Abstract
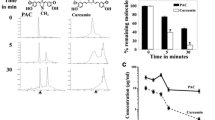
We have investigated here the anti-breast cancer properties of two novel curcumin analogues, EAC and PAC. Apoptosis was assessed by the annexin V/propidium iodide (PI) assay on different breast cancer and normal cells. Immunoblotting analysis determined the effects of these agents on different apoptotic and oncogenic proteins. Furthermore, flow cytometry and Elispot were utilised to investigate the effects on the cell cycle and the production of cytokines, respectively. Breast cancer tumour xenografts were developed in nude mice. Finally, 18F-radiolabeled PAC and curcumin were produced to study their bioavailability and tissue biodistribution in mice. PAC is five times more efficient than curcumin and EAC in inducing apoptosis, mainly via the internal mitochondrial route. This effect was 10-fold higher against ER-negative as compared to ER-positive cells, and ectopic expression of ERα rendered ER-negative breast cancer cells more resistant to PAC. In addition, PAC delayed the cell cycle at G2/M phase with a stronger effect on ER-negative cells. Moreover, PAC exhibited strong capacity as an immuno-inducer through reducing the secretion of the two major Th2 cytokines IL-4 and IL-10. Importantly, PAC significantly reduced tumour size, and triggered apoptosis in vivo. Furthermore, PAC inhibited survivin, NF-kB and its downstream effectors cyclin D1 and Bcl-2, and strongly up-regulated p21WAF1 both in vitro and in tumours. Besides, PAC exhibited higher stability in blood and greater biodistribution and bioavailability than curcumin in mice. These results indicate that PAC could constitute a powerful, yet not toxic, new chemotherapeutic agent against ER-negative breast tumours.







Similar content being viewed by others
References
Smigal C, Jemal A, Ward E, Cokkinides V, Smith R, Howe HL, Thun M (2006) Trends in breast cancer by race and ethnicity: update 2006. CA Cancer J Clin 56:168–183
Garg AK, Buchholz TA, Aggarwal BB (2005) Chemosensitization and radiosensitization of tumors by plant polyphenols. Antioxid Redox Signal 7:1630–1647
Goel A, Kunnumakkara AB, Aggarwal BB (2007) Curcumin as “Curecumin”: from kitchen to clinic. Biochem Pharmacol 19:19
Anand P, Thomas SG, Kunnumakkara AB, Sundaram C, Harikumar KB, Sung B, Tharakan ST, Misra K, Priyadarsini IK, Rajasekharan KN et al (2008) Biological activities of curcumin and its analogues (Congeners) made by man and mother nature. Biochem Pharmacol 76:1590–1611
Bhattacharyya S, Mandal D, Saha B, Sen GS, Das T, Sa G (2007) Curcumin prevents tumor-induced T cell apoptosis through Stat-5a-mediated Bcl-2 induction. J Biol Chem 282:15954–15964
Labbozzetta M, Notarbartolo M, Poma P, Maurici A, Inguglia L, Marchetti P, Rizzi M, Baruchello R, Simoni D, D’Alessandro N (2009) Curcumin as a possible lead compound against hormone-independent, multidrug-resistant breast cancer. Ann NY Acad Sci 1155:278–283
Kim HI, Huang H, Cheepala S, Huang S, Chung J (2008) Curcumin inhibition of integrin (alpha6beta4)-dependent breast cancer cell motility and invasion. Cancer Prev Res (Phila PA) 1:385–391
Moiseeva EP, Almeida GM, Jones GD, Manson MM (2007) Extended treatment with physiologic concentrations of dietary phytochemicals results in altered gene expression, reduced growth, and apoptosis of cancer cells. Mol Cancer Ther 6:3071–3079
Anand P, Sundaram C, Jhurani S, Kunnumakkara AB, Aggarwal BB (2008) Curcumin and cancer: an “old-age” disease with an “age-old” solution. Cancer Lett 267:133–164
Aggarwal BB, Shishodia S, Takada Y, Banerjee S, Newman RA, Bueso-Ramos CE, Price JE (2005) Curcumin suppresses the paclitaxel-induced nuclear factor-kappaB pathway in breast cancer cells and inhibits lung metastasis of human breast cancer in nude mice. Clin Cancer Res 11:7490–7498
Anand P, Kunnumakkara AB, Newman RA, Aggarwal BB (2007) Bioavailability of curcumin: problems and promises. Mol Pharm 4:807–818
Amolins MW, Peterson LB, Blagg BS (2009) Synthesis and evaluation of electron-rich curcumin analogues. Bioorg Med Chem 17:360–367
Simoni D, Rizzi M, Rondanin R, Baruchello R, Marchetti P, Invidiata FP, Labbozzetta M, Poma P, Carina V, Notarbartolo M et al (2008) Antitumor effects of curcumin and structurally beta-diketone modified analogs on multidrug resistant cancer cells. Bioorg Med Chem Lett 18:845–849
Adams BK, Cai J, Armstrong J, Herold M, Lu YJ, Sun A, Snyder JP, Liotta DC, Jones DP, Shoji M (2005) EF24, a novel synthetic curcumin analog, induces apoptosis in cancer cells via a redox-dependent mechanism. Anticancer Drugs 16:263–275
Padhye S, Banerjee S, Chavan D, Pandye S, Swamy KV, Ali S, Li J, Dou QP, Sarkar FH (2009) Fluorocurcumins as cyclooxygenase-2 inhibitor: molecular docking, pharmacokinetics and tissue distribution in mice. Pharm Res 26:2438–2445
Padhye S, Yang H, Jamadar A, Cui QC, Chavan D, Dominiak K, McKinney J, Banerjee S, Dou QP, Sarkar FH (2009) New difluoro Knoevenagel condensates of curcumin, their Schiff bases and copper complexes as proteasome inhibitors and apoptosis inducers in cancer cells. Pharm Res 26:1874–1880
Selvendiran K, Tong L, Vishwanath S, Bratasz A, Trigg NJ, Kutala VK, Hideg K, Kuppusamy P (2007) EF24 induces G2/M arrest and apoptosis in cisplatin-resistant human ovarian cancer cells by increasing PTEN expression. J Biol Chem 282:28609–28618
Youssef KM, El-Sherbeny MA, El-Shafie FS, Farag HA, Al-Deeb OA, Awadalla SA (2004) Synthesis of curcumin analogues as potential antioxidant, cancer chemopreventive agents. Arch Pharm (Weinheim) 337:42–54
Abukhdeir AM, Blair BG, Brenner K, Karakas B, Konishi H, Lim J, Sahasranaman V, Huang Y, Keen J, Davidson N et al (2006) Physiologic estrogen receptor alpha signaling in non-tumorigenic human mammary epithelial cells. Breast Cancer Res Treat 99:23–33
Al-Mohanna MA, Manogaran PS, Al-Mukhalafi Z, Al-Hussein AK, Aboussekhra A (2004) The tumor suppressor p16(INK4a) gene is a regulator of apoptosis induced by ultraviolet light and cisplatin. Oncogene 23:201–212
Aggarwal BB, Kumar A, Bharti AC (2003) Anticancer potential of curcumin: preclinical and clinical studies. Anticancer Res 23:363–398
Rowlands TM, Pechenkina IV, Hatsell S, Cowin P (2004) Beta-catenin and cyclin D1: connecting development to breast cancer. Cell Cycle 3:145–148
Guttridge DC, Albanese C, Reuther JY, Pestell RG, Baldwin AS Jr (1999) NF-kappaB controls cell growth and differentiation through transcriptional regulation of cyclin D1. Mol Cell Biol 19:5785–5799
Hinz M, Krappmann D, Eichten A, Heder A, Scheidereit C, Strauss M (1999) NF-kappaB function in growth control: regulation of cyclin D1 expression and G0/G1-to-S-phase transition. Mol Cell Biol 19:2690–2698
Altieri DC (2008) Survivin, cancer networks and pathway-directed drug discovery. Nat Rev Cancer 8:61–70
Lacroix M, Toillon RA, Leclercq G (2006) p53 and breast cancer, an update. Endocr Relat Cancer 13:293–325
Dotto GP (2000) p21(WAF1/Cip1): more than a break to the cell cycle? Biochim Biophys Acta 1471:M43–M56
Gartel AL, Tyner AL (2002) The role of the cyclin-dependent kinase inhibitor p21 in apoptosis. Mol Cancer Ther 1:639–649
Hickman ES, Moroni MC, Helin K (2002) The role of p53 and pRB in apoptosis and cancer. Curr Opin Genet Dev 12:60–66
Stanelle J, Putzer BM (2006) E2F1-induced apoptosis: turning killers into therapeutics. Trends Mol Med 12:177–185
Cao Y, Karin M (2003) NF-kappaB in mammary gland development and breast cancer. J Mammary Gland Biol Neoplasia 8:215–223
Haffner MC, Berlato C, Doppler W (2006) Exploiting our knowledge of NF-kappaB signaling for the treatment of mammary cancer. J Mammary Gland Biol Neoplasia 11:63–73
Van Waes C (2007) Nuclear factor-kappaB in development, prevention, and therapy of cancer. Clin Cancer Res 13:1076–1082
Paul S, Dey A (2008) Wnt signaling and cancer development: therapeutic implication. Neoplasma 55:165–176
Prasad CP, Gupta SD, Rath G, Ralhan R (2007) Wnt signaling pathway in invasive ductal carcinoma of the breast: relationship between beta-catenin, dishevelled and cyclin D1 expression. Oncology 73:112–117
Bartkova J, Lukas J, Strauss M, Bartek J (1995) Cyclin D1 oncoprotein aberrantly accumulates in malignancies of diverse histogenesis. Oncogene 10:775–778
Yang C, Trent S, Ionescu-Tiba V, Lan L, Shioda T, Sgroi D, Schmidt EV (2006) Identification of cyclin D1- and estrogen-regulated genes contributing to breast carcinogenesis and progression. Cancer Res 66:11649–11658
Tanaka K, Iwamoto S, Gon G, Nohara T, Iwamoto M, Tanigawa N (2000) Expression of survivin and its relationship to loss of apoptosis in breast carcinomas. Clin Cancer Res 6:127–134
Callagy GM, Pharoah PD, Pinder SE, Hsu FD, Nielsen TO, Ragaz J, Ellis IO, Huntsman D, Caldas C (2006) Bcl-2 is a prognostic marker in breast cancer independently of the Nottingham Prognostic Index. Clin Cancer Res 12:2468–2475
Nagai S, Toi M (2000) Interleukin-4 and breast cancer. Breast Cancer 7:181–186
Mocellin S, Marincola FM, Young HA (2005) Interleukin-10 and the immune response against cancer: a counterpoint. J Leukoc Biol 78:1043–1051
Ryu EK, Choe YS, Lee KH, Choi Y, Kim BT (2006) Curcumin and dehydrozingerone derivatives: synthesis, radiolabeling, and evaluation for beta-amyloid plaque imaging. J Med Chem 49:6111–6119
Adams BK, Ferstl EM, Davis MC, Herold M, Kurtkaya S, Camalier RF, Hollingshead MG, Kaur G, Sausville EA, Rickles FR et al (2004) Synthesis and biological evaluation of novel curcumin analogs as anti-cancer and anti-angiogenesis agents. Bioorg Med Chem 12:3871–3883
Acknowledgments
We are grateful to Ben Ho Park for providing us with the ER-expressing pIRESneo3 plasmid. We would like also to thank Dr. Ra’afat El-Sayed, Falah Al-Mohanna and Jonathan Caijas for their help with animal studies. We are also thankful to Dr. Bedri Karakas and Dr. Abde Abukhdeir for their help and fruitful discussions. This work was performed under the RAC proposal # 2050039 and was supported by King Abdelaziz city of Sciences and technology (KACST proposal # 08-MED476-20).
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Al-Hujaily, E.M., Mohamed, A.G., Al-Sharif, I. et al. PAC, a novel curcumin analogue, has anti-breast cancer properties with higher efficiency on ER-negative cells. Breast Cancer Res Treat 128, 97–107 (2011). https://doi.org/10.1007/s10549-010-1089-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-010-1089-3