27:317–341
Gowda PSB, Xu GW, Frederiksen RA, Magill CW (1995) DNA markers for downy mildew resistance genes in sorghum. Genome 38:823–826
Grewal RPS (1988) Genetic basis of resistance to zonate leaf spot disease in forage sorghum. Theor Appl Genet 76:550–554
Islam MR, Mayo G (1990) Compendium on host genes in flax conferring resistance to flax rust. Plant Breed 104:89–100
Kim JS, Klein PE, Klein RR, Price HJ, Mullet JE, Stelly DM (2005) Chromosome identification and nomenclature of Sorghum bicolor. Genetics 169:1169–1173
Klein RR, Rodriguez-Herrera R, Schlueter JA, Klein PE, Yu ZH, Rooney WL (2001) Identification of genomic regions that affect grain mold incidence and other traits of agronomic importance in sorghum. Theor Appl Genet 102:307–319
Koch MF, Parlevliet JE (1991) Genetic analysis of, and selection for, factors affecting quantitative resistance to Xanthomonas campestris pv oryzae in rice. Euphytica 53:235–245
Kosambi DD (1944) The estimation of map distances from recombination values. Ann Eugen 12:172–175
Lander ES, Botstein D (1989) Mapping Mendelian factors underlying quantitative traits using RFLP linkage maps. Genetics 121:185–199
Leukel RW (1948) Periconia circinata and its relation to milo disease. J Agric Res 77:201–222
Lillemo M, Singh RP, Huerta-Espino J, Chen XM, He ZH, Brown JKM (2007) Leaf rust resistance gene Lr34 is involved in powdery mildew resistance of CIMMYT bread wheat line Saar. In: Buck HT, Nisi JE, Salomon N (eds) Developments in plant breeding, vol 12: wheat productions in stressed environments (Proc 7th Int Wheat Conf). Mar del Plata, pp 97–102
Mathur K, Bunker RN (2002) Drechslera leaf blight of sorghum caused by Dreschslera australiensis—new report from India. Intl Sorghum Millets Newsl 43:60
Mingeot D, Chantret N, Baret PV, Dekeyser A, Boukhatem N, Sourdille P, Doussinault G, Jacquemin JM (2002) Mapping QTL involved in adult plant resistance to powdery mildew in the winter wheat line RE714 in two susceptible genetic backgrounds. Plant Breed 121:133–140
Mode CJ, Robinson HF (1959) Pleiotropism and the genetic variance and covariance. Biometrics 15:518–537
Mohan SM, Madhusudhana R, Mathur K, Howarth CJ, Srinivas G, Satish K, Reddy RN, Seetharama N (2009) Co-localization of quantitative trait loci for foliar disease resistance in sorghum. Plant Breed 128:532–535
Nicholson RL, Kollipara SS, Vincent JR, Lyons PC, Cadena-Gomez G (1987) Phytoalexin synthesis by the sorghum mesocotyl in response to infection by pathogenic and nonpathogenic fungi. Proc Natl Acad Sci USA 84:5520–5524
Oh BJ, Frederiksen RA, Magill CW (1994) Identification of molecular markers linked to head smut resistance gene (Shs) in sorghum by RFLP and RAPD analyses. Phytopathology 84:830–833
Paterson AH, Lander ES, Hewitt JD, Peterson S, Lincoln SE, Tanksley SD (1988) Resolution of quantitative traits into Mendelian factors by using a complete RFLP linkage map. Nature 335:721–726
Pawson T, Scott JD (1997) Signaling through scaffold, anchoring, and adaptor proteins. Science 278:2075–2080
Peng JH, Ronin Y, Fahima T, Röder MS, Li YC, Nevo E, Korol AB (2003) Domestication quantitative trait loci in Triticum dicoccides, the progenitor of wheat. Proc Natl Acad Sci USA 100:2489–2494
Perumal R, Menz MA, Mehta PJ, Katilé S, Gutierrez-Rojas LA, Klein RR, Klein PE, Prom LK, Schlueter JA, Rooney WL, Magill CW (2008) Molecular mapping of Cg1, a gene for resistance to anthracnose (Colletotrichum sublineolum) in sorghum. Euphytica 165:597–606
Portyanko VA, Chen G, Rines HW, Phillips RL, Leonard KJ, Ochocki GE, Stuthman DD (2005) Quantitative trait loci for partial resistance to crown rust, Puccinia coronata, in cultivated oat, Avena sativa L. Theor Appl Genet 111:313–324
Rana BS, Tripathi DP, Rao NG (1976) Genetic analysis of some exotic × Indian crosses in sorghum. XV. Inheritance of resistance to sorghum rust. Indian J Genet Plant Breed 36:244–249
Rana BS, Rao MH, Indira S, Singh BU, Chari Appaji, Tonapi VA (1999) Technology for increasing sorghum production and value addition. National Research Center for Sorghum, Rajendranagar, Hyderabad
Richter K, Schondelmaier J, Jung C (1998) Mapping of quantitative trait loci affecting Drechslera teres resistance in barley with molecular markers. Theor Appl Genet 97:1225–1234
Ryals JA, Neuenschwander UH, Willits MG, Molina A, Steiner HY, Hunt MO (1996) Systemic acquired resistance. Plant Cell 8:1809–1819
Satish K, Srinivas G, Madhusudhana R, Padmaja PG, Nagaraja Reddy R, Murali Mohan S, Seetharama N (2009) Identification of quantitative trait loci for resistance to shoot fly in sorghum [Sorghum bicolor (L.) Moench]. Theor Appl Genet 119:1425–1439
Sharma HC (1980) Screening of sorghum for leaf-disease resistance in India. In: Williams RJ, Frederiksen RA, Mughogho LK (eds) Sorghum diseases: a world review (Proc Int Workshop Sorghum Diseases). ICRISAT, Patancheru, pp 249–264
Sharma KD, Winter P, Kahl G, Muehlbauer J (2004) Molecular mapping of Fusarium oxysporum f. sp. ciceris race 3 resistance gene in chickpea. Theor Appl Genet 108:1243–1248
Singh M, Chaudhary K, Boora KS (2006) RAPD-based SCAR marker SCA 12 linked to recessive gene conferring resistance to anthracnose in sorghum [Sorghum bicolor (L.) Moench]. Theor Appl Genet 114:187–192
Singh RP, Ma H, Rajaram S (1995) Genetic analysis of resistance to scab in spring wheat cultivar Frontana. Plant Dis 79:238–240
Srinivas G, Satish K, Murali Mohan S, Nagaraja Reddy R, Madhusudhana R, Balakrishna D, Venkatesh Bhat B, Howarth CJ, Seetharama N (2008) Development of genic-microsatellite markers for sorghum stay-green QTL using a comparative genomic approach with rice. Theor Appl Genet 117:283–296
Srinivas G, Satish K, Madhusudhana R, Nagaraja Reddy R, Murali Mohan S, Seetharama N (2009) Identification of quantitative trait loci for agronomically important traits and their association with genic-microsatellite markers in sorghum. Theor Appl Genet 118:1439–1454
Stefenson BJ, Hayes PM, Kleinhofs A (1996) Genetics of seedling and adult plant resistance to net blotch (Pyrenophora teres f. teres) and spot blotch (Cochliobolus sativus) in barley. Theor Appl Genet 92:552–558
Stein M, Dittgen J, Sánchez-Rodríguez C, Hou B-H, Molina A, Schulze-Lefert P, Lipka V, Somervillea S (2006) Arabidopsis PEN3/PDR8, an ATP binding cassette transporter, contributes to non-host resistance to inappropriate pathogens that enter by direct penetration. Plant Cell 18:731–746
Szabo A, Korszun R, Hartl FU, Flanagan J (1996) A zinc finger-like domain of the molecular chaperone DnaJ is involved in binding to denatured protein substrates. EMBO J 15:408–417
Tao YZ, Jordan DR, Henzell RG, McIntyre CL (1998) Construction of a genetic map in a sorghum RIL population using probes from different sources and its alignment with other sorghum maps. Aust J Agric Res 49:729–736
Thakur RP, Rao VP, Wu BM, Subbarao KV, Mathur K, Tailor HC, Kushwaha US, Dwivedi RP, Krishnaswamy R, Hiremath RV, Indira S (2007) Genetic resistance to foliar anthracnose in sorghum and pathogenic variability in Colletotrichum graminicola. Indian Phytopathol 60:13–23
Tonapi VA, Dayakar Rao B, Raghavendra Rao KV, Seetharama N (2007) NRCS-perspective Plan—Vision 2025. http://www.sorghum.res.in/vision2025/1b.pdf
Torres-Montalvo H, Mendoza-Onofre L, Gonzalez-Hernandez V, Williams-Alanis H (1992) Reaction of tan and non-tan isogenic genotypes to head blight. Intl Sorghum Millets Newsl 33:36
Umaerus V, Umaerus M (1994) Inheritance of resistance to late blight. In: Bradshaw JE, Mackay GR (eds) Potato genetics. Oxon CAB Int, Wallingford, pp 365–401
Ungerer M, Halldorsdottir S, Modliszewski J, Mackay T, Purugganan M (2002) Quantitative trait loci for inflorescence development in Arabidopsis thaliana. Genetics 160:1133–1151
Upadyayula N, Da Silva HS, Bohn MO, Rocheford TR (2006) Genetic and QTL analysis of maize tassel and ear inflorescence architecture. Theor Appl Genet 112:592–606
Van Ooijen JW (2005) Map-QTL 5: Software for the mapping quantitative trait loci in mapping populations. Kyazma B.V, Wageningen
Van Ooijen JW, Voorrips RE (2001) JOINMAP 3.0. Software for the calculation of genetic linkage maps. Plant Res Int, Wageningen
Vear F, Gentzbittel L, Philippon J, Mouzeyar S, Mestries E, Roeckel-Drevet P, Tourvieille de labrouhe D, Nicolas P (1997) The genetics of resistance to five races of downy mildew (Plasmopara halstedii) in sunflower (Helianthus annuus L.). Theor Appl Genet 95:584–589
Wastie RL (1991) Breeding for resistance. In: Ingram DS, Williams PH (eds) Advances in plant pathology. Academic, London, pp 193–224
Xiong LZ, Liu KD, Dai XK, Xu CG, Zhang Q (1999) Identification of genetic factors controlling domestication-related traits of rice using an F2 population of a cross between Oryza sativa and O. rufipogon. Theor Appl Genet 98:243–251
Yoshimura A, Mew TW, Khush GS, Omura T (1983) Inheritance of resistance to bacterial blight in rice cultivar Cas 209. Phytopathology 73:1409–1411
Young ND (1996) QTL mapping and quantitative disease resistance in plants. Annu Rev Phytopathol 34:479–501
Zhu S, Leonard KJ, Kaeppler HF (2003) Quantitative trait loci associated with seedling resistance to isolates of Puccinia coronata in oat. Phytopathology 93:860–866
Acknowledgments
The authors acknowledge the DBT, Govt of India for supporting this work under the network project on “Development and applications of Biotechnological tools for millet improvement”. The authors thank Dr. B.V.S. Reddy, ICRISAT, India for providing the initial mapping population.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Murali Mohan, S., Madhusudhana, R., Mathur, K. et al. Identification of quantitative trait loci associated with resistance to foliar diseases in sorghum [Sorghum bicolor (L.) Moench]. Euphytica 176, 199–211 (2010). https://doi.org/10.1007/s10681-010-0224-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10681-010-0224-x