Abstract
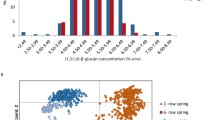
Cell wall degradation is a crucial process within the malting process of barley. Therefore, the haplotype diversity of genes for two cell wall degrading enzymes, (1 → 3),(1 → 4)-β-d-Glucan-4-glucanohydrolase and (1 → 4)-β-Xylan-endohydrolase 1, was investigated and associations to malting quality parameters were performed. The (1 → 3),(1 → 4)-β-d-Glucan-4-glucanohydrolase gene glb2 had two major haplotypes defined by three SNPs and one INDEL, which explained 8.9 and 9.5% of the total variation of malt extract content and viscosity in the spring barley gene pool, respectively. The most significant associations of (1 → 4)-β-Xylan-endohydrolase 1 gene X-1 were found for diastatic power, saccharification VZ45 and soluble nitrogen with 18, 12 and 8% of the total variation explained by SNP3 in the spring barleys. High-throughput markers were developed for both genes which can be used for marker assisted selection.







Similar content being viewed by others
References
Altschul SF, Gish W, Miller W, Myers EW, Lipman DJ (1990) Basic alignment search tool. J Mol Biol 215:403–441
Altschul SF, Madden TL, Schäfer AA, Zhang JZ, Zhang Z, Miller W, Lipman DJ (1997) Gapped BLAST and PSI-BLAST; a new generation of protein database search programs. Nucleic Acids Res 25:3389–3402. doi:10.1093/nar/25.17.3389
Banik M, Garrett TPJ, Fincher GB (1996) Molecular cloning of cDNAs encoding (1 → 4)-β-xylan endohydrolases from the aleurone layer of germinated barley (Hordeum vulgare). Plant Mol Biol 31:1163–1172. doi:10.1007/BF00040833
Banik M, Li CD, Langridge P, Fincher GB (1997) Structure, hormonal regulation, and chromosomal location of genes encoding barley (1 → 4)-β-xylan endohydrolases from the aleurone layer of germinated barley (Hordeum vulgare). Mol Gen Genet 253:599–608. doi:10.1007/s004380050362
Bradbury PJ, Zhang Z, Kroon DE, Casstevens TM, Ramdoss Y, Buckler ES (2007) TASSEL: software for association mapping of complex traits in diverse samples. Bioinformatics 23(19):2633–2635. doi:10.1093/bioinformatics/btm308
Caicedo AL, Williamson SH, Hernandez RD, Boyko A, Fledel-Alon, York TL, Polato NR, Olsen KM, Nielsen R, McCouch S, Bustamente CD, Purugganan MD (2007) Genome-wide patterns of nucleotide polymorphism in domesticated rice. PLoS Genet 3:e163. doi:10.1371/journal.pgen.0030163
Caspers MPM, Lok F, Sinjorgo KMC, van Zeijl MJ, Nielsen KA, Cameron-Mills V (2001) Synthesis, processing and export of cytoplasmatic endo-β-1, 4-xylanase from barley aleurone during germination. Plant J 26:191–204. doi:10.1046/j.0960-7412.2001.01019.x
Choi I-Y, Hyten DL, Matukumalli LK, Song QJ, Chaky JM, Quigley CV, Chase K, Lark KG, Reiter RS, Yoon M-S, Hwang E-Y, Yi S-I, Young ND, Shoemaker RC, van Tassell CP, Specht JE, Cregan PB (2007) A soybean transcript map: gene distribution, haplotype and single-nucleotide polymorphism analysis. Genetics 176:685–696. doi:10.1534/genetics.107.070821
Cockram J, White J, Leigh FJ, Lea VJ, Chiapparino E, Laurie DA, Mackay IJ, Powell W, O’Sullivan DM (2008) Association mapping of partitioning loci in barley. BMC Genetics 9:16. doi:10.1186/147-2156-9-16
Corpet F (1988) Multiple sequence alignment with hierarchical clustering. Nucleic Acids Res 16(22):10881–10890. doi:10.1093/nar/16.22.10881
D’hoop BB, Paulo MJ, Mank RA, van Eck HJ, van Eeuwijk F (2008) Association mapping of quality traits in potato (Solanum tuberosum L.). Euphytica 161:47–60. doi:10.1007/s10681-007-9565-5
Evanno G, Regnaut S, Goudet J (2005) Detecting the number of clusters of individuals using the software STRUCTURE: a simulation study. Mol Ecol 14:2611–2620. doi:10.1111/j.1365-294X.2005.02553.x
Falush D, Stephens M, Pritchard JK (2003) Inference of population structure using multilocus genotype data: linked loci and correlated allele frequencies. Genetics 164:1567–1587
Fincher GB (1989) Molecular and cellular biology associated with endosperm mobilization in germinating cereal grains. Annu Rev Plant Physiol Plant Mol Biol 40:305–346. doi:10.1146/annurev.pp.40.060189.001513
Fincher GB (1992) Barley: genetics, biochemistry, molecular biotechnology and biotechnology. In: Shewry PR (ed) Cell wall metabolism in barley. CAB International, Wallingford, pp 413–437
Fincher GB, Lock PA, Morgan MM, Lingelbach K, Wettenhall REH, Mercer JFB, Brandt A, Thomsen KK (1986) Primary structure of the (1 → 3),(1 → 4)-β-d-glucan 4-glucohydrolase from barley aleurone. Proc Natl Acad Sci USA 83:2081–2085. doi:10.1073/pnas.83.7.2081
Haseneyer G, Ravel C, Dardevet M, Balfourier F, Sourdille P, Charmet G, Brunel D, Sauer S, Geiger HH, Graner A, Stracke S (2008) High level of conservation between genes coding for the GAMYB transcription factor in barley (Hordeum vulgare L.) and bread wheat (Triticum aestivum L.) collections. Theor Appl Genet 117:321–331. doi:10.1007/s00122-008-0777-4
Hyten DL, Choi IY, Song Q, Shoemaker RC, Nelson RL, Costa JM, Specht JE, Cregan PB (2007) Highly variable patterns of linkage disequilibrium in multiple soybean populations. Genetics 175:1937–1944. doi:10.1534/genetics.106.069740
Islam AKMR, Shepherd KW, Sparrow DHB (1981) Isolation and characterization of euplasmic wheat–barley chromosome addition lines. Heredity 46:161–174. doi:10.1038/hdy.1981.24
Johnson GC, Esposito L, Barratt BJ, Smith AN, Heward J, Di Genova G, Ueda H, Cordell HJ, Eaves IA, Dudbridge F, Twells RC, Payne F, Hughes W, Nutland S, Stevens H, Carr P, Tuomilehto-Wolf E, Tuomilehto J, Gough SC, Clayton DG, Todd JA (2001) Haplotype tagging for the identification of common disease genes. Nat Genet 29:233–237. doi:10.1038/ng1001-233
Kantartzi SK, Stewart JMD (2008) Association analysis of fibre traits in Gossypium arboretum accessions. Plant Breed 127:173–179. doi:10.1111/j.1439-0523.2008.01490.x
Kim S, Zhao K, Jiang R, Molitor J, Borevitz JO, Nordborg M, Marjoram P (2006) Association mapping with single-feature polymorphisms. Genetics 173:1125–1133. doi:10.1534/genetics.105.052720
Kraakman ATW, Niks RE, Van der Berg PM, Stam P, Van Eeuwijk FA (2004) Linkage disequilibrium mapping of yield and yield stability in modern spring barley cultivars. Genetics 168:435–446. doi:10.1534/genetics.104.026831
Kraakman ATW, Martinez F, Mussiraliev B, van Eeuwijk FA, Niks RE (2006) Linkage disequilibrium mapping of morphological resistance, and other agronomically relevant traits in modern spring barley cultivars. Mol Breed 17:41–58. doi:10.1007/s11032-005-1119-8
Kuenne C, Lange M, Funke T, Miehe H, Thiel T, Grosse I, Scholz U (2005) CR-EST: a resource for crop ESTs. Nucleic Acids Res 33(Database issue):D619–D621. doi:10.1093/nar/gki119
Li L, Paulo MJ, Strahwald J, Lübeck J, Hofferbert HR, Tacke E, Junghans, Wunder J, Draffehn A, van Eeuwijk F, Gebhardt C (2008) Natural variation at candidate loci is associated with potato chip color, tuber starch content, yield and starch yield. Theor Appl Genet 116:1167–1181. doi:10.1007/s00122-008-0746-y
Litts JC, Simmons CR, Karrer EE, Huang N, Rodriguez RL (1990) The isolation and characterization of a barley 1, 3 → 1, 4 → β-glucanase gene. Eur J Biochem 194:831–838. doi:10.1111/j.1432-1033.1990.tb19476.x
Loi L, Ahluwaila B, Fincher GB (1988) Chromosomal location of genes encoding barley (1 → 3),(1 → 4)-β-glucan 4-glucanohydrolases. Plant Physiol 87:300–302. doi:10.1104/pp.87.2.300
Mackay I, Powell W (2007) Methods for linkage disequilibrium mapping in crops. Trends Plant Sci 12:57–63. doi:10.1016/j.tplants.2006.12.001
MacLeod LC, Lance RCM, Brown AHD (1991) Chromosomal mapping of the Glb 1 locus encoding (1 → 3),(1 → 4)-β-d-Glucan 4-glucanohydrolase EI in barley. J Cereal Sci 13:291–298
Malysheva-Otto L, Röder MS (2006) Haplotype diversity in the endosperm specific β-amylase gene Bmy1 of cultivated barley (Hordeum vulgare L.). Mol Breed 18:143–156. doi:10.1007/s11032-006-9023-4
Mather KA, Caicedo AL, Polato NR, Olsen KM, McCouch S, Purugganan MD (2007) The extent of linkage disequilibrium in rice (Oryza sativa L.). Genetics 177:2223–2232. doi:10.1534/genetics.107.079616
Matthies IE, Weise S, Röder MS (2009) Association of haplotype diversity in the α-amylase gene amy1 with malting quality parameters in barley. Mol Breed 23(1):139–152. doi:10.1007/s11032-008-9221-3
Nordborg M, Hu TT, Ishino Y, Jhaveri J, Toomajian C, Zheng H, Bakker E, Calabrese P, Gladstone J, Goyal R, Jakobsson M, Kim S, Morozov Y, Padhukasahasram B, Plagnol V, Rosenberg NA, Shah C, Wall JD, Wang J, Zhao K, Kalbfleisch T, Schulz V, Kreitman M, Bergelson J (2005) The pattern of polymorphism in Arabidopsis thaliana. PLoS Biol 3:e196. doi:10.1371/journal.pbio.0030196
Plaschke J, Ganal MW, Röder MS (1995) Detection of genetic diversity in closely related bread wheat using microsatellite markers. Theor Appl Genet 91:1001–1007. doi:10.1007/BF00223912
Pritchard JK, Stephens M, Donnelly PJ (2000) Inference of population structure using multilocus genotype data. Genetics 155:945–959
Rafalski A (2002) Applications of single nucleotide polymorphisms in crop genetics. Curr Opin Plant Biol 5:94–100. doi:10.1016/S1369-5266(02)00240-6
Remington DL, Thornsberry JM, Matsuoka Y, Wilson LM, Whitt SR, Doebley J, Kresovich S, Goodman MM, Buckler ES 4th (2001) Structure of linkage disequilibrium and phenotypic associations in the maize genome. Proc Natl Acad Sci USA 98:11479–11484. doi:10.1073/pnas.201394398
Risch NJ (2000) Searching for genetic determinants in the new millennium. Nature 405:847–856. doi:10.1038/35015718
Röder MS, Korzun V, Wendehake K, Plaschke J, Tixier MH, Leroy P, Ganal MW (1998) A microsatellite map of wheat. Genetics 149:2007–2023
Rostoks N, Ramsay L, MacKenzie K, Cardle L, Bhat PR, Roose ML, Svensson JT, Stein N, Varshney RK, Marshall DF, Graner A, Close TJ, Waugh R (2006) Recent history of artificial outcrossing facilitates whole-genome association mapping in elite inbred crop varieties. Proc Natl Acad Sci USA 103:18656–18661. doi:10.1073/pnas.0606133103
Schlueter SD, Dong Q, Brendel V (2003) GeneSeqer@PlantGDB: gene structure prediction in plant genomes. Nucleic Acids Res 31(13):3597–3600. doi:10.1093/nar/gkg533
Searle SR (1987) Linear models for unbalanced data. Wiley, NY
Slade AM, Høj PB, Morrice NA, Fincher GB (1989) Purification and characterization of three (1→ 4)-β-D-xylan endohydrolases from germinated barley. Eur J Biochem 185:533–539
Slakesi N, Fincher GB (1992) Developmental regulation of (1 → 3),(1 → 4)-β-Glucanase gene expression in barley–Tissue-specific expression of individual isozymes. Plant Physiol 99:1126–1231
Slakesi N, Baulcombe DC, Devos KM, Ahluwalia B, Doan DNP, Fincher GB (1990) Structure and tissue-specific regulation of genes encoding barley (1 → 3),(1 → 4)-β-glucan endohydrolases. Mol Gen Genet 224:437–449
Stracke S, Haseneyer G, Veyrieras JB, Geiger HH, Sauer S, Graner A, Piepho HP (2009) Association mapping reveals gene action and interactions in the determination of flowering time in barley. Theor Appl Genet 118(2):259–273. doi10.1007/s00122-008-0896-y
Thornsberry JM, Goodman MM, Doebly J, Kresovich S, Nielsen D, Buckler ES (2001) Dwarf8 polymorphisms associate with variation in flowering time. Nat Genet 28:4673–4680
Weise S, Scholz U, Röder MS, Matthies IE (2009) A comprehensive database of malting quality traits in brewing barley. Barley Genet Newsl 39:1–4. http://wheat.pw.usda.gov/ggpages/bgn/39/
Wolf N (1992) Structure of the genes encoding Hordeum vulgare (1 → 3),(1 → 4)-β-glucanase isoenzymes I and II and functional analysis of their promoters in barley aleurone protoplasts. Mol Gen Genet 234:33–42
Woodward JR, Fincher GB (1982a) Purification and chemical properties of two (1 → 3),(1 → 4)-β-glucan endohydrolases from germinating barley. Eur J Biochem 121:663–669. doi:10.1111/j.1432-1033.1982.tb05837.x
Woodward JR, Fincher GB (1982b) Substrate specifities and kinetic properties of two (1 → 3),(1 → 4)-β-glucan endohydrolases from germinating barley (Hordeum vulgare). Carbohydr Res 106:111–122. doi:10.1016/S0008-6215(00)80737-5
Woodward JR, Morgan JR, Fincher GB (1982) Amino acid sequence homology in two (1 → 3),(1 → 4)-β-glucan endohydrolases from germinating barley (Hordeum vulgare). FEBS Lett 138:198–200. doi:10.1016/0014-5793(82)80440-7
Zhang G, Chen J, Wang J, Ding S (2001) Cultivar and environmental effects on (1 → 3),(1 → 4)-β-d-glucan and protein content in malting barley. J Cereal Sci 34:295–301. doi:10.1006/jcrs.2001.0414
Acknowledgments
We thank A. Flieger, E. Weiß and S. Kirsten for excellent technical assistance. The project was funded by a grant (project no. 0313125A) in the GABI program of the BMBF.
Author information
Authors and Affiliations
Corresponding author
Additional information
The three Supplemental Tables can also be viewed in the following links.
http://pgrc.ipk-gatersleben.de/glb.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Matthies, I.E., Weise, S., Förster, J. et al. Association mapping and marker development of the candidate genes (1 → 3),(1 → 4)-β-d-Glucan-4-glucanohydrolase and (1 → 4)-β-Xylan-endohydrolase 1 for malting quality in barley. Euphytica 170, 109–122 (2009). https://doi.org/10.1007/s10681-009-9915-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10681-009-9915-6