Abstract
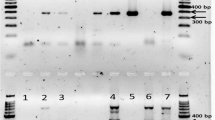
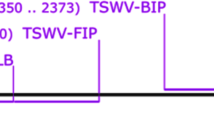
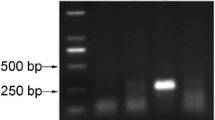
Pospiviroids cause serious economic damage to solanaceous crops, including tomato (Solanum lycopersicum), and cause symptomless infections in many horticultural plants. Symptomless infection and seed transmission are obstacles in visual inspection for plant quarantine. Therefore, in the present study, a comprehensive detection and identification system for molecular diagnosis of eight pospiviroids was developed, based on real-time reverse transcription polymerase chain reaction (RT-PCR). In this system, two real-time RT-PCR methods based on SYBR Green and TaqMan technologies were combined. First, tomato seed samples were screened for all the eight pospiviroids by the SYBR Green method using three primer pairs, including one pair of universal primers. Samples that were positive using the universal primer set were subjected to secondary screening for the identification to species level of six viroids by TaqMan real-time RT-PCR method, with specific primer and probe sets. This two-phase system could specifically detect a viroid in a sample of 400 seeds containing a single contaminated seed. The developed system could be an effective and simple tool to avoid the introduction of pospiviroids through tomato seeds.



Similar content being viewed by others
References
Antignus, Y., Lachman, O., & Pearlsman, M. (2007). Spread of Tomato apical stunt viroid (TASVd) in greenhouse tomato crops is associated with seed transmission and bumble bee activity. Plant Disease, 91(1), 47–50.
Behjatnia, S. A. A., Dry, I. B., Krake, L. R., Condé, B. D., Connelly, M. I., & Randles, J. W. (1996). New Potato spindle tuber viroid and Tomato leaf curl geminivirus strains from a wild Solanum sp. Phytopathology, 86(8), 880–886.
Boonham, N., Gonzáles, P. L., Mendez, M. S., Lilia, P. E., Blockly, A., Walsh, K., Barker, I., & Mumford, R. A. (2004). Development of a real-time RT-PCR assay for the detection of Potato spindle tuber viroid. Journal of Virological Methods, 116(2), 139–146.
Bostan, H., Nie, X., & Singh, R. P. (2004). An RT-PCR primer pair for the detection of pospiviroid and its application in surveying ornamental plants for viroids. Journal of Virological Methods, 116(2), 189–193.
Botermans, M., van de Voseenberg, B. T., Verhoeven, J. T., Roenhorst, J. W., Hooftman, M., & Dekter, R. (2013). Development and validation of a real-time RT-PCR assay for generic detection of pospiviroids. Journal of Virological Methods, 187(1), 43–50.
Brunschot, S. L., Verhoeven, J. T. J., Persley, D. M., Geering, A. D. W., Drenth, A., & Thomas, J. E. (2014). An outbreak of Potato spindle tuber viroid in tomato is linked to imported seed. European Journal of Plant Pathology, 139(1), 1–7.
Chambers, G. A., Seyb, A. M., Mackie, J., Constable, F. E., Rodoni, B. C., & Letham, D. (2013). First report of Pepper chat fruit viroid in traded tomato seed, an interception by Australian biosecurity. Plant Disease, 97(10), 1386.
Flores, R., Gas, M. E. G., Molina-Serrano, D., Nohales, M. A., Carbonell, A., & Gago, S. (2009). Viroid replication: rolling-circles, enzymes and ribozymes. Viruses, 1(2), 317–334.
Hadidi, A., Flores, R., Randles, J. W., & Semancik, J. S. (Eds.). (2003). Viroids. Collingwood: CSIRO.
Hoshino, S., Okuta, T., Isaka, M., Tsutsumi, N., Miyai, N., Ikeshiro, T., Saito, N., Ohara, T., & Takahashi, T. (2006). Detection of Potato spindle tuber viroid (PSTVd) in tomato and potato seeds. Research bulletin of the Plant Protection Service Japan, 42, 75–79.
Kryczynski, S., Paduch-Cichal, E., & Skrzeczkowski, L. J. (1988). Transmission of three viroids through seed and pollen of tomato plants. Journal of Phytopathology, 121(1), 51–57.
Mano, J., Shigemitsu, N., Futo, S., Akiyama, H., Teshima, R., & Hino, A. (2009). Real-time PCR array as a universal platform for the detection of genetically modified crops and its application in identifying unapproved genetically modified crops in Japan. Journal of Agricultural and Food Chemistry, 57(1), 26–37.
Matsushita, Y., & Tsuda, S. (2014). Distribution of Potato spindle tuber viroid in reproductive organs of petunia during its developmental stages. Phytopathology, 104(9), 964–969.
Matsushita, Y., & Tsuda, S. (2016). Seed transmission of potato spindle tuber viroid, tomato chlorotic dwarf viroid, tomato apical stunt viroid, and Columnea latent viroid in horticultural plants. European Journal of Plant Pathology. doi:10.1007/s10658-016-0868-z.
Matsushita, Y., Usugi, T., & Tsuda, S. (2011). Distribution of tomato chlorotic dwarf viroid in floral organs of tomato. European Journal of Plant Pathology, 130(4), 441–447.
Monger, W., Tomlinson, J., Booonham, N., Marn, M. V., Plesko, I. M., & Molinero-Demilly, V. (2010). Development and inter-laboratory evaluation of real-time PCR assays for the detection of pospiviroids. Journal of Virological Methods, 169(1), 207–210.
Singh, R. P., Dilworth, A. D., Ao, X., Singh, M., & Baranwal, V. K. (2009). Citrus exocortis viroid transmission through commercially-distributed seeds of Impatiens and Verbena plants. European Journal of Plant Pathology, 124(4), 691–694.
Thompson, J. D., Higgins, D. G., & Gibson, T. J. (1994). CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Research, 22(22), 4673–4680.
Verhoeven, J. T. J., Jansen, C. C. C., Willemen, T. M., Kox, L. F. F., Owens, R. A., & Roenhorst, J. W. (2004). Natural infections of tomato by Citrus exocortis viroid, Columnea latent viroid, Potato spindle tuber viroid and Tomato chlorotic dwarf viroid. European Journal of Plant Pathology, 110(8), 823–831.
Verhoeven, J. T. J., Jansen, C. C., Roenhorst, J. W., Flores, R., & de la Peña, M. (2009). Pepper chat fruit viroid: biological and molecular properties of a proposed new species of the genus pospiviroid. Virus Research, 144(1), 209–214.
Verhoeven, J. T. J., Roenhorst, J. W., & Owens, R. A. (2011). Mexican papita viroid and tomato planta macho viroid belong to a single species in the genus pospiviroid. Archives of Virology, 156(8), 1433–1437.
Acknowledgements
This study was supported by a Grant-in-Aid for “Regulatory research projects for food safety, animal health and plant protection” and “Development of detection and identification techniques of pests in research and development for global warming adaptation and abnormal weather correspondence” from the Ministry of Agriculture, Forestry, and Fisheries of Japan. We also thank J. Th. J. Verhoeven (Naktuinbouw, The Netherlands) and K. Reanwarakorn (Kasetsart University, Thailand) for supplying the viroid infected materials. We thank Y. Matsumura, Y. Narita, and J. Sato for preparing the experimental materials.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yanagisawa, H., Shiki, Y., Matsushita, Y. et al. Development of a comprehensive detection and identification molecular based system for eight pospiviroids. Eur J Plant Pathol 149, 11–23 (2017). https://doi.org/10.1007/s10658-017-1157-1
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10658-017-1157-1