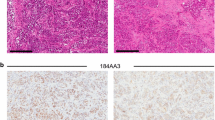
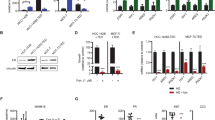
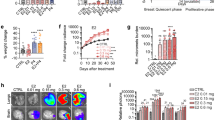
Summary
Currently, a number of breast cancer cell lines exist that serve as models for both estrogen receptor alpha (ERα) positive and ERα negative disease. Models are also available for pre-neoplastic breast epithelial cells that do not express ERα; however, there are no ideal systems for studying pre-neoplastic cells that are ERα positive. This has been largely due to the inability to establish an estrogen growth stimulated, non-tumorigenic breast epithelial cell line, as most human breast epithelial cells engineered to overexpress ERα have been found to be growth inhibited by estrogens. We have developed independently derived clones from the non-cancerous MCF-10A human breast cell line that express ERα and are growth stimulated by 17-beta-estradiol (E2) in the absence of epidermal growth factor (EGF), a cytokine normally required for MCF-10A cell proliferation. This effect is blocked by the selective estrogen receptor modulator (SERM), Tamoxifen and the selective estrogen receptor downregulator, ICI 182,780 (Faslodex, Fulvestrant). Exposure of these cells to EGF and E2 results in a growth inhibitory phenotype similar to previous reports. These data present a reconciling explanation for the previously described paradoxical effects of ERα overexpression, and provide a model for examining the carcinogenic effects of estrogens in non-tumorigenic human breast epithelial cells.
Similar content being viewed by others
References
Clarke RB, Howell A, Potten CS, Anderson E (1997) Dissociation between steroid receptor expression and cell proliferation in the human breast. Cancer Res 57(22): 4987–4991
Petersen OW, Hoyer PE, van Deurs B (1987) Frequency and distribution of estrogen receptor-positive cells in normal, nonlactating human breast tissue. Cancer Res 47(21): 5748–5751
Soule HD, Maloney TM, Wolman SR, Peterson WD Jr., Brenz R, McGrath CM, Russo J, Pauley RJ, Jones RF, Brooks SC (1990) Isolation and characterization of a spontaneously immortalized human breast epithelial cell line, MCF-10. Cancer Res 50(18): 6075–6086
Tang B, Vu M, Booker T, Santner SJ, Miller FR, Anver MR, Wakefield LM (2003) TGF-beta switches from tumor suppressor to prometastatic factor in a model of breast cancer progression. J Clin Invest 112(7): 1116–1124
Shekhar MP, Nangia-Makker P, Wolman SR, Tait L, Heppner GH, Visscher DW (1998) Direct action of estrogen on sequence of progression of human preneoplastic breast disease. Am J Pathol 152(5): 1129–1132
Shekhar PV, Werdell J, Basrur VS (1997) Environmental estrogen stimulation of growth and estrogen receptor function in preneoplastic and cancerous human breast cell lines. J Natl Cancer Inst 89(23): 1774–1782
Yoon DS, Wersto RP, Zhou W, Chrest FJ, Garrett ES, Kwon TK, Gabrielson E (2002) Variable levels of chromosomal instability and mitotic spindle checkpoint defects in breast cancer. Am J Pathol 161(2): 391–397
Iavarone A, Massague J (1997) Repression of the CDK activator Cdc25A and cell-cycle arrest by cytokine TGF-beta in cells lacking the CDK inhibitor p15. Nature 387(6631): 417–422
Thomas TJ, Faaland CA, Adhikarakunnathu S, Watkins LF, Thomas T (1998) Induction of p21 (CIP1/WAF1/SID1) by estradiol in a breast epithelial cell line transfected with the recombinant estrogen receptor gene: a possible mechanism for a negative regulatory role of estradiol. Breast Cancer Res Treat 47(2): 181–193
Pilat MJ, Christman JK, Brooks SC (1996) Characterization of the estrogen receptor transfected MCF10A breast cell line 139B6. Breast Cancer Res Treat 37(3): 253–266
Jiang SY, Jordan VC (1992) Growth regulation of estrogen receptor-negative breast cancer cells transfected with complementary DNAs for estrogen receptor. J Natl Cancer Inst 84(8): 580–591
Mueller H, Loop P, Liu R, Wosikowski K, Kueng W, Eppenberger U (1994) Differential signal transduction of epidermal-growth-factor receptors in hormone-dependent and hormone-independent human breast cancer cells. Eur J Biochem 221(2): 631–637
Mohamood AS, Gyles P, Balan KV, Hollis VW, Eckberg WR, Asseffa A, Han Z, Wyche JH, Anderson WA (1997) Estrogen receptor, growth factor receptor and protooncogene protein activities and possible signal transduction crosstalk in estrogen dependent and independent breast cancer cell lines. J Submicrosc Cytol Pathol 29(1): 1–17
Davidson NE, Gelmann EP, Lippman ME, Dickson RB (1987) Epidermal growth factor receptor gene expression in estrogen receptor-positive and negative human breast cancer cell lines. Mol Endocrinol 1(3): 216–223
Lee CS, Hall RE, Alexander IE, Koga M, Shine J, Sutherland RL (1990) Inverse relationship between estrogen receptor and epidermal growth factor receptor mRNA levels in human breast cancer cell lines. Growth Factors 3(2): 97–103
Briand P, Lundholt BK, Skouv J, Lykkesfeldt AE (1999) Growth response of breast epithelial cells to estrogen is influenced by EGF. Mol Cell Endocrinol 153(1–2): 1–9
Moyret C, Madsen MW, Cooke J, Briand P, Theillet C (1994) Gradual selection of a cellular clone presenting a mutation at codon 179 of the p53 gene during establishment of the immortalized human breast epithelial cell line HMT-3522. Exp Cell Res 215(2): 380–385
Chou JL, Fan Z, DeBlasio T, Koff A, Rosen N, Mendelsohn J (1999) Constitutive overexpression of cyclin D1 in human breast epithelial cells does not prevent G1 arrest induced by deprivation of epidermal growth factor. Breast Cancer Res Treat 55(3): 267–283
Keen JC, Zhou Q, Park BH, Pettit C, Mack KM, Blair B, Brenner K, Davidson NE (2005) Protein phosphatase 2A regulates estrogen receptor {alpha} (ER) expression through modulation of ER mRNA stability. J Biol Chem 280(33): 29519–29524
Bachman KE, Blair BG, Brenner K, Bardelli A, Arena S, Zhou S, Hicks J, De Marzo AM, Argani P, Park BH (2004) p21(WAF1/CIP1) Mediates the growth response to TGF-beta in human epithelial cells. Cancer Biol Ther 3(2): 221–225
Seth P, Porter D, Lahti-Domenici J, Geng Y, Richardson A, Polyak K (2002) Cellular and molecular targets of estrogen in normal human breast tissue. Cancer Res 62(16): 4540–4544
Ignatoski KM, Lapointe AJ, Radany EH, Ethier SP (1999) erbB-2 Overexpression in human mammary epithelial cells confers growth factor independence. Endocrinology 140(8): 3615–3622
Kobayashi M, Yamauchi Y, Tanaka A, Shimamura S (1996) Improved dicistronic mRNA expression vectors for efficient selection of transfectants highly expressing foreign genes. Biotechniques 21(3): 398–402
Anderson NG, Ahmad T, Chan K, Dobson R, Bundred NJ (2001) ZD1839 (Iressa), A novel epidermal growth factor receptor (EGFR) tyrosine kinase inhibitor, potently inhibits the growth of EGFR-positive cancer cell lines with or without erbB2 overexpression. Int J Cancer 94(6): 774–782
Fitzpatrick SL, LaChance MP, Schultz GS (1984) Characterization of epidermal growth factor receptor and action on human breast cancer cells in culture. Cancer Res 44(8): 3442–3447
Arteaga CL, Ramsey TT, Shawver LK, Guyer CA (1997) Unliganded epidermal growth factor receptor dimerization induced by direct interaction of quinazolines with the ATP binding site. J Biol Chem 272(37): 23247–23254
Baulida J, Kraus MH, Alimandi M, Di Fiore PP, Carpenter G (1996) All ErbB receptors other than the epidermal growth factor receptor are endocytosis impaired. J Biol Chem 271(9): 5251–5257
Friday BB, Adjei AA (2005) K-ras as a target for cancer therapy. Biochim Biophys Acta 1756(2): 127–144
Migliaccio A, Di Domenico M, Castoria G, de Falco A, Bontempo P, Nola E, Auricchio F (1996) Tyrosine kinase/p21ras/MAP-kinase pathway activation by estradiol–receptor complex in MCF-7 cells. Embo J 15(6): 1292–1300
Gilhooly EM, Rose DP (1999) The association between a mutated ras gene and cyclooxygenase-2 expression in human breast cancer cell lines. Int J Oncol 15(2): 267–270
Wilhelm SM, Carter C, Tang L, Wilkie D, McNabola A, Rong H, Chen C, Zhang X, Vincent P, McHugh M, Cao Y, Shujath J, Gawlak S, Eveleigh D, Rowley B, Liu L, Adnane L, Lynch M, Auclair D, Taylor I, Gedrich R, Voznesensky A, Riedl B, Post LE, Bollag G, Trail PA (2004) BAY 43–9006 exhibits broad spectrum oral antitumor activity and targets the RAF/MEK/ERK pathway and receptor tyrosine kinases involved in tumor progression and angiogenesis. Cancer Res 64(19): 7099–7109
Gruber CJ, Gruber DM, Gruber IM, Wieser F, Huber JC (2004) Anatomy of the estrogen response element. Trends Endocrinol Metab 15(2): 73–78
DiRenzo J, Signoretti S, Nakamura N, Rivera-Gonzalez R, Sellers W, Loda M, Brown M (2002) Growth factor requirements and basal phenotype of an immortalized mammary epithelial cell line. Cancer Res 62(1): 89–98
Acknowledgements
We thank Lizzy Weiner for technical assistance and members of the Park Lab for critical review of the manuscript. We also thank Cindy Zahnow and members of the Sukumar lab for helpful discussions regarding pharmacologic EGFR inhibition. We thank Dr Kornelia Polyak for the ERE GFP promoter/reporter vector.
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was supported by The Flight Attendant’s Medical Research Institute (FAMRI), The American Cancer Society (#IRG-58-005-41), NIH Breast SPORE Grant P50 CA88843, the Maryland Cigarette Restitution Fund, The Entertainment Industry Foundation, The Department of Defense Breast Cancer Research Program (DAMD17-03-1-0241), and the Avon Foundation. B.H.P. is an Avon Scholar for Breast Cancer Research and also receives generous support from The V Foundation for Cancer Research.
Rights and permissions
About this article
Cite this article
Abukhdeir, A.M., Blair, B.G., Brenner, K. et al. Physiologic estrogen receptor alpha signaling in non-tumorigenic human mammary epithelial cells. Breast Cancer Res Treat 99, 23–33 (2006). https://doi.org/10.1007/s10549-006-9177-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-006-9177-0