Abstract
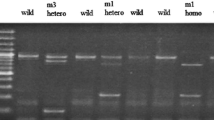
Cytochrome P-450 1A1 (CYP1A1) is involved in the 2-hydroxylation of estrogens and mammary carcinogens into 2-hydroxy catechol metabolites. Many commonly occurring single nucleotide polymorphism (SNP) are reported in CYP1A1 in various populations that include, isoleucine to valine substitution at 462 codon in heme binding region in exon 7 (A to G transition at position 2455; M2), threonine to asparagine substitution at codon 461 (C to A transversion at position 2453; M4), T to C transition at 3801 position (M1) and T to C transition at position 3205 (M3) in 3′ non-coding region. Epidemiological studies have shown inconsistent patterns between CYP1A1 polymorphism and breast cancer risk among various populations. Most of the studies have shown significant association between CYP1A1 genotype polymorphism and breast cancer risk. The present investigation was therefore undertaken to investigate the association of M1, M2, M3 and M4 polymorphisms and their subsequent contribution in premenopausal and postmenopausal women with breast cancer risk in north Indian women. Genomic DNA was isolated from case controls and breast cancer patients, specific segments of genomic DNA were amplified and restriction fragment length polymorphism (RFLP) was performed. CYP1A1 expression and catalytic activity were also assessed in premenopausal and postmenopausal case controls and patients. Polymorphism at M1, M2 and M4 alleles was detected and odds ratio for W/M1 and␣M1/M1 was calculated as 1.07 (95% CI, 0.59–1.87) and 0.74 (95% CI, 0.28–1.96) respectively. Odds ratio for W/M1 and M1/M1 alleles in premenopausal and postmenopausal women was 1.09 (95% CI, 0.45–2.49)/0.62 (95% CI, 0.10–2.66) and 1.60 (95% CI, 0.60–4.22)/1.06 (95% CI, 0.22–7.33) respectively. Odds ratio for W/M4 and M4/M4 allele was 1.20 (95% CI, 0.65–2.24)/4.55 (95% CI, 0.44–226.2) and 0.96 (95% CI, 0.36–2.64)/4.51 (95% CI, 0.23–273.0) respectively in total and premenopausal women. In postmenopausal women odds ratio was calculated as 1.16 (95% CI, 0.45–2.94) for M4/W but it could not be detected for M4/M4 since this genotype was not found in any postmenopausal case controls. Odds ratio for W/M2 genotype was calculated 0.57 (95% CI, 0.28–1.02), 1.06 (95% CI, 0.40–2.47) and 0.33 (95% CI, 0.12–0.89) respectively for total, premenopausal and postmenopausal women, however, in any group the odds ratio for M2/M2 could not be detected as M2/M2 genotype was not found in breast cancer patients. Polymorphism at M1 and M4 alleles was not found significantly associated with breast cancer risk and only wild type genotype was found in case controls and patients for M3 allele. Lack of protective association between CYP1A1 M2 genotype was also observed, however, in postmenopausal women a significant protective association with breast cancer risk was found (odds ratio, 0.33; 95% CI, 0.12–0.89; P-value 0.03). Similarly, no significant alteration in CYP1A1 expression and catalytic activity was observed in wild type and variant genotypes both in premenopausal and postmenopausal patients as compared with their respective controls. The results obtained from the present investigation thus suggest that probably CYP1A1 (M1, M2, M3, and M4) polymorphism alone does not play a significant role in the breast cancer risk in north Indian women.
Similar content being viewed by others

References
Mitrunen K, Hirvonen A (2003) Molecular epidemiology of sporadic breast cancer. The role of polymorphic genes involved in estrogen biosynthesis and metabolism. Mutat Res 544:9–41
McManus ME, Burgess WM, Veronese ME, Huggett A, Quattrochi LC, Tukey RH (1990) Metabolism of 2-acetyleaminofluorene and benzo(a)pyrene and activation of food-derived heterocyclic amine mutagens by human cytochrome P-450. Cancer Res 50:3367–3376
Thompson PA, Ambrosone C (2000) Molecular epidemiology of genetic polymorphisms in estrogen metabolizing enzymes in human breast cancer. J Natl Cancer Inst Monogr 27:125–134
Cavalieri E, Frenkel K, Liehr JG, Rogan E, Roy D (2000) Estrogens as endogenous genotoxic agents-DNA adducts and mutations. J Natl Cancer Inst Monogr 27:75–93
Lakhani NJ, Sarkar MA, Venitz J, Figg WD (2003) 2-Methoxyestradiol, a promising anticancer agent. Pharmacotherapy 23:165–172
Brueggemeier RW, Bhat AS, Lovely CJ, Coughenour HD, Joomprabutra S, Weitzel DH, Vandre DD, Yusuf F, Burak WE Jr (200l) 2-Methoxyestradiol: a new 2-methoxyestrogen analog that exhibit antiproliferative activity and alters tubulin dynamics. J Steroid Biochem Mol Biol 78:145–156
Schneider J, Huh MM, Bradlow HL, Fishman J (1984) Antiestrogen action of 2-hydroxyestrone on MCF-7 human breast cancer cells. J Biol Chem 259:4840–4845
Fotsis T, Zhang Y, Pepper MS, Aldercreutz H, Montesano R, Nawroth PP, Schweilgerer L (1994) The endogenous estrogen metabolite 2-methoxyestradiol inhibits angiogenesis and suppresses tumor growth. Nature 368:237–239
Tabakovic K, Gleason WB, Ojala WH, Abdul-Hajj YJ (1996) Oxidative transformation of 2-hydroxyestrone. Stability and reactivity of 2,3-estrone quinone and its relationship to estrogen carcinogenicity. Chem Res Toxicol 9:860–865
Liehr JG, Ricc MJ (1996) 4-Hydroxylation of estrogens as marker of human mammary tumors. Proc Natl Acad Sci USA 93:3294–3296
Masson LF, Sharp L, Cotton SC, Little J (2005) Cytochrome P-4501A1 gene polymorphisms and risk of breast cancer: a HuGE review. Am J Epidemiol 161:901–915
Kawajiri K, Watanabe J, Gotoh O, Tagashira Y, Sogawa K, Fujii-Kuriyama Y (1986) Structure and drug inducibility of human cytochrome P-450c gene. Eur J Biochem 159:219–225
McKay JA, Murray GI, Ah-See AK, Greenlee WF, Marcus CB, Burke MD, Melvin WT (1996) Differential expression of CYP1A1 and CYP1B1 in human breast cancer. Biochem Soc Trans 24:327S
Boyapati SM, Shu XO, Gao YT, Cai Q, Jin F, Zheng W (2005) Polymorphism in CYP1A1 and breast carcinoma risk in a population based case–control study of Chinese women. Cancer 103:2228–2235
Okobia M, Bunker C, Zmuda J, Kammerer C, Vogel V, Uche E, Anyanwu S, Ezeome E, Ferrell R, Kuller L (2005) Cytochrome P4501A1 genetic polymorphisms and breast cancer risk in Nigerian Women. Breast Cancer Res Treat 94:285–293
Hefler LA, Tempfer CB, Grimm C, Lebrecht A, Ulbrich E, Heinze G, Leodolter S, Schneeberger C, Mueller MW Muendlein A, Koelbl H (2004) Estrogen-metabolizing gene polymorphism in the assessment of breast carcinoma risk and fibrodenema risk in Caucasian Women. Cancer 101:264–269
Modugno F, Zmuda JM, Potter D, Cai C, Ziv E, Cummings SR, Stone KL, Morin PA, Greene D, Cauley JA (2005) Estrogen metabolizing polymorphisms and breast cancer risk among older white women. Breast Cancer Res Treat 93:261–270
Kawajiri K, Nakach K, Imai K, Yoshii A, Shinoda N, Wanatabe J (1990) Identification of genetically high risk individuals to lung cancer by DNA polymorphisms of the cytochrome P-4501A1 gene. FEBS Lett 263:131–133
Hayashi S, Wanatabe J, Nakachi K, Kawajiri K (1991) Genetic linkage of lung associated MspI polymorphisms with amino acid replacement in the heme binding region of the human cytochrome P-450 1A1 gene. J Biochem (Tokyo) 110:407–411
Crofts F, Cosma GN, Currie D, Taioli E, Toniolo P, Garte SJ (1993) A novel CYP1A1 gene polymorphism in African Americans. Carcinogenesis 14:1729–1731
Cascorbi I, Brockmoller J, Roots I (1996) A C4887A polymorphism in exon 7 of human CYP1A1: population frequency, mutation linkages, and impact on lung cancer susceptibility. Cancer Res 56:4965–4969
Tailoi E, Bradlow HL, Garbers SV, Sepkovic DW, Osborne MP, Trachman J, Ganguly S, Garte SJ (1999) Role of estradiol metabolism and CYP1A1 polymorphisms in breast cancer risk. Cancer Detect Prev 23:232–237
Taioli E, Trachman J, Chen X, Toniolo P, Garte SJ (1995) A CYP1A1 restriction fragment length polymorphism is associated with breast cancer in African American women. Cancer Res 55:3757–3758
Rebbeck TR, Rosvold EA, Duggan DJ, Zhang J, Buetow KH (1994) Genetics of CYP1A1: coamplification of specific alleles by polymerase chain reaction and association with breast cancer. Cancer Epidemiol Biomarkers Prev 3:511–514
Ambrosone CB, Freudenheim JL, Graham S, Marshall JR, Vena JE, Brasure JR, Laughlin R, Nemoto T, Michalek AM, Harrington A (1995) Cytochrome P4501A1 and glutathione-S-transferase (M1) genetic polymorphisms and postmenapusal breast cancer risk. Cancer Res 55:3483–3485
Miller SA, Dykes DD, Polesky HF (1988) A simple salting out procedure for extracting DNA from human nucleated cells. Nucl Acids Res 16:1215
Li Y, Millikan RC, Bell DA, Cui L, Tse CK, Newman B, Conway K (2004) Cigarette smoking, Cytochrome P450 polymorphisms, and breast cancer among African-American and white women. Breast Cancer Res 6:R460–R473
Boyum A (1964) Separation of white blood cells. Nature 204:793–794
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Fasco MJ, Treanor CP, Spivack S, Figge HL, Kaminsky LS (1995) Quantitative RNA-polymerase chain reaction – DNA analysis by capillary electrophoresis and laser induced fluorescence. Anal Biochem 224:140–147
Kemp TJ, Causton HC, Clerk A (2003) Changes in gene expression induced by H2O2 in cardiac myocytes. Biochem Biophys Res Commun 307:416–421
Roberta JP, Fouts JR (1980) A rapid method for assaying the metabolism of 7-Ethoxyresorufin by Microsomal Subcellular Fractions. Anal Biochem 107:150–155
Zhu K, Hunter S, Payne-Wilks K, Sutcliffe C, Bentfey C, Roland CL, Williams SM (2006) Potential differences in breast cancer risk factors based on CYP1A1 MspI and African-American-specific genotypes. Ethn Dis 16:207–215
Ishibe N, Hankinson SE, Colditz GA, Spiegelman D, Willett WC, Speizer FE, Kelsey KT, Hunter DJ (1998) Cigarette smoking, cytochrome P4501A1 polymorphisms, and breast cancer risk in the Nurses Health Study. Cancer Res 58:667–671
Bailey LR, Roodi N, Verrier CS, Yee CJ, Dupont WD, Parl FF (1998) Breast cancer and CYP1A1, GSTM1 and GSTT1 polymorphisms: evidence of a lack of association in Caucasians and African Americans. Cancer Res 58:65–70
Chacko P, Joseph T, Mathew BS, Rajan B, Pillai MR (2005) Role of xenobiotic metabolizing gene polymorphisms in breast cancer susceptibility and treatment outcome. Mutation Research 581:153–163
Moysich KB, Shields PG, Freudenheim JL, Schisterman EF, Vena JE, Kostyniak P, Greizerstein H, Marshall JR, Graham S, Ambrosome CB (1999) Polychlorinated biphenyls, cytochrome p4501A1 polymorphism, and postmenopausal breast cancer risk. Cancer Epidemiol Biomarkers Prev 8:41–44
Cribb AE, Kinght MJ, Dryer D, Guernsey J, Hender K, Tesch M, Saleh TM (2006) Role of polymorphic human cytochrome p450 enzymes in estrone oxidation. Cancer Epidemiol Biomarkers Prev 15:551–558
Shen Y, Li DK, Wu J, Zhang Z, Gao E (2006) Joint effects of the CYP1A1 MspI, ER alpha, PvuII, and ER alpha XbaI polymorphisms on the risk of breast cancer: results from a population-based case–control study in Shanghai, China. Cancer Epidemiol Biomarkers Prev 15:342–347
Acknowledgements
Authors sincerely thank University Grant Commission (UGC), New Delhi, India for providing research fellowship to Virendra Singh and Council of Scientific and Industrial Research (CSIR), New Delhi, India for providing research fellowship to Abhai Kumar. Authors also thank Director, ITRC, Lucknow, India for providing necessary facilities required for this study.
Author information
Authors and Affiliations
Corresponding author
Additional information
An erratum to this article is available at http://dx.doi.org/10.1007/s10549-006-9421-7.
Rights and permissions
About this article
Cite this article
Singh, V., Rastogi, N., Sinha, A. et al. A Study on the Association of Cytochrome-P450 1A1 Polymorphism and Breast Cancer Risk in North Indian Women. Breast Cancer Res Treat 101, 73–81 (2007). https://doi.org/10.1007/s10549-006-9264-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10549-006-9264-2