Abstract
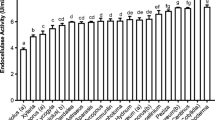
Agaricus brasiliensis CS1, Pleurotus ostreatus H1 and Aspergillus flavus produced holocellulases when grown in solid and submerged liquid cultures containing agro-industrial residues, including sugar cane bagasse and dirty cotton residue, as substrates. These isolates proved to be efficient producers of holocellulases under the conditions used in this screening. Bromatological analysis of agro-industrial residues showed differences in protein, fiber, hemicellulose, cellulose and lignin content. Maximal holocellulase activity (hemicellulase, cellulase and pectinase) was obtained using solid-state cultivation with 10% substrate concentration. In this case, remarkably high levels of xylanase and polygalacturonase activity (4,008 and 4,548 IU/l, respectively) were produced by A. flavus when grown in media containing corn residue, followed by P. ostreatus H1 with IU/l values of 1,900 and 3,965 when cultivated on 5% and 10% sugar cane bagasse, respectively. A. brasiliensis CS1 showed the highest reducing sugar yield (11.640 mg/ml) when grown on medium containing sugar cane bagasse. A. brasiliensis was also the most efficient producer of protein, except when cultivated on dirty cotton residue, which induced maximal production in A. flavus. Comparison of enzymatic hydrolysis of sugar cane bagasse and dirty cotton residue by crude extracts of A. brasiliensis CS1, P. ostreatus H1 and A. flavus showed that the best reducing sugar yield was achieved using sugar cane bagasse as a substrate.




Similar content being viewed by others
References
Andreaus J, Filho EXF, Bon EPS (2008) Biotechnology of holocellulose-degrading enzymes. In: Hou CT, Shaw J-F (eds) Biocatalysis and bioenergy. Willey, United States of America, pp 197–229
Association of Official Analytical Chemists (1995) Official methods of analysis of the association of the analytical chemists. 16th ed. Washington
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein dye binding. Anal Biochem 72:248–254
Cali JP, Bowers GN Jr, Young DS (1973) A Referee method for the determination of total calcium in serum. Clin Chem 19:1208–1213
Cen P, Xia L (1999) Production of cellulose by solid-state fermentation. Adv Biochem Eng 65:70–92
Erden E, Ucar MC, Gezer T, Pazarlioglu NK (2009) Screening for lignolytic enzymes from autochthonous fungi and applications for decolorization of remazole marine blue. Braz J Microbiol 40:346–353
Gomide JL, Demuner EBJ (1986) Determinação do teor de lignina em material lenhoso: método Klason modificado. O papel 47:36–38
Kristensen JB, Felby C, Jørgensen H (2009) Yield-determining factors in high-solids enzymatic hydrolysis of lignocelluloses. Biotechnol Biofuels 2:1–10
Kumar R, Singh S, Singh OV (2008) Bioconversion of lignocellulosic biomass: biochemical and molecular properties. J Ind Microbiol Biotechnol 35:377–391
Mandels M, Andreotti R, Roche C (1976) Measurement of saccharifying cellulose. Biotechnol Bioeng Sympos 16:21–33
Mellon JE, Cotty PJ, Dowd MK (2007) Aspergillus flavus hydrolases: their roles in pathogenesis and substrate utilization. Appl Microbiol Biotechnol 77:497–504
Miller GL (1959) Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal Chem 31:426–428
Roig B, Gonzalez C, Thomas O (1999) Simple UV/UV-visible method for nitrogen and phosphorous measurement in waste water. Talanta 50:751–758
Salles BC, Te’o VSJ, Gibbs MD, Bergquist PL, Filho EXF, Ximenes EA, Nevalainen KMH (2007) Identification of two novel xylnase-encoding genes (xyn5 and xyn6) from Acrophialophora nainiana and heterologous expression of xyn6 in Trichoderma reesei. Bioechnol Lett 29:1195–1201
Sanchez C (2009) Lignocellulosic residues: biodegradation and bioconversion by fungi. Biotechnol Adv 27:185–194
Silva DJ, Queiroz AC (2002) Análise de alimentos: métodos químicos e biológicos. University of Viçosa, Brazil
Siqueira FG, Siqueira EG, Jaramillo PMD, Silveira MLH, Andreaus J, Couto FA, Batista LR, Filho EXF (2010) The potential of agro-industrial residues for production of holocellulase from filamentous fungi. Int Biodeterior Biodegrad 64:20–26
Trinder P (1969) Determination of glucose in blood using glucose oxidase with an alternative oxygen acceptor. Ann Clin Biochem 6:24–25
Tuohy MG, Buckley RJ, Griffin TO, Connelly IC, Shanley NA, Filho EXF, Hughes MM, Grogan P, Coughlan MP (1989) Enzyme production by solid-state cultures of aerobic fungi on lignocellulosic substrates. In: Coughlan MP (ed) Enzyme systems for lignocellulose degradation. Elsevier Applied Science, London, England, pp 293–312
van Soest PJ (1963) Use of detergents in the analyses of fibrous feeds. 2. A rapid method for determination of fiber and lignin. J Assoc Off Anal Chem 46:829–835
van Soest PJ, Wine RH (1967) Use of detergents in the analysis of fibrous feeds IV: determination of plant cell wall constituents. J Assoc Off Anal Chem 50:50–55
Zhang Y-HP, Himmel ME, Mielenz JR (2006) Outlook for cellulase improvement: screening and selection strategies. Biotechnol Adv 24:452–481
Acknowledgements
This work was funded by the Department of Education of Federal District (Brazil), the Foundation for Research Support of the Federal District (Brazil, research grant number 193.000.470/2008) and the Brazilian National Research Council (research grant number 470358/2007-6). E.X.F.F. acknowledges the receipt of research fellowship from Brazilian National Research Council.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
de Siqueira, F.G., de Siqueira, A.G., de Siqueira, E.G. et al. Evaluation of holocellulase production by plant-degrading fungi grown on agro-industrial residues. Biodegradation 21, 815–824 (2010). https://doi.org/10.1007/s10532-010-9346-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10532-010-9346-z