Abstract
Objectives
To explore the role of thioesterases in Rhodococcus opacus PD630 by endogenously overexpression in this bacteria for increased lipid production.
Results
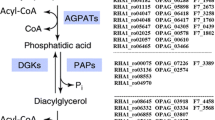
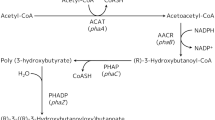
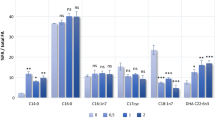
Overexpression of four thioesterases from R. opacus PD630 in E. coli led to a 2- to 8-fold increase in C16:1 and C18:1 fatty acids while, when overexpressed in R. opacus PD630, only two recombinants had significant effect on the quantities and compositions of total fatty acid. The contents of total fatty acids (FAs) in two recombinants, pJTE2 (OPAG_00508 thioesterase) and pJTE4 (WP_012687673.1 thioesterase), were 400–460 mg/g (CDW) which is 1.5 times of wild-type strain PD630 (300-350 mg/g CDW), and 20–30 % (w/w) more than that of the control strain PDpJAM2 (330-370 mg/g CDW). The contents of 17:1 and 18:1 fatty acids increased by about 27 and 35 %, respectively, in pJTE2 and by 35 and 20 %, respectively, in pJTE4 compared with the control strain.
Conclusions
The engineered strains showed improved production of lipid (as total fatty acids), and could also tailor the composition of the fatty acid profile when cultured in mineral salts medium using glucose as sole carbon source.





Similar content being viewed by others
References
Alvarez HM, Kalscheuer R, Steinbuchel A (2000) Accumulation and mobilization of storage lipids by Rhodococcus opacus PD630 and Rhodococcus ruber NCIMB 40126. Appl Microbiol Biotechnol 54:218–223
Alvarez AF, Alvarez HM, Kalscheuer R, Waeltermann M, Steinbuechel A (2008) Cloning and characterization of a gene involved in triacylglycerol biosynthesis and identification of additional homologous genes in the oleaginous bacterium Rhodococcus opacus PD630. Microbiology 154:2327–2335
Burnette WN (1981) “Western blotting”: electrophoretic transfer of proteins from sodium dodecyl sulfate-polyacrylamide gels to unmodified nitrocellulose and radiographic detection with antibody and radioiodinated protein A. Anim Biochem 112:195–203
Cantu CD, Chen Y, Reilly JP (2010) Thioesterases: a new perspective based on their primary and tertiary structures. Prot Sci 19:1281–1295
Fan L, Liu J, Nie K, Liu L, Wang F, Tan T, Deng L (2013) Synthesis of medium chain length fatty acid ethyl esters in engineered Escherichia coli using endogenously produced medium chain fatty acids. Enz Microb Technol 53:128–133
Hanisch J, Waltermann M, Robenek H, Steinbüchel A (2006) The Ralstonia eutropha H16 phasin PhaP1 is targeted to intracellular triacylglycerol inclusions in Rhodococcus opacus PD630 and Mycobacterium smegmatis mc2155, and provides an anchor to target other proteins. Microbiology 152:3271–3280
Hernandez MA, Arabolaza A, Rodriguez E, Gramajo H, Alvarez HM (2013) The atf2 gene is involved in triacylglycerol biosynthesis and accumulation in the oleaginous Rhodococcus opacus PD630. Appl Microbiol Biotechnol 97:2119–2130
Hernandez MA, Comba S, Arabolaza A, Gramajo H, Alvarez HM (2015) Overexpression of a phosphatidic acid phosphatase type 2 leads to an increase in triacylglycerol production in oleaginous Rhodococcus strains. Appl Microbiol Biotechnol 99:2191–2207
Hetzler S, Broeker D, Steinbüchel A (2013) Saccharification of cellulose by recombinant Rhodococcus opacus PD630 strains. Appl Environ Microbiol 79:5159–5166
Holder JW, Ulrich JC, Debono AC et al (2011) Comparative and functional genomics of Rhodococcus opacus PD630 for biofuels development. PLoS Genet 7(9):e1002219
Jiang P, CronanJr JE (1994) Inhibition of fatty acid synthesis in Escherichia coli in the absence of phospholipid synthesis and release of inhibition by thioesterase action. Bacteriol 176:2814–2821
Jing F, David CC, Jarmila T, Jay PC, Basil JN, Marna DY, Peter JR (2011) Phylogenetic and experimental characterization of an acyl-ACP thioesterase family reveals significant diversity in enzymatic specificity and activity. BMC Biochem 12:44–59
Kalscheuer R, Arenskötter M, Steinbüchel A (1999) Establishment of a gene transfer system for Rhodococcus opacus PD630 based on electroporation and its application for recombinant biosynthesis of poly-3-hydroxyalkanoic acids. Appl Microbiol Biotechnol 52:508–515
Lee S, Park S, Lee J (2013) Improvement of fatty acid production in Escherichia coli using codon-optimized Streptococcus pyogenes acyl-ACP thioesterase. Bioprocess Biosyst Eng 36(10):1519–1525
Ratledge C, Wynn JP (2002) The biochemistry and molecular biology of lipid accumulation in oleaginous microorganisms. Adv Appl Microbiol 51:1–51
Sakuradani E, Shimizu S (2003) Gene cloning and functional analysis of a second delta 6-fatty acid desaturase from an arachidonic acid-producing Mortierella fungus. Biosci Biotechnol Biochem 67(4):704–711
Schlegel HG, Kaltwasser H, Gottschalk G (1961) Ein Submersverfahren zur Kultur wasserstoffoxydierender Bakterien: wachstumsphysiologische Untersuchungen. Arch Mikrobiol 38:209–222
Serrano-Vega MJ, Garces R, Martinez-Force E (2005) Cloning, characterization and structural model of a FatA-type thioesterase from sunflower seeds. Planta 221:868–880
Shao Z, Dick WA, Behki RM (1995) An improved Escherichia coli-Rhodococcus shuttle vector and plasmid transformation in Rhodococcus spp. using electroporation. Lett Appl Microbiol 21:261–266
Torella JP, Ford TJ, Kim SN, Chen AM, Way JC, Silver PA (2013) Tailored fatty acid synthesis via dynamic control of fatty acid elongation. Proc Natl Acad Sci 110:11290–11295
Triccas JA, Parish T, Britton WJ, Giquel B (1998) An inducible expression system permitting the efficient purification of a recombinant antigen from Mycobacterium smegmatis. FEMS Microbiol Lett 167:151–156
Villalba MS, Alvarez HM (2014) Identification of a novel ATP-binding cassette transporter involved in long-chain fatty acid import and its role in triacylglycerol accumulation in Rhodococcus jostii RHA1. Microbiology 160:1523–1532
Zhang X, Li A, Agrawal A, San KY (2011) Efficient free acid production in Escherichia coli using plant acyl-ACP thioesterases. Metab Eng 13:713–722
Zheng Y, Li L, Liu Q et al (2012) Boosting the fatty acid synthesis of Escherichia coli by expression of a cytosolic Acinetobacter baylyi thioesterase. Biotechnol Biofuel 5:76–88
Acknowledgments
This study was funded by the National Natural Science Foundation of China (31271812), the National High Technology Research and Development Program of China (2012AA022105C), and the Strategic Merieux Research Grant.
Supporting Information
Supplementary Table 1—List of plasmids and primers used.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical statement
This article does not contain any studies with human participants or animals performed by any of the authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Huang, L., Zhao, L., Zan, X. et al. Boosting fatty acid synthesis in Rhodococcus opacus PD630 by overexpression of autologous thioesterases. Biotechnol Lett 38, 999–1008 (2016). https://doi.org/10.1007/s10529-016-2072-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-016-2072-9