Abstract
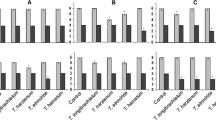
Trichoderma spp. are known for their biocontrol activity against several plant pathogens. A specific isolate of Trichoderma harzianum, 303/02, has the potential to inhibit the growth of Sclerotinia sclerotiorum, an important agent involved in several crop diseases. In this study, the interaction between T. harzianum 303/02 and mycelia, sclerotia and apothecia of S. sclerotiorum was studied by scanning electron microscopy. RT-qPCR was used to examine the expression of 11 genes potentially involved in biocontrol. T. harzianum 303/02 parasitizes S. sclerotiorum by forming branches that coil around the hyphae. The fungus multiplied abundantly at the sclerotia and apothecia surface, forming a dense mycelium that penetrated the inner surface of these structures. The levels of gene expression varied according to the type of structure with which T. harzianum was interacting. The data also showed the presence of synergistic action between the cell-wall degrading enzymes.


Similar content being viewed by others
References
Ait-Lahsen H, Soler A, Rey M De, La Cruz J, Monte E, Llobel A (2001) An antifungal exo-α-1,3-glucanase (AGN13.1) from the biocontrol fungus Trichoderma harzianum. Appl Environ Microbiol 67:5833–5839
Almeida FB, Cerqueira FM, Silva RN, Ulhoa CJ (2007) Mycoparasitism studies of Trichoderma harzianum strains against Rhizoctonia solani: evaluation of coiling and hydrolytic enzyme production. Biotechnol Lett 29:1189–1193
Amin F, Razdan VK, Mohid FA, Bhat KA, Banday S (2010) Potential of Trichoderma species as biocontrol agents of soil borne fungal propagules. J Phytol 10:38–41
Benhamou N, Chet I (1996) Parasitism of sclerotia of Sclerotium rolfsii by Trichoderma harzianum: ultrastructural and cytochemical aspects of the interaction. Phytophatology 86:405–416
Benitez T, Rincon AM, Limon MC, Codon AC (2004) Biocontrol mechanisms of Trichoderma strains. Intern Microbiol 7:249–260
Boland GJ, Hall R (1994) Index of plant hosts of Sclerotinia sclerotiorum. Can J Plant Pathol 16:93–108
Bowman SM, Free SJ (2006) The structure and synthesis of the fungal cell wall. Bio Essays 28:799–808
De La Cruz J, Hidalgo-Gallego A, Lora JM, Benitez T, Pintor-Toro JA, Llobell A (1992) Isolation and characterization of three chitinases from Trichoderma harzianum. Eur J Biochem 206:859–867
Djonovic S, Vittone G, Mendoza-Herrera A, Kenerley CM (2007) Enhanced biocontrol activity of Trichoderma virens transformants constitutively coexpressing beta-1,3- and beta-1,6-glucanase genes. Mol Plant Pathol 8:469–480
Druzhinina IS, Kubicek CP, Komoń-Zelazowska M, Mulaw TB, Bissett J (2010) The Trichoderma harzianum demon: complex speciation history resulting in coexistence of hypothetical biological species, recent agamospecies and numerous relict lineages. BMC Evol Biol 1(10):94
Druzhinina IS, Seidl-Seiboth V, Herrera-Estrella A, Horwitz BA, Kenerley CM, Monte E, Mukherjee PK, Zeilinger S, Grigoriev IV, Kubicek CP (2011) Trichoderma: the genomics of opportunistic success. Nat Rev Microbiol 9:749–759
Dubey SC, Bhavani R, Singh B (2011) Integration of soil application and seed treatment formulations of Trichoderma species for management of wet root rot of mungbean caused by Rhizoctonia solani. Pest Manag 67:1163–1168
Eneyskaya EV, Kulminskaya AA, Savel’ev AN, Shabalin KA, Golubev AM, Neustroev KN (1998) Alpha-Mannosidase from Trichoderma reesei participates in the postsecretory deglycosylation of glycoproteins. Biochem Biophys Res Commun. 245:43–49
Garcia RA, Juliatti FC, Barbosa KAG (2013) Effect of fungicides and herbicides in the control of Sclerotinia sclerotiorum. Biosci J 29:1989–1996
Geraldine AM, Lopes FAC, Carvalho DDC, Barbosa ET, Rodrigues AR, Brandão RS, Ulhoa CJ, Lobo-Junior M (2013) Cell wall-degrading enzymes and parasitism of sclerotia are key factors on field biocontrol of white mold by Trichoderma spp. Biol Control 67:308–316
Haran S, Schickler H, Oppenheim A, Chet I (1996) Differential expression of Trichoderma harzianum chitinases during mycoparasitism. Phytopathology 86:980–985
Jones D (1970) Ultrastructure and composition of the cell walls of Sclerotinia sclerotiorum. Trans Br Mycol Soc 54:351–360
Kullnig C, Mach RL, Lorito M, Kubicek CP (2000) Enzyme division from Trichoderma atroviride (=T. harzianum P1) to Rhizoctonia solani is a prerequisite for triggering of Trichoderma ech42 gene expression before mycoparasitic contact. Applied and Environ Microbiol 66:2232–2234
Liu Y, Yang Q (2007) Cloning and heterlogous expression of aspartic protease SA76 related to biocontrol in Trichoderma harzianum. FEMS Microbiol Lett 277:173–181
Liu Y, Yang Q, Song J (2009) A new serine protease gene from Trichoderma harzianum is expressed in Saccharomyces cerevisiae. Prikl Biokhim Mikrobiol 45:28–32
Liu Z, Yang X, Sun D, Song J, Chen G, Juba O, Yang Q (2010) Expressed sequence tags-based identification of genes in a biocontrol strain Trichoderma asperellum. Mol Biol Rep 37:3673–3681
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(−Delta Delta C(T)). Methods 25:402–408
Lopes FAC, Steindorff AS, Geraldine AM, Brandão RS, Monteiro VN, Lobo M Jr, Coelho ASG, Ulhoa CJ, Silva RN (2012) Biochemical and metabolic profiles of Trichoderma strains isolated from common bean crops in the Brazilian Cerrado, and potential antagonism against Sclerotinia sclerotiorum. Fungal Biol. 116:815–824
Mellado E, Dubreucq G, Mol P, Sarfati J, Paris S, Diaquin M, Holden DW, Rodriguez-Tudela JL, Latgé JP (2003) Cell wall biogenesis in a double chitin synthase mutant (chsG-/chsE-) of Aspergillus fumigatus. Fungal Genet Biol 38:98–109
Neustroyev KN, Firsov LM (1990) Acid proteinase and the multiplicity of Aspergillus awamori glucoamylase forms. Biokhimiya 55:776–785
Qualhato TF, Lopes FA, Steindorff AS, Brandão RS, Jesuino RS, Ulhoa CJ (2013) Mycoparasitism studies of Trichoderma species against three phytopathogenic fungi: evaluation of antagonism and hydrolytic enzyme production. Biotechnol Lett 35:1461–1468
Sanz L, Montero M, Redondo J, Llobell A, Mont E (2005) Expression of an alpha-1,3-glucanase during mycoparasitic interaction of Trichoderma asperellum. FEBS J 272:493–499
Steindorff A, Ramada MH, Coelho AS, Miller RN, Pappas G, Ulhoa CJ, Noronha EF (2014) Identification of mycoparasitism-related genes against the phytopathogen Sclerotinia sclerotiorum through transcriptome and expression profile analysis in Trichoderma harzianum. BMC Genom 15:204
Vieira PM, Coelho AS, Steindorff AS, de Siqueira SJ, Silva RN, Ulhoa CJ (2013) Identification of differentially expressed genes from Trichoderma harzianum during growth on cell wall of Fusarium solani as a tool for biotechnological application. BMC Gen 14:177
Viterbo A, Harel M, Chet I (2004) Isolation of two aspartyl proteases from Trichoderma asperellum expressed during colonization of cucumber roots. FEMS Microbiol Lett 238:151–158
Wei H, Scherer M, Singh A, Liese R, Fischer R (2001) Aspergillus nidulans alpha-1,3 glucanase (mutanase), mutA, is expressed during sexual development and mobilizes mutan. Fungal Genet Biol 34:217–227
Yang DQ (2005) Isolation of wood-inhabiting fungi from Canadian hardwood logs. Can J Microbiol 51:1–6
Acknowledgments
This work was funded by FINEP (Research and Projects Financing), the National Council for Scientific and Technological Development (CNPq) and the State of Goiás Research Foundation (FAPEGO). T.F.Q. was supported by a scholarship from CNPq. We kindly thank Dr. Robert Poque for reading the manuscript and providing suggestions.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Troian, R.F., Steindorff, A.S., Ramada, M.H.S. et al. Mycoparasitism studies of Trichoderma harzianum against Sclerotinia sclerotiorum: evaluation of antagonism and expression of cell wall-degrading enzymes genes. Biotechnol Lett 36, 2095–2101 (2014). https://doi.org/10.1007/s10529-014-1583-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-014-1583-5