Abstract
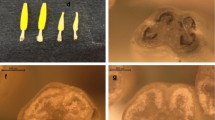
Cytoplasmic male sterility (CMS) is an indispensable trait for F1 hybrid seed production in bunching onion (Allium fistulosum L.). Expansion of the cytoplasmic diversity of F1 hybrid cultivars by introduction of various CMS resources has great potential to eliminate vulnerability to cytoplasm type-specific diseases. This study aimed to evaluate appearance frequency of male sterile plants in several bunching onion accessions and to identify CMS resources. In eight (‘Nogiwa Aigara’, ‘Bansei Hanegi’, ‘Amarume’, ‘Kimnung’, ‘Zhangqiu’, ‘INT/CHN/1990/GOTOU’, ‘Natsunegi’ and ‘Guangzhou’) of 135 accessions collected from Japan, China, Mongolia, Korea and Taiwan, male sterile plants appeared with varied frequencies from 1.7% (‘Nogiwa Aigara’ and ‘Bansei Hanegi’) to 24.5% (‘Zhangqiu’). The inheritance mode of Zhangqiu- and Guangzhou-derived male sterility was confirmed to be CMS by sib-crossings and interbreed crossings. Microscopic examination of microsporogenesis in the CMS plants revealed that microspore protoplasm rapidly degenerated without mitotic division after the release of microspores from tetrads. The CMS germplasm described here would be useful for the development of “A” lines to be used in F1 hybrid seed production of bunching onion. Male fertility in ‘Nogiwa Aigara’, ‘Bansei Hanegi’, ‘Kimnung’, ‘INT/CHN/1990/GOTOU’ and ‘Natsunegi’ was verified to be controlled by a single fertility restoration locus.

Similar content being viewed by others
References
Berninger E (1965) Contribution to the study of male sterility in the onion (Allium cepa L.). Ann Amelior Plant 15:183–199
Buiteveld J, Suo Y, van Lookeren Campagne MM, Creemers-Molenaar J (1998) Production and characterization of somatic hybrid plants between leek (Allium ampeloprasum L.) and onion (Allium cepa L.). Theor Appl Genet 96:765–775
Desloire S, Gherbi H, Laloui W, Mardour S, Clouet V, Cattolico L, Falentin C, Giancola S, Renard M, Budar F, Small I, Caboche M, Delourme R, Bendshmane A (2003) Identification of the fertility restoration locus, Rfo, in radish, as a member of the pentatricopeptide-repeat protein family. Embo Rep 4:588–594
Edwardson JR (1970) Cytoplasmic male sterility. Bot Rev 22:296–738
Engelke T, Terefe D, Tatlioglu T (2003) A PCR-based marker system monitoring CMS-(S), CMS-(T) and (N)-cytoplasm in the onion (Allium cepa L.). Theor Appl Genet 107:162–167
Havey MJ (1995) Identification of cytoplasms using the polymerase chain reaction to aid in the extraction of maintainer lines from open-pollinated populations of onion. Theor Appl Genet 90:263–268
Havey MJ (1999) Seed yield, floral morphology, and lack of male-fertile restoration of male sterile onion (Allium cepa) population possessing the cytoplasm of Allium galanthum. J Am Soc Hortic Sci 124:626–629
Havey MJ (2000) Diversity among male-sterility-inducing and male-fertile cytoplasms of onion. Theor Appl Genet 101:778–782
Holford P, Craft JH, Newbury HJ (1991) Differences between, and possible origins of, the cytoplasms found in fertile and male-sterile onions (Allium cepa L.). Theor Appl Genet 82:737–744
Inden H, Asahira T (1990) Japanese bunching onion (Allium fistulosum L.). In: Rabinowitch HD, Brewster JL (eds) Onions and allied crops, vol 2. CRC Press, Boca Raton, Florida, pp 159–178
Isshiki S, Kawajiri N (2002) Effect of cytoplasm of Solanum violaceum Ort. on fertility of eggplant (S. melongena L.). Sci Hortic 93:9–18
Japan Horticultural Production and Research Institute (2006) In: Itou T (ed) Sosai no Shinhinshu, vol 16. Seibundoushinkousha Publ., Tokyo, Japan, pp 157–164 (in Japanese)
Japan Seed Trade Association (2009) In: Matsunaga K (ed) Yasai Hinshu Meikan. Japan Seed Trade Association, Tokyo (in Japanese)
Jones HA, Clarke AE (1943) Inheritance of male-sterility in the onion and the production of hybrid seed. Proc Am Soc Hortic Sci 43:189–194
Jones HA, Emsweller SL (1937) A male sterile onion. Proc Am Soc Hortic Sci 34:582–585
Katsumata H (1985) Cytoplasmic male sterility and F1 hybrid breeding. In: Yamaguchi H (ed) Cytoplasmic male sterility. CMC press, Tokyo, Japan, pp 183–194
Kik C, Wietsma WA, Verbeek WHJ (1998) Onion. In: Banga SS, Banga SK (eds) Hybrid cultivar development: concepts and methodologies. Narosa Publ. House, New Delhi, India, pp 476–485
Kim S, Lee EL, Cho DY, Han T, Bang H, Patil BS, Ahn YK, Yoon MK (2009) Identification of a novel chimeric gene, orf 725, and its use in development of a molecular marker for distinguishing among three cytoplasm types in onion (Allium cepa L.). Theor Appl Genet 118:433–441
Kumazawa S, Katsumata H (1965) Negi (Japanese bunching onion). In: Kumazawa S (ed) Sosai-engei kakuron (Vegetable crops). Yokendo Publ., Tokyo, pp 280–289 (in Japanese)
Levingth CS III (1991) The Texas cytoplasm of maize: cytoplasmic male sterility and disease susceptibility. Science 250:942–947
Martin WJ, McCallum J, Shigyo M, Jakne J, Kuhl JC, Yamane N, Pither-Joyce M, Gökçe AF, Sink KC, Town CD, Havey MJ (2005) Genetic mapping of expressed sequences in onion and in silico comparisons with rice show scant colinearity. Mol Gen Genom 274:197–204
McCallum J, Pither-Joyce M, Clarke A, Kik C, Van Heusden S, Jakse J, Havey MJ (2005) Towards a framework PCR-based map of onion (Allium cepa L.). Acta Hortic IV IS on Edible Alliaceae 688:173–178
McVetty PBE (1997) Cytoplasmic male sterility. In: Shivanna KR, Sawhney VK (eds) Pollen biotechnology for crop production and improvement. Cambridge University Press, Winnipeg, Manitoba, pp 155–182
Ministry of Agriculture, Forestry and Fisheries (2008) Shuyou-nousanbutsu no Sanshutsugaku (2006) (in Japanese). http://www.e-stat.go.jp/SG1/estat/List.do?lid=000001031687
Moue T, Uehara T (1985) Inheritance of cytoplasmic male sterility in Allium fistulosum L. (welsh onion). J Jpn Soc Hortic Sci 53:432–437 (in Japanese)
Nishimura Y, Shibano M (1972) Male sterility in A. fistulosum L. Cytological and anatomical studies. J Jpn Soc Hortic Sci (Suppl 1):180–181 (in Japanese)
Sato Y (1998) PCR amplification of CMS-specific mitochondrial nucleotide sequences to identify cytoplasmic genotypes of onion (Allium cepa L.). Theor Appl Genet 96:367–370
Schweisguth B (1973) Study of a new type of male sterility in onion, Allium cepa L. Ann Amelior Plant 23:221–233
Shinjo C (1969) Cytoplasmic-genetic male sterility in cultivated rice, Oryza sativa L. II. The inheritance of male sterility. Jpn J Genet 44:149–156
Tsukazaki H, Yamashita K, Yaguchi S, Masuzaki S, Fukuoka H, Yonemaru J, Kanamori H, Kono I, Hang TTM, Shigyo M, Kojima A, Wako T (2008) Construction of SSR-based chromosome map in bunching onion (Allium fistulosum). Theor Appl Genet 117:1213–1223
Uyttewaal M, Arnal N, Quadrado M, Martin-Canadell A, Vrielynck N, Hiard S, Bendahman A, Budar F, Mireau H (2008) Characterization of Raphanus sativus pentatricopeptide repeat proteins encoded by the fertility restorer locus for Ogura cytoplasmic male sterility. Plant Cell 20:3331–3345
van der Meer QP, van Bennecom JL (1969) Effect of temperature on the occurrence of male sterility in onion (Allium cepa L.). Euphytica 18:389–394
Wako T, Tsukazaki H, Ohara T, Yoshida M, Shimazaki S, Ando T, Yamashita K, Kojima A (2009) Verification of variety classification of bunching onion by principal component analysis of morphological traits and growth habit. Bull Natl Inst Veg Tea Sci 8:121–130 (in Japanese)
Wang Z, Zou Y, Zhang X, Chen L, Wu H, Su D, Chen Y, Guo J, Luo D, Long Y, Zhong Y, Liu YG (2006) Cytoplasmic male sterility of rice with boro II cytoplasm is caused by a cytotoxic peptide and is restored by two related PPR motif genes via distinct modes of RNA silencing. Plant Cell 18:676–687
Yabuno T (1977) Genetic studies on the interspecific cytoplasm substitution lines of Japonica varieties of Oryza sativa L. and O. Glaberrima Steud. Euphytica 26:451–463
Yamashita K, Tashiro Y (1999) Possibility of developing male sterile lines of shallot (Allium cepa L. Aggregatum group) with cytoplasm from A. galanthum Kar. et Kir. J Jpn Soc Hortic Sci 68:256–262
Yamashita K, Tashiro Y (2004) Seed productivity test of CMS lines of Japanese bunching onion (Allium fistulosum L.) possessing the cytoplasm of a wild species, A. galanthum Kar. et Kir. Euphytica 136:327–331
Yamashita K, Arita H, Tashiro Y (1999a) Cytoplasm of a wild species, Allium galanthum Kar. et Kir., is useful for developing the male sterile line of A. fistulosum L. J Jpn Soc Hortic Sci 68:788–797
Yamashita K, Arita H, Tashiro Y (1999b) Isozyme and RAPD markers linked to fertility restoring gene for cytoplasmic male sterile Allium fistulosum L. with cytoplasm of A. galanthum Kar. et Kir. J Jpn Soc Hortic Sci 68:954–959
Yamashita K, Takatori Y, Tashiro Y (2002) Development of sequence characterized amplified region (SCAR) markers linked to the fertility restoring gene for cytoplasmic male sterile Allium fistulosum L. possessing the cytoplasm of A. galanthum Kar. et Kir. J Jpn Soc Hortic Sci 71:777–779
Yamashita K, Takatori Y, Tashiro Y (2005) Chromosomal location of a pollen fertility-restoring gene, Rf, for CMS in Japanese bunching onion (Allium fistulosum L.) possessing the cytoplasm of A. galanthum Kar. et Kir. revealed by genomic in situ hybridization. Theor Appl Genet 111:15–22
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yamashita, Ki., Tsukazaki, H., Kojima, A. et al. Inheritance mode of male sterility in bunching onion (Allium fistulosum L.) accessions. Euphytica 173, 357–367 (2010). https://doi.org/10.1007/s10681-009-0101-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10681-009-0101-7