Abstract
Vincristine (VCR), an alkaloid isolated from vinca, is a commonly used chemotherapeutic drug. However, VCR therapy can lead to dose-dependent peripheral neurotoxicity, mainly manifesting as neuropathic pain, which is one of the dominant reasons for limiting its utility. Experimentally, we discovered that VCR-induced neuropathic pain (VINP) was accompanied by astrocyte activation; the upregulation of phospho-CaMKII (p-CaMKII), CaV3.2, and Connexin-43 (Cx43) expression; and the production and release of inflammatory cytokines and chemokines in the spinal cord. Similar situations were also observed in astrocyte cultures. Interestingly, these alterations were all reversed by intrathecal injection of KN-93 (a CaMKII inhibitor) or l-Ascorbic acid (a CaV3.2 inhibitor). In addition, KN-93 and l-Ascorbic acid inhibited the increase in [Ca2+]i associated with astrocyte activation. We also verified that knocking down or inhibiting Cx43 level via intrathecal injection of Cx43 siRNA or Gap27 (a Cx43 mimetic peptide) relieved pain hypersensitivity and reduced the release of inflammatory factors; however, they did not affect astrocyte activation or p-CaMKII and CaV3.2 expression. Besides, the overexpression of Cx43 through the transfection of the Cx43 plasmid did not affect p-CaMKII and CaV3.2 expressions in vitro. Therefore, CaMKII and CaV3.2 may activate astrocytes by increasing [Ca2+]i, thereby mediating Cx43-dependent inflammation in VINP. Moreover, we demonstrated that the CaMKII signalling pathway was involved in VCR-induced inflammation, apoptosis, and mitochondrial damage. Collectively, our findings show a novel mechanism by which CaMKII and CaV3.2 mediate Cx43-dependent inflammation by activating astrocytes in neuropathic pain induced by VCR.
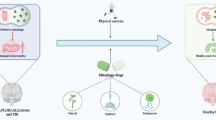
Graphical abstract











Similar content being viewed by others
Data availability
All data generated or analysed during this study are included in this published article.
References
Balayssac D, Ferrier J, Descoeur J, Ling B, Pezet D, Eschalier A, et al. Chemotherapy-induced peripheral neuropathies: from clinical relevance to preclinical evidence. Expert Opin Drug Saf. 2011;10(3):407–17. https://doi.org/10.1517/14740338.2011.543417.
Bennett MV, Garre JM, Orellana JA, et al. Connexin and pannexin hemichannels in inflammatory responses of glia and neurons. Brain Res. 2012;1487:3–15. https://doi.org/10.1016/j.brainres.2012.08.042.
Bourinet E, Alloui A, Monteil A, Barrère C, Couette B, Poirot O, et al. Silencing of the Cav3.2 T-type calcium channel gene in sensory neurons demonstrates its major role in nociception. EMBO J. 2005;24(2):315–24. https://doi.org/10.1038/sj.emboj.7600515.
Cavaletti G, Alberti P, Argyriou AA, Lustberg M, Staff NP, Tamburin S, et al. Chemotherapy-induced peripheral neurotoxicity: a multifaceted, still unsolved issue. J Peripher Nerv Syst. 2019;24(Suppl 2):S6–S12. https://doi.org/10.1111/jns.12337.
Chen JY, Chang CY, Feng PH, Chu CC, So EC, Hu ML. Plasma vitamin C is lower in postherpetic neuralgia patients and administration of vitamin C reduces spontaneous pain but not brush-evoked pain. Clin J Pain. 2009;25(7):562–9. https://doi.org/10.1097/AJP.0b013e318193cf32.
Chen L, Xu B, Liu L, Luo Y, Yin J, Zhou H, et al. Hydrogen peroxide inhibits mTOR signaling by activation of AMPKalpha leading to apoptosis of neuronal cells. Lab Investig. 2010;90(5):762–73. https://doi.org/10.1038/labinvest.2010.36.
Chen JY, Chu CC, Lin YS, So EC, Shieh JP, Hu ML. Nutrient deficiencies as a risk factor in Taiwanese patients with postherpetic neuralgia. Br J Nutr. 2011;106(5):700–7. https://doi.org/10.1017/S0007114511000481.
Chen G, Park CK, Xie RG, Berta T, Nedergaard M, Ji RR. Connexin-43 induces chemokine release from spinal cord astrocytes to maintain late-phase neuropathic pain in mice. Brain. 2014;137(Pt 8):2193–209. https://doi.org/10.1093/brain/awu140.
Chen XJ, Wang L, Song XY. Mitoquinone alleviates vincristine-induced neuropathic pain through inhibiting oxidative stress and apoptosis via the improvement of mitochondrial dysfunction. Biomed Pharmacother. 2020;125:110003. https://doi.org/10.1016/j.biopha.2020.110003.
Cheng G, Kong R, Zhang L, Zhang JN. Mitochondria in traumatic brain injury and mitochondrial-targeted multipotential therapeutic strategies. Br J Pharmacol. 2012;167(4):699–719. https://doi.org/10.1111/j.1476-5381.2012.02025.x.
Circu ML, Aw TY. Reactive oxygen species, cellular redox systems, and apoptosis. Free Radic Biol Med. 2010;48(6):749–62. https://doi.org/10.1016/j.freeradbiomed.2009.12.022.
Dai W, Zhang L, Han L, et al. Regulation of the K-JNK gap junction signaling pathway by immunomodulator astragaloside IV attenuates neuropathic pain. Reg Anesth Pain Med. 2020;45(12):955–63. https://doi.org/10.1136/rapm-2020-101411.
Diniz LP, Tortelli V, Garcia MN, Araújo APB, Melo HM, Seixas da Silva GS, et al. Astrocyte transforming growth factor beta 1 promotes inhibitory synapse formation via CaM kinase II signaling. Glia. 2014;62(12):1917–31. https://doi.org/10.1002/glia.22713.
Dixon WJ. Efficient analysis of experimental observations. Annu Rev Pharmacol Toxicol. 1980;20:441–62. https://doi.org/10.1146/annurev.pa.20.040180.002301.
Dosch M, Zindel J, Jebbawi F, Melin N, Sanchez-Taltavull D, Stroka D, et al. Connexin-43-dependent ATP release mediates macrophage activation during sepsis. Elife. 2019;8(undefined), undefined. https://doi.org/10.7554/eLife.42670.
François A, Schüetter N, Laffray S, Sanguesa J, Pizzoccaro A, Dubel S, et al. The low-threshold calcium channel Cav3.2 determines low-threshold mechanoreceptor function. Cell Rep. 2015;10(3):370–82. https://doi.org/10.1016/j.celrep.2014.12.042.
Franklin JL. Redox regulation of the intrinsic pathway in neuronal apoptosis. Antioxid Redox Signal. 2011;14(8):1437–48. https://doi.org/10.1089/ars.2010.3596.
Gao YJ, Ji RR. Chemokines, neuronal-glial interactions, and central processing of neuropathic pain. Pharmacol Ther. 2010;126(1):56–68. https://doi.org/10.1016/j.pharmthera.2010.01.002.
Gold MS, Gebhart GF. Nociceptor sensitization in pain pathogenesis. Nat Med. 2010;16(11):1248–57. https://doi.org/10.1038/nm.2235.
Guindon J, Deng L, Fan B, Wager-Miller J, Hohmann AG. Optimization of a cisplatin model of chemotherapy-induced peripheral neuropathy in mice: use of vitamin C and sodium bicarbonate pretreatments to reduce nephrotoxicity and improve animal health status. Mol Pain. 2014;10:56. https://doi.org/10.1186/1744-8069-10-56.
Guo W, Wang H, Watanabe M, Shimizu K, Zou S, LaGraize SC, et al. Glial-cytokine-neuronal interactions underlying the mechanisms of persistent pain. J Neurosci. 2007;27(22):6006–18. https://doi.org/10.1523/jneurosci.0176-07.2007.
Gupta KK, Bharne SS, Rathinasamy K, Naik NR, Panda D. Dietary antioxidant curcumin inhibits microtubule assembly through tubulin binding. FEBS J. 2006;273(23):5320–32. https://doi.org/10.1111/j.1742-4658.2006.05525.x.
Hu Y, Kodithuwakku ND, Zhou L, et al. Levo-corydalmine alleviates neuropathic cancer pain induced by tumor compression via the CCL2/CCR2 pathway. Molecules. 2017;22(6), undedfined. https://doi.org/10.3390/molecules22060937.
Kantarjian H, Thomas D, O’Brien S, et al. Long-term follow-up results of hyperfractionated cyclophosphamide, vincristine, doxorubicin, and dexamethasone (Hyper-CVAD), a dose-intensive regimen, in adult acute lymphocytic leukemia. Cancer. 2004;101(12):2788–801. https://doi.org/10.1002/cncr.20668.
Kawasaki Y, Xu Z, Wang X, et al. Distinct roles of matrix metalloproteases in the early- and late-phase development of neuropathic pain. Nat Med. 2008;14(3):331–6. https://doi.org/10.1038/nm1723.
Kim J, Sharma RP. Calcium-mediated activation of c-Jun NH2-terminal kinase (JNK) and apoptosis in response to cadmium in murine macrophages. Toxicol Sci. 2004;81(2):518–27. https://doi.org/10.1093/toxsci/kfh221.
Koopman W, Nijtmans L, Dieteren C, et al. Mammalian mitochondrial complex I: biogenesis, regulation, and reactive oxygen species generation. Antioxid Redox Signal. 2010;12(12):1431–70. https://doi.org/10.1089/ars.2009.2743.
Li R, Shen L, Yu X, Ma C, Huang Y. Vitamin C enhances the analgesic effect of gabapentin on rats with neuropathic pain. Life Sci. 2016;157:25–31. https://doi.org/10.1016/j.lfs.2016.05.035.
Li Y, Tatsui CE, Rhines LD, North RY, Harrison DS, Cassidy RM, et al. Dorsal root ganglion neurons become hyperexcitable and increase expression of voltage-gated T-type calcium channels (Cav3.2) in paclitaxel-induced peripheral neuropathy. Pain. 2017;158(3):417–29. https://doi.org/10.1097/j.pain.0000000000000774.
Li GZ, Hu YH, Li DY, Zhang Y, Guo HL, Li YM, et al. Vincristine-induced peripheral neuropathy: a mini-review. Neurotoxicology. 2020;81:161–71. https://doi.org/10.1016/j.neuro.2020.10.004.
Lu R, Kallenborn-Gerhardt W, Geisslinger G, Schmidtko A. Additive antinociceptive effects of a combination of vitamin C and vitamin E after peripheral nerve injury. PLoS One. 2011;6(12):e29240. https://doi.org/10.1371/journal.pone.0029240.
Marambaud P, Dreses-Werringloer U, Vingtdeux V. Calcium signaling in neurodegeneration. Mol Neurodegener. 2009;4:20. https://doi.org/10.1186/1750-1326-4-20.
Maschio M, Zarabla A, Maialetti A, Marchesi F, Giannarelli D, Gumenyuk S, et al. Prevention of bortezomib-related peripheral neuropathy with docosahexaenoic acid and alpha-lipoic acid in patients with multiple myeloma: preliminary data. Integr Cancer Ther. 2018;17(4):1115–24. https://doi.org/10.1177/1534735418803758.
McCallum JB, Kwok WM, Mynlieff M, et al. Loss of T-type calcium current in sensory neurons of rats with neuropathic pain. Anesthesiology. 2003;98(1):209–16. https://doi.org/10.1097/00000542-200301000-00032.
Miao H, Xu J, Xu D, et al. Nociceptive behavior induced by chemotherapeutic paclitaxel and beneficial role of antioxidative pathways. Physiol Res. 2019;68(3):491–500. https://doi.org/10.33549/physiolres.933939.
Morioka N, Fujii S, Kondo S, Zhang FF, Miyauchi K, Nakamura Y, et al. Downregulation of spinal astrocytic connexin43 leads to upregulation of interleukin-6 and cyclooxygenase-2 and mechanical hypersensitivity in mice. Glia. 2018;66(2):428–44. https://doi.org/10.1002/glia.23255.
Morioka N, Nakamura Y, Zhang FF, Hisaoka-Nakashima K, Nakata Y. Role of connexins in chronic pain and their potential as therapeutic targets for next-generation analgesics. Biol Pharm Bull. 2019;42(6):857–66. https://doi.org/10.1248/bpb.b19-00195.
Naranjo JR, Mellstrom B. Ca2+-dependent transcriptional control of Ca2+ homeostasis. J Biol Chem. 2012;287(38):31674–80. https://doi.org/10.1074/jbc.R112.384982.
Nedergaard M, Verkhratsky A. Artifact versus reality--how astrocytes contribute to synaptic events. Glia. 2012;60(7):1013–23. https://doi.org/10.1002/glia.22288.
Nedergaard M, Rodriguez JJ, Verkhratsky A. Glial calcium and diseases of the nervous system. Cell Calcium. 2010;47(2):140–9. https://doi.org/10.1016/j.ceca.2009.11.010.
Neher E, Sakaba T. Multiple roles of calcium ions in the regulation of neurotransmitter release. Neuron. 2008;59(6):861–72. https://doi.org/10.1016/j.neuron.2008.08.019.
Orellana J, Martinez A, Retamal M. Gap junction channels and hemichannels in the CNS: regulation by signaling molecules. Neuropharmacology. 2013;75:567–82. https://doi.org/10.1016/j.neuropharm.2013.02.020.
Park J, Jang K, Park K. Apamin suppresses LPS-induced neuroinflammatory responses by regulating SK channels and TLR4-mediated signaling pathways. Int J Mol Sci. 2020;21(12), undedfined. https://doi.org/10.3390/ijms21124319.
Retamal M, Froger N, Palacios-Prado N, et al. Cx43 hemichannels and gap junction channels in astrocytes are regulated oppositely by proinflammatory cytokines released from activated microglia. J Neurosci. 2007;27(50):13781–92. https://doi.org/10.1523/jneurosci.2042-07.2007.
Sáez J, Green C. Involvement of connexin hemichannels in the inflammatory response of chronic diseases. Int J Mol Sci. 2018;19(9), undedfined. https://doi.org/10.3390/ijms19092469.
Schouten SM, van de Velde ME, Kaspers GJL, Mokkink LB, van der Sluis IM, van den Bos C, et al. Measuring vincristine-induced peripheral neuropathy in children with cancer: validation of the Dutch pediatric-modified Total Neuropathy Score. Support Care Cancer. 2020;28(6):2867–73. https://doi.org/10.1007/s00520-019-05106-3.
Shen Y, Zhang ZJ, Zhu MD, Jiang BC, Yang T, Gao YJ. Exogenous induction of HO-1 alleviates vincristine-induced neuropathic pain by reducing spinal glial activation in mice. Neurobiol Dis. 2015;79:100–10. https://doi.org/10.1016/j.nbd.2015.04.012.
Shimizu N, Ban N, Watanabe Y, Rikitake A, Watanabe R, Tanaka S, et al. The elevation of cardio-ankle vascular index in a patient with malignant lymphoma treated with a combination therapy of rituximab and cyclophosphamide, doxorubicin, vincristine, and prednisolone. J Clin Med Res. 2017;9(8):729–32. https://doi.org/10.14740/jocmr3071w.
Siau C, Bennett GJ. Dysregulation of cellular calcium homeostasis in chemotherapy-evoked painful peripheral neuropathy. Anesth Analg. 2006;102(5):1485–90. https://doi.org/10.1213/01.ane.0000204318.35194.ed.
Siddiqui M, Abdellatif B, Zhai K, et al. Flavonoids alleviate peripheral neuropathy induced by anticancer drugs. Cancers (Basel). 2021;13(7), undedfined. https://doi.org/10.3390/cancers13071576.
Sisignano M, Baron R, Scholich K, Geisslinger G. Mechanism-based treatment for chemotherapy-induced peripheral neuropathic pain. Nat Rev Neurol. 2014;10(12):694–707. https://doi.org/10.1038/nrneurol.2014.211.
Sompol P, Norris C. Ca, Astrocyte activation and calcineurin/NFAT signaling in age-related neurodegenerative diseases. Front Aging Neurosci. 2018;10:199. https://doi.org/10.3389/fnagi.2018.00199.
Song B, Lai B, Zheng Z, Zhang Y, Luo J, Wang C, et al. Inhibitory phosphorylation of GSK-3 by CaMKII couples depolarization to neuronal survival. J Biol Chem. 2010;285(52):41122–34. https://doi.org/10.1074/jbc.M110.130351.
Sticozzi C, Belmonte G, Frosini M, Pessina F. Nitric oxide/cyclic GMP-dependent calcium signalling mediates IL-6- and TNF-α-induced expression of glial fibrillary acid protein. J Mol Neurosci. 2020;71(4):854–66. https://doi.org/10.1007/s12031-020-01708-3.
Surmeier D, Guzman J, Sanchez-Padilla J. Calcium, cellular aging, and selective neuronal vulnerability in Parkinson’s disease. Cell Calcium. 2010;47(2):175–82. https://doi.org/10.1016/j.ceca.2009.12.003.
Tonkin R, Bowles C, Perera C, et al. Attenuation of mechanical pain hypersensitivity by treatment with Peptide5, a connexin-43 mimetic peptide, involves inhibition of NLRP3 inflammasome in nerve-injured mice. Exp Neurol. 2018;300:1–12. https://doi.org/10.1016/j.expneurol.2017.10.016.
Vicario N, Zappala A, Calabrese G, et al. Connexins in the central nervous system: physiological traits and neuroprotective targets. Front Physiol. 2017;8:1060. https://doi.org/10.3389/fphys.2017.01060.
Vicario N, Pasquinucci L, Spitale FM, Chiechio S, Turnaturi R, Caraci F, et al. Simultaneous activation of Mu and delta opioid receptors reduces allodynia and astrocytic connexin 43 in an animal model of neuropathic pain. Mol Neurobiol. 2019;56(11):7338–54. https://doi.org/10.1007/s12035-019-1607-1.
Vicario N, Turnaturi R, Spitale FM, Torrisi F, Zappalà A, Gulino R, et al. Intercellular communication and ion channels in neuropathic pain chronicization. Inflamm Res. 2020;69(9):841–50. https://doi.org/10.1007/s00011-020-01363-9.
Wang A, Xu C. The role of connexin43 in neuropathic pain induced by spinal cord injury. Acta Biochim Biophys Sin Shanghai. 2019;51(6):555–61. https://doi.org/10.1093/abbs/gmz038.
Welsby P, Wang H, Wolfe J, et al. A mechanism for the direct regulation of T-type calcium channels by Ca2+/calmodulin-dependent kinase II. J Neurosci. 2003;23(31):10116–21. https://doi.org/10.1523/jneurosci.23-31-10116.2003.
Wen X, Li Y, Liu X, Sun C, Lin J, Zhang W, et al. Roles of CaMKIIbeta in the neurotoxicity induced by ropivacaine hydrochloride in dorsal root ganglion. Artif Cells Nanomed Biotechnol. 2019;47(1):2948–56. https://doi.org/10.1080/21691401.2019.1642208.
Wu A, Green CR, Rupenthal ID, Moalem-Taylor G. Role of gap junctions in chronic pain. J Neurosci Res. 2012;90(2):337–45. https://doi.org/10.1002/jnr.22764.
Xiao WH, Bennett GJ. Effects of mitochondrial poisons on the neuropathic pain produced by the chemotherapeutic agents, paclitaxel and oxaliplatin. Pain. 2012;153(3):704–9. https://doi.org/10.1016/j.pain.2011.12.011.
Yang H, Yan H, Li X, Liu J, Cao S, Huang B, et al. Inhibition of connexin 43 and phosphorylated NR2B in spinal astrocytes attenuates bone cancer pain in mice. Front Cell Neurosci. 2018;12:129. https://doi.org/10.3389/fncel.2018.00129.
Zamora N, Cheli V, Santiago González D, et al. Deletion of voltage-gated calcium channels in astrocytes during demyelination reduces brain inflammation and promotes myelin regeneration in mice. J Neurosci. 2020;40(17):3332–47. https://doi.org/10.1523/jneurosci.1644-19.2020.
Zhang ZJ, Cao DL, Zhang X, Ji RR, Gao YJ. Chemokine contribution to neuropathic pain: respective induction of CXCL1 and CXCR2 in spinal cord astrocytes and neurons. Pain. 2013;154(10):2185–97. https://doi.org/10.1016/j.pain.2013.07.002.
Zhou L, Hu Y, Li C, Yan Y, Ao L, Yu B, et al. Levo-corydalmine alleviates vincristine-induced neuropathic pain in mice by inhibiting an NF-kappa B-dependent CXCL1/CXCR2 signaling pathway. Neuropharmacology. 2018;135:34–47. https://doi.org/10.1016/j.neuropharm.2018.03.004.
Zhou L, Ao L, Yan Y, Li C, Li W, Ye A, et al. Levo-corydalmine attenuates vincristine-induced neuropathic pain in mice by upregulating the Nrf2/HO-1/CO pathway to inhibit connexin 43 expression. Neurotherapeutics. 2020a;17(1):340–55. https://doi.org/10.1007/s13311-019-00784-7.
Zhou L, Ao L, Yan Y, Li W, Ye A, Hu Y, et al. The therapeutic potential of chemokines in the treatment of chemotherapy- induced peripheral neuropathy. Curr Drug Targets. 2020b;21(3):288–301. https://doi.org/10.2174/1389450120666190906153652.
Zsurka G, Kunz W. Mitochondrial dysfunction and seizures: the neuronal energy crisis. Lancet Neurol. 2015;14(9):956–66. https://doi.org/10.1016/s1474-4422(15)00148-9.
Acknowledgements
The authors acknowledge technical and equipment support for this work provided by the Key Laboratory of Pediatrics, Children’s Hospital of Nanjing Medical University.
Funding
This work was supported by Grants from the National Natural Science Foundation of China (No. 81803642 and 82073845), the China Postdoctoral Science Foundation (No. 2019M651903), the National Major Scientific and Technological Special Project for “Significant New Drugs Development” during the Thirteenth Five-year Plan Period (No. 2018ZX09301043-001), and the Jiangsu Research Hospital Association for Precision Medication (No. JY202010).
Author information
Authors and Affiliations
Contributions
Yunman Li, Yahui Hu, and Feng Chen developed the idea of the study, participated in its design and coordination, and revised the manuscript. Guizhou Li, Yahui Hu, Qingyan Yang, and Di Fu performed the experiments and analysed data. Guizhou Li, Yini Lu, and Yahui Hu drafted the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics statement
The animal study was reviewed and approved by Institutional Animal Care and Use Committee of China Pharmaceutical University (Nanjing, China; licence number: SYXK (Su) 2016-0011).
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Highlights
1. CaMKII, CaV3.2, and Cx43 play vital roles in neuropathic pain.
2. CaMKII and CaV3.2 activate astrocytes by mediating calcium ions.
3. CaMKII and CaV3.2 facilitate Cx43-mediated inflammatory factor release in spinal dorsal horn astrocytes.
4. CaMKII mediates VCR-induced inflammation and apoptosis.
Supplementary information
Supplementary S1.
(A, B) The PWT and PWL of mice in sham and sham treated with Gap27 (144 nM) groups. (C, D) The PWT and PWL of mice in sham and sham treated with KN-93 (70 nM) groups. (E, F) The PWT and PWL of mice in sham and sham treated with l-Ascorbic acid (300 μM) groups. ANOVA followed by Tukey’s post hoc test. n = 8 mice/group. All data are represented as means ± SEM. (PNG 429 kb)
Rights and permissions
About this article
Cite this article
Li, Gz., Hu, Yh., Lu, Yn. et al. CaMKII and CaV3.2 T-type calcium channel mediate Connexin-43-dependent inflammation by activating astrocytes in vincristine-induced neuropathic pain. Cell Biol Toxicol 39, 679–702 (2023). https://doi.org/10.1007/s10565-021-09631-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10565-021-09631-y