Abstract
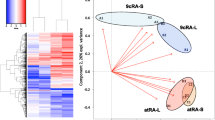
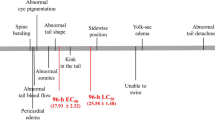
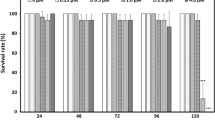
Zebrafish (Danio rerio) embryos were comparably exposed to seven known agonists of retinoid X receptors (RXRs) including two endogenous compounds (9-cis-retinoic acid and docosahexaenoic acid), four man-made selective ligands (LGD1069, SR11237, fluorobexarotene and CD3254), and a biocide (triphenyltin). The dominant phenotypes of malformation were sharp mouths and small caudal fins in 1 mg/L SR11237-treated group after 5 days exposure. 9-cis-retinoic acid and LGD1069 induced multiple malformations including small eyes, bent notochords, reduced brain, enlarged proctodaems, absence of fins, short tails and edema after 5 days exposure. Fluorobexarotene and CD3254 induced similar phenotypes of malformations after 5 days exposure at low concentration (20 μg/L) to those after the 1st d exposure at high concentrations (50 and 100 μg/L). Triphenlytin induced multiple malformations including deformed eyes, bent notochords, bent tails, and edema in hearts after 5 days exposure at concentrations of 1–10 μg Sn/L. In contrast, no discernible malformations were observed in triphenlytin-treated groups after each separate day exposure. These agonists not only showed different ability of teratogenicity but also induced different phenotypes of malformation in zebrafish embryos. In addition, the sensitive stages of zebrafish embryos were different in response to these agonists. Therefore, our results suggest that the agonists of RXRs had divergent teratogenicity in zebrafish embryos.





Similar content being viewed by others
References
Ali S, Van Mil HGJ, Richardson MK (2011) Large-scale assessment of the zebrafish embryo as a possible predictive model in toxicity testing. PLoS ONE 6(6):e21076
Brannen KC, Panzica-Kelly JM, Danberry TL, Augustine-Rauch KA (2010) Development of a zebrafish embryo teratogenicity assay and quantitative prediction model. Birth Defects Res B 89(1):66–77
Burkhardt-Holm P, Oulmi Y, Schroeder A, Braunbeck T (1999) Toxicity of 4-chloroaniline in early life stages of zebrafish (Danio rerio): II. cytopathology and regeneration of liver and gills after prolonged exposure to waterborne 4-chloroaniline. Arch Environ Contam Toxicol 37:85–102
Chawla A (2001) Nuclear receptors and lipid physiology: opening the x-files. Science 294(5548):1866–1870
Dawson MI, Xia ZB (2012) The retinoid X receptors and their ligands. Biochim Biophys Acta 1821(1):21–56
de Urquiza AM (2000) Docosahexaenoic acid, a ligand for the retinoid X receptor in mouse brain. Science 290(5499):2140–2144
Embry MR, Belanger SE, Braunbeck TA, Galay-Burgos M, Halder M, Hinton DE, Léonard MA, Lillicrap A, Norberg-King T, Whale G (2010) The fish embryo toxicity test as an animal alternative method in hazard and risk assessment and scientific research. Aquat Toxicol 97:79–87
Fent K, Meier W (1994) Effects of triphenyltin on fish early life stages. Arch Environ Contam Toxicol 27:224–231
Görge G, Nagel R (1990) Toxicity of lindane, atrazine, and deltamethrin to early life stages of zebrafish (Danio rerio). Ecotoxicol Environ Saf 20(3):246–255
Grün F (2006) Endocrine-disrupting organotin compounds are potent inducers of adipogenesis in vertebrates. Mol Endocrinol 20(9):2141–2155
Guo SZ, Qian LJ, Shi HH, Barry T, Cao QZ, Liu JQ (2010) Effects of tributyltin (TBT) on Xenopus tropicalis embryos at environmentally relevant concentrations. Chemosphere 79(5):529–533
Hu JY, Zhang ZB, Wei QQ, Zhen HJ, Zhao YB, Peng H, Wan Y, Giesy JP, Li LX, Zhang B (2009) Malformations of the endangered Chinese sturgeon, Acipenser sinensis, and its causal agent. Proc Natl Acad Sci USA 106(23):9339–9344
Iguchi T, Katsu Y (2008) Commonality in signaling of endocrine disruption from snail to human. Bioscience 58(11):1061
Kimmel CB, Ballard WW, Kimmel SR, Ullmann B, Schilling TF (1995) Stages of embryonic development of the zebrafish. Dev Dyn 203(3):253–310
Kraft JC, Schuh T, Juchau M, Kimelman D (1994) The retinoid X receptor ligand, 9-cis-retinoic acid, is a potential regulator of early Xenopus development. Proc Natl Acad Sci USA 91:3067–3071
le Maire A, Bourguet W, Balaguer P (2010) A structural view of nuclear hormone receptor: endocrine disruptor interactions. Cell Mol Life Sci 67(8):1219–1237
Lefebvre P, Benomar Y, Staels B (2010) Retinoid X receptors: common heterodimerization partners with distinct functions. Trends Endocrinol Metab 21(11):676–683
Li J, Ma M, Wang Z (2008) A two-hybrid yeast assay to quantify the effects of xenobiotics on retinoid X receptor-mediated gene expression. Toxicol Lett 176(3):198–206
McGrath P, Li CQ (2008) Zebrafish: a predictive model for assessing drug-induced toxicity. Drug Discov Today 13(9–10):394–401
Minucci S, Saint-Jeannet JP, Toyama R, Scita G, Deluca LM, Taira M, Levin AA, Ozato K, Dawid IB (1996) Retinoid X receptor-selective ligands produce malformations in Xenopus embryos. Proc Natl Acad Sci USA 93:1803–1807
Minucci S, Leid M, Toyama R, Saint-Jeannet JP, Peterson VJ, Horn V, Ishmael JE, Bhattacharyya N, Dey A, Dawid IB, Ozato K (1997) Retinoid X receptor (RXR) within the RXR-retinoic acid receptor heterodimer binds its ligand and enhances retinoid-dependent gene expression. Mol Cell Biol 17(2):644–655
Nahoum V, Perez E, Germain P, Rodriguez-Barrios F, Manzo F, Kammerer S, Lemaire G, Hirsch O, Royer CA, Gronemeyer H, de Lera AR, Bourguet W (2007) Modulators of the structural dynamics of the retinoid X receptor to reveal receptor function. Proc Natl Acad Sci USA 104(44):17323–17328
Nishikawa J, Mamiya S, Kanayama T, Nishikawa T, Shiraishi F, Horiguchi T (2004) Involvement of the retinoid X receptor in the development of imposex caused by organotins in gastropods. Environ Sci Technol 38:6271–6276
Panzica-Kelly JM, Zhang CX, Danberry TL, Flood A, DeLan JW, Brannen KC, Augustine-Rauch KA (2010) Morphological score assignment guidelines for the dechorionated zebrafish teratogenicity assay. Birth Defects Res B 89(5):382–395
Papalopulu N, Clarke JD, Bradley L, Wilkinson D, Krumlauf R, Holder N (1991) Retinoic acid causes abnormal development and segmental patterning of the anterior hindbrain in Xenopus embryos. Development 113:1145–1158
Strmac M, Braunbeck T (1999) Effects of triphenlytin acetate on survival, hatching success, and liver ultrastructure of early life stages of zebrafish (Danio rerio). Ecotoxicol Environ Saf 44:25–39
OECD series on testing and assessment (2006) Current approaches in the statistical analysis of ecotoxicity data: a guidance to application. OECD Series on Testing and Assessment No 54. ENV/JM/MONO(2006)18
Yang LX, Ho NY, Alshut R, Legradi J, Weiss C, Reischl M, Mikut R, Liebel U, Müller F, Strähle U (2009) Zebrafish embryos as models for embryotoxic and teratological effects of chemicals. Reprod Toxicol 28(2):245–253
Yu L, Zhang XL, Yuan J, Cao QZ, Liu JQ, Zhu P, Shi HH (2011) Teratogenic effects of triphenyltin on embryos of amphibian (Xenopus tropicalis): a phenotypic comparison with the retinoid X and retinoic acid receptor ligands. J Hazard Mater 192(3):1860–1868
Yuan J, Zhang XL, Yu L, Sun Z, Zhu P, Wang XH, Shi HH (2011) Stage-specific malformations and phenotypic changes induced in embryos of amphibian (Xenopus tropicalis) by triphenyltin. Ecotoxicol Environ Saf 74(7):1960–1966
Acknowledgments
This work was supported by grants from the Natural Science Foundation of China (20877023) and the State Key Laboratory of Estuarine and Coastal Research (2010RCDW01).
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Shi, H., Zhu, P., Sun, Z. et al. Divergent teratogenicity of agonists of retinoid X receptors in embryos of zebrafish (Danio rerio). Ecotoxicology 21, 1465–1475 (2012). https://doi.org/10.1007/s10646-012-0900-9
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10646-012-0900-9