Summary
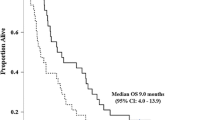
Gemcitabine and cisplatin combination chemotherapy have been shown to have promising efficacy for the treatment of advanced biliary tract cancer (BTC) as a first-line chemotherapy. However, this treatment has not been approved for clinical practice in Japan. Oral fluoropyrimidines (e.g., S-1 and capecitabine) are also promising agents that are widely used with or without gemcitabine. Unfortunately, there is no standard chemotherapy for patients refractory to gemcitabine and oral fluoropyrimidine. We conducted a feasibility study of gemcitabine and cisplatin combination chemotherapy for patients with advanced BTC who are refractory to gemcitabine and S-1. Gemcitabine (1,000 mg/m2) and cisplatin (25 mg/m2) were administered intravenously on days 1 and 8, and this regimen was repeated every 3 weeks. Tumor response was assessed every two cycles using the Response Evaluation Criteria in Solid Tumors version 1.0. Twenty patients with pathologically confirmed BTC were enrolled. Gemcitabine and cisplatin combination chemotherapy was administered as a second-line chemotherapy in thirteen patients and as a third-line chemotherapy in seven patients. Tumor response did not occur in any of the cases. Fourteen patients demonstrated stable diseases, and the disease control rate was 70%. Median overall survival and time-to-progression were 5.9 months (95% CI, 3.9–11.3 months) and 3.6 months (95% CI, 2.2–4.2 months), respectively. Grade 3/4 toxicities included leucopenia (35%), neutropenia (35%), anemia (20%), and thrombocytopenia (15%). Two patients treated for approximately 1 year developed cisplatin-related toxicities. In conclusion, gemcitabine and cisplatin combination chemotherapy produces a limited tumor response in BTC, but may prolong patient’s survival.


Similar content being viewed by others
References
Hezel AF, Zhu AX (2008) Systemic therapy for biliary tract cancers. Oncologist 13(4):415–423
Eckel F, Schmid RM (2007) Chemotherapy in advanced biliary tract carcinoma: a pooled analysis of clinical trials. Br J Cancer 96(6):896–902
Yonemoto N, Furuse J, Okusaka T, Yamao K, Funakoshi A, Ohkawa S, Boku N, Tanaka K, Nagase M, Saisho H, Sato T (2007) A multi-center retrospective analysis of survival benefits of chemotherapy for unresectable biliary tract cancer. Jpn J Clin Oncol 37(11):843–851
Valle J, Wasan H, Palmer DH, Cunningham D, Anthoney A, Maraveyas A, Madhusudan S, Iveson T, Hughes S, Pereira SP, Roughton M, Bridgewater J (2010) Cisplatin plus gemcitabine versus gemcitabine for biliary tract cancer. N Engl J Med 362(14):1273–1281
Furuse J, Okusaka T, Miyazaki M, Tainai H, Nimura Y (2009) A randomized study of gemcitabine/cisplatin versus single-agent gemcitabine in patients with biliary tract cancer. Proc Am Soc Clin Oncol 27:15s, Abstract No: 4579
Okusaka T, Ishii H, Funakoshi A, Yamao K, Ohkawa S, Saito S, Saito H, Tsuyuguchi T (2006) Phase II study of single-agent gemcitabine in patients with advanced biliary tract cancer. Cancer Chemother Pharmacol 57(5):647–653
Ueno H, Okusaka T, Ikeda M, Takezako Y, Morizane C (2004) Phase II study of S-1 in patients with advanced biliary tract cancer. Br J Cancer 91(10):1769–1774
Furuse J, Okusaka T, Boku N, Ohkawa S, Sawaki A, Masumoto T, Funakoshi A (2008) S-1 monotherapy as first-line treatment in patients with advanced biliary tract cancer: a multicenter phase II study. Cancer Chemother Pharmacol 62(5):849–855
Sasaki T, Isayama H, Yashima Y, Yagioka H, Kogure H, Arizumi T, Togawa O, Matsubara S, Ito Y, Nakai Y, Sasahira N, Hirano K, Tsujino T, Tada M, Kawabe T, Omata M (2009) S-1 monotherapy in patients with advanced biliary tract cancer. Oncology 77(1):71–74
Sasaki T, Isayama H, Nakai Y, Ito Y, Kogure H, Togawa O, Toda N, Yasuda I, Hasebe O, Maetani I, Sasahira N, Hirano K, Tsujino T, Tada M, Omata M (2010) Multicenter, phase II study of gemcitabine and S-1 combination chemotherapy in patients with advanced biliary tract cancer. Cancer Chemother Pharmacol 65(6):1101–1107
Murakami Y, Uemura K, Sudo T, Hayashidani Y, Hashimoto Y, Nakamura H, Nakashima A, Sueda T (2009) Adjuvant gemcitabine plus S-1 chemotherapy improves survival after aggressive surgical resection for advanced biliary carcinoma. Ann Surg 250(6):950–956
Lee MA, Woo IS, Kang JH, Hong YS, Lee KS (2004) Gemcitabine and cisplatin combination chemotherapy in intrahepatic cholangiocarcinoma as second-line treatment: report of four cases. Jpn J Clin Oncol 34(9):547–550
Therasse P, Arbuck SG, Eisenhauer EA, Wanders J, Kaplan RS, Rubinstein L, Verweij J, Van Glabbeke M, Van Oosterom AT, Christian MC, Gwyther SG (2000) New guidelines to evaluate the response to treatment in solid tumors. European Organization for Research and Treatment of Cancer, National Cancer Institute of the United States, National Cancer Institute of Canada. J Natl Cancer Inst 92(3):205–216
Paule B, Herelle MO, Rage E, Ducreux M, Adam R, Guettier C, Bralet MP (2007) Cetuximab plus gemcitabine-oxaliplatin (GEMOX) in patients with refractory advanced intrahepatic cholangiocarcinomas. Oncology 72(1–2):105–110
Pino MS, Milella M, Gelibter A, Sperduti I, De Marco S, Nuzzo C, Bria E, Carpanese L, Ruggeri EM, Carlini P, Cognetti F (2009) Capecitabine and celecoxib as second-line treatment of advanced pancreatic and biliary tract cancers. Oncology 76(4):254–261
Lee S, Oh SY, Kim BG, Kwon HC, Kim SH, Rho MH, Kim YH, Rho MS, Jeong JS, Kim HJ (2009) Second-line treatment with a combination of continuous 5-fluorouracil, doxorubicin, and mitomycin-C (conti-FAM) in gemcitabine-pretreated pancreatic and biliary tract cancer. Am J Clin Oncol 32(4):348–352
Oh SY, Jeong CY, Hong SC, Kim TH, Ha CY, Kim HJ, Lee GW, Hwang IG, Jang JS, Kwon HC, Kang JH (2010) Phase II study of second line gemcitabine single chemotherapy for biliary tract cancer patients with 5-fluorouracil refractoriness. Invest New Drugs. doi:10.1007/s10637-010-9417-3
Sasaki T, Isayama H, Nakai Y, Togawa O, Kogure H, Ito Y, Yamamoto K, Mizuno S, Yagioka H, Yashima Y, Kawakubo K, Arizumi T, Matsubara S, Sasahira N, Hirano K, Tsujino T, Toda N, Tada M, Omata M, Koike K (2010) Prognostic factors in patients with advanced biliary tract cancer receiving chemotherapy. Cancer Chemother Pharmacol (in press)
Conflict of interest statement
None declared.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sasaki, T., Isayama, H., Nakai, Y. et al. Feasibility study of gemcitabine and cisplatin combination chemotherapy for patients with refractory biliary tract cancer. Invest New Drugs 29, 1488–1493 (2011). https://doi.org/10.1007/s10637-010-9485-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10637-010-9485-4