Abstract
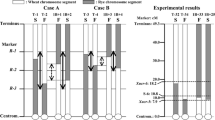
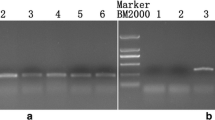
In a previous study, we developed cytoplasmic male sterile lines of Allium fistulosum possessing the cytoplasm of A. galanthum, a wild species, by continuous backcrossing. Furthermore, we reported the presence of a pollen fertility-restoring gene (Rf) for cytoplasmic male sterility (CMS) in A. fistulosum from segregation of pollen fertility of backcross progenies. In the present study, genomic in situ hybridization (GISH), using genomic DNA of A. galanthum as the probe DNA and that of A. fistulosum as the blocking DNA, was applied to F1 hybrids between both species and backcross progenies to determine the chromosomal location of the Rf locus. By means of GISH, eight chromosomes from A. galanthum were clearly discriminated from those of A. fistulosum in the F1 hybrids, and chromosome substitution process through continuous backcrossing was visualized. Interestingly, the chromosome region from A. galanthum, specific to male fertile plants, was detected in one chromosome of BC4 to BC7 generations. Based on the karyotype analysis of the male fertile plants, the chromosome was identified as the 5F chromosome. Our results confirm that the Rf locus is located on the 5F chromosome of the male fertile plants. This is the first report that identified the chromosomal location of the pollen fertility-restoring gene in A. fistulosum.




Similar content being viewed by others
References
Benabdelmouna A, Peltier D, Humbert C, Abirached-Darmency M (1999) Southern and fluorescence in situ hybridization detect three RAPD-generated PCR products useful as introgression markers in Petunia. Theor Appl Genet 98:10–17
Buiteveld J, Suo Y, van Lookeren Campagne MM, Creemers-Molenaar J (1998) Production and characterization of somatic hybrid plants between leek (Allium ampeloprasum L.) and onion (Allium cepa L.). Theor Appl Genet 96:65–775
Emsweller SL, Jones HA (1935) An interspecific hybrid in Allium. Hilgardia 9:265–273
Entwistle AR, Rabinowitch HD, Brewster JL (1990) Root diseases. In: Rabinowitch HD, Brewster JL (eds) Onion and allied crops, vol II. CRC, Boca Raton, pp 103–154
Gokce AF, McCallum J, Sato Y, Havey MJ (2002) Molecular tagging of the Ms locus in onion. J Am Soc Hort Sci 127: 576–582
Havey MJ (1999) Seed yield, floral morphology, and lack of male-fertile restoration of male sterile onion (Allium cepa) population possessing the cytoplasm of Allium galanthum. J Am Soc Hort Sci 124:626–629
Heusden AW van, Shigyo M, Tashiro Y, Vrielink-van Ginkel R, Kik C (2000) AFLP linkage group assignment to the chromosomes of Allium cepa L. via monosomic addition lines. Theor Appl Genet 100:480–486
Hou A, Peffley EB (2000) Recombinant chromosomes of advanced backcross plants between Allium cepa L. and A. fistulosum L. revealed by in situ hybridization. Theor Appl Genet 100:1190–1196
Inden H, Asahira T (1990) Japanese bunching onion (Allium fistulosum L.). In: Rabinowitch HD, Brewster JL (eds) Onions and allied crops, vol II. CRC, Boca Raton, pp 159–178
Kalkman ER (1984) Analysis of the C-banded karyotype of Allium cepa L. standard system of nomenclature and polymorphism. Genetica 65: 141–148
Keller ERJ, Schubert I, Fuchs J, Meister A (1996) Interspecific crosses of onion with distant Allium species and characterization of the presumed hybrids by means of flow cytometry, karyotype analysis and genomic in situ hybridization. Theor Appl Genet 92:417–424
Khrustaleva LI, Kik C (1998) Cytogenetical studies in the bridge cross Allium cepa × (A. fistulosum × A. roylei). Theor Appl Genet 96:8–14
Khrustaleva LI, Kik C (2000) Introgression of Allium fistulosum into A. cepa mediated by A. roylei. Theor Appl Genet 100: 17–26
Khrustaleva LI, Kik C (2001) Localization of single-copy T-DNA insertion in transgenic shallots (Allium cepa) by using ultra-sensitive FISH with tyramide signal amplification. Plant J 25:699–707
King JJ, Bradeen JM, Bark O, McCallum JA, Havey MJ (1998) A low density genetic map of onion reveals a role for tandem duplication in the evolution of an extremely large diploid genome. Theor Appl Genet 96: 52–62
Kumazawa S, Katsumata H (1965) Negi (Japanese bunching onion). In: Kumazawa S (ed) Sosai-engei kakuron (vegetable crops). Yokendo, Tokyo, pp 280–289
Meer QP van der, van Bennekom JL (1978) Improving the onion crop (Allium cepa L.) by transfer of characters from Allium fistulosum L. Bull Warzywhiczy 22:87–91
Moue T, Uehara T (1985) Inheritance of cytoplasmic male sterility in Allium fistulosum L. (Welsh onion). J Jpn Soc Hort Sci 53: 432–437
Mukai Y, Appels R (1996) Direct chromosome mapping of plant genes by in situ polymerase chain reaction (in situ PCR). Chromosome Res 4:401–404
Nishimura Y, Shibano M (1972) Male sterility in Allium fistulosum L. Cytological and anatomical studies. Abstr Jpn Soc Hort Sci Spring Meet:180–181
Ohara T, Wako T, Tsukazaki H, Nunome T, Kojima A (2003) Construction of genetic linkage map based on AFLP markers in bunching onion (Allium fistulosum). Abstr Breed Sci 5(Suppl 1):74
Peterka H, Budahn H, Schrader O, Havey MJ (2002) Transfer of a male-sterility-inducing cytoplasm from onion to leek (Allium ampeloprasum). Theor Appl Genet 105:173–181
Puizina J, Javornik B, Bohanec B, Schweizer D, Maluszynska J, Papes D (1999) Random amplified polymorphic DNA analysis, genome size, and genomic in situ hybridization of triploid viviparous onions. Genome 42:1208–1216
Shigyo M, Tashiro Y, Miyazaki S (1994) Chromosomal locations of glutamate oxaloacetate transaminase gene loci in Japanese bunching onion (Allium fistulosum L.) and shallot (A. cepa L. Aggregatum group). Jpn J Genet 69:417–424
Shigyo M, Tashiro Y, Isshiki S, Miyazaki S (1995a) Chromosomal locations of five isozyme gene loci (Lap-1, Got-1, 6-Pgdh-2, Adh-1 and Gdh-1) in shallot (Allium cepa L. Aggregatum group). Jpn J Genet 70:399–407
Shigyo M, Tashiro Y, Isshiki S, Miyazaki S (1995b) Chromosomal locations of isocitrate dehydrogenase and phosphoglucoisomerase gene loci in shallot (Allium cepa L. Aggregatum group). Jpn J Genet 70:627–632
Shigyo M, Tashiro Y, Isshiki S, Miyazaki S (1996) Establishment of a series of alien monosomic addition lines of Japanese bunching onion (Allium fistulosum L.) with extra chromosomes from shallot (A. cepa L. Aggregatum group). Genes Genet Syst 71:363–371
Shigyo M, Miyazaki T, Isshiki S, Tashiro Y (1997) Assignment of randomly amplified polymorphic DNA markers to all chromosomes of shallot (Allium cepa L. Aggregatum group). Genes Genet Syst 72:249–252
Shigyo M, Imamura K, Iino M, Yamashita K, Tashiro Y (1998) Identification of alien chromosomes in a series of Allium fistulosum-A. cepa monosomic addition lines by means of genomic in situ hybridization. Genes Genet Syst 73:311–315
Shigyo M, Wako T, Kojima A, Yamauchi N, Tashiro Y (2003) Transmission of alien chromosomes from selfed progenies of a complete set of Allium monosomic addition lines: the development of a reliable method for the maintenance of a monosomic addition set. Genome 46:1098–1103
Shigyo M, Yamane N, Martin WJ, Yamauchi N, Havey MJ (2005) Assignment of onion RFLP-linkage map to chromosomes by using alien monosomic additions. Abstr J Jpn Soc Hort Sci 74 (Suppl 1):426
Song YS, Suwabe K, Wako T, Tsukazaki H, Ohara T, Nunome T, Kojima A (2004) Development of microsatellite markers in bunching onion (Allium fistulosum L.). Abstracts of 4th International ISHS symposium on edible Alliaceae (ISEA):197
Tashiro Y, Tsutsumi M, Shigyo M (2000) Production and gene analyses of alien monosomic addition lines of Allium fistulosum L. with extra chromosomes from wild species in section Cepa of Allium. Acta Horticult 521:211–217
Tsukazaki H, Ohara T, Wako T, Song YS, Yamashita K, Kojima A (2004) DNA markers linked to seedling growth in fall-sown bunching onion (Allium fistulosum). Abstracts of 4th international ISHS symposium on edible Alliaceae (ISEA):198
Yamashita K, Tashiro Y (1999) Possibility of developing male sterile lines of shallot (Allium cepa L. Aggregatum group) with cytoplasm from A. galanthum Kar. et Kir. J Jpn Soc Hort Sci 68:256–262
Yamashita K, Tashiro Y (2004) Seed productivity test of CMS lines of Japanese bunching onion (Allium fistulosum L.) possessing the cytoplasm of a wild species, A. galanthum Kar. et Kir. Euphytica 136:327–331
Yamashita K, Arita H, Tashiro Y (1999a) Cytoplasm of a wild species, Allium galanthum Kar. et Kir., is useful for developing the male sterile line of A. fistulosum L. J Jpn Soc Hort Sci 68:788–797
Yamashita K, Arita H, Tashiro Y (1999b) Isozyme and RAPD markers linked to fertility restoring gene for cytoplasmic male sterile Allium fistulosum L. with cytoplasm of A. galanthum Kar. et Kir. J Jpn Soc Hort Sci 68:954–959
Yamashita K, Iino M, Shigyo M, Tashiro Y (2000a) Visualization of nucleus substitution between Allium galanthum and shallot (A. cepa) by genomic in situ hybridization. J Jpn Soc Hort Sci 69:189–191
Yamashita K, Noda R, Tashiro Y (2000b) Use of mitochondrial DNA polymorphisms to distinguish cytoplasms of cultivated and wild species in section Cepa of Allium. J Jpn Soc Hort Sci 69:396–402
Yamashita K, Hisatsune Y, Sakamoto T, Ishizuka K, Tashiro Y (2001) Chromosome and cytoplasm analyses of somatic hybrids between onion (Allium cepa L.) and garlic (A. sativum L.). Euphytica 125:163–167
Yamashita K, Takatori Y, Tashiro Y (2002) Development of sequence characterized amplified region (SCAR) markers linked to the fertility restoring gene for cytoplasmic male sterile Allium fistulosum L. possessing the cytoplasm of A. galanthum Kar. et Kir. J Jpn Soc Hort Sci 71:777–779
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by J.S. Heslop-Harrison
Rights and permissions
About this article
Cite this article
Yamashita, Ki., Takatori, Y. & Tashiro, Y. Chromosomal location of a pollen fertility-restoring gene, Rf, for CMS in Japanese bunching onion (Allium fistulosum L.) possessing the cytoplasm of A. galanthum Kar. et Kir. revealed by genomic in situ hybridization. Theor Appl Genet 111, 15–22 (2005). https://doi.org/10.1007/s00122-005-1941-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00122-005-1941-8