Abstract
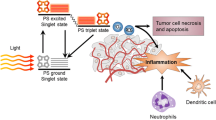
Photodynamic therapy (PDT) utilizes the destructive power of reactive oxygen species generated via visible light irradiation of a photosensitive dye accumulated in the cancerous tissue/cells, to bring about their obliteration. PDT activates multiple signalling pathways in cancer cells, which could give rise to all three cell death modalities (at least in vitro). Simultaneously, PDT is capable of eliciting various effects in the tumour microenvironment thereby affecting the tumour-associated/-infiltrating immune cells and by extension, leading to infiltration of various immune cells (e.g. neutrophils) into the treated site. PDT is also associated to the activation of different immune phenomena, e.g. acute-phase response, complement cascade and production of cytokines/chemokines. It has also come to light that, PDT is capable of activating ‘anti-tumour adaptive immunity’ in both pre-clinical as well as clinical settings. Although the ability of PDT to induce ‘anti-cancer vaccine effect’ is still debatable, yet it has been shown to be capable of inducing exposure/release of certain damage-associated molecular patterns (DAMPs) like HSP70. Therefore, it seems that PDT is unique among other approved therapeutic procedures in generating a microenvironment suitable for development of systemic anti-tumour immunity. Apart from this, recent times have seen the emergence of certain promising modalities based on PDT like-photoimmunotherapy and PDT-based cancer vaccines. This review mainly discusses the effects exerted by PDT on cancer cells, immune cells as well as tumour microenvironment in terms of anti-tumour immunity. The ability of PDT to expose/release DAMPs and the future perspectives of this paradigm have also been discussed.


Similar content being viewed by others
Abbreviations
- AIF:
-
Apoptosis-inducing factor
- ALA:
-
Amino levulinic acid
- APCs:
-
Antigen-presenting cells
- APR:
-
Acute-phase response
- ATP:
-
Adenosine triphosphate
- CD:
-
Cluster of differentiation
- DAMP:
-
Damage-associated molecular patterns
- DC:
-
Dendritic cell
- ER:
-
Endoplasmic reticulum
- GRP:
-
Glucose-regulated protein
- HIF:
-
Hypoxia-inducible factor
- HMGB1:
-
High-mobility group box-1
- HSP:
-
Heat shock protein
- IFN:
-
Interferon
- IL:
-
Interleukin
- MAPK:
-
Mitogen-activated protein kinase
- MHC:
-
Major histocompatibility complex
- MMP:
-
Mitochondrial membrane permeabilization
- NK cells:
-
Natural killer cells
- NO:
-
Nitric oxide
- PDT:
-
Photodynamic therapy
- RIP1:
-
Receptor interacting protein 1
- ROS:
-
Reactive oxygen species
- TAA:
-
Tumour-associated antigen(s)
- TGF:
-
Transforming growth factor
- TLR:
-
Toll-like receptor(s)
- TNF:
-
Tumour necrosis factor
References
Buytaert E, Dewaele M, Agostinis P (2007) Molecular effectors of multiple cell death pathways initiated by photodynamic therapy. Biochim Biophys Acta 1776:86–107
Castano AP, Mroz P, Hamblin MR (2006) Photodynamic therapy and anti-tumour immunity. Nat Rev Cancer 6:535–545
Korbelik M (2006) PDT-associated host response and its role in the therapy outcome. Lasers Surg Med 38:500–508
Golab J, Wilczynski G, Zagozdzon R et al (2000) Potentiation of the anti-tumour effects of Photofrin-based photodynamic therapy by localized treatment with G-CSF. Br J Cancer 82:1485–1491
Nowis D, Makowski M, Stoklosa T, Legat M, Issat T, Golab J (2005) Direct tumor damage mechanisms of photodynamic therapy. Acta Biochim Pol 52:339–352
Agostinis P, Vantieghem A, Merlevede W, de Witte PA (2002) Hypericin in cancer treatment: more light on the way. Int J Biochem Cell Biol 34:221–241
Zhou F, Xing D, Wu S, Chen WR (2010) Intravital imaging of tumor apoptosis with FRET probes during tumor therapy. Mol Imaging Biol 12:63–70
Bhuvaneswari R, Gan YY, Soo KC, Olivo M (2009) Targeting EGFR with photodynamic therapy in combination with Erbitux enhances in vivo bladder tumor response. Mol Cancer 8:94
Banihashemi B, Vlad R, Debeljevic B, Giles A, Kolios MC, Czarnota GJ (2008) Ultrasound imaging of apoptosis in tumor response: novel preclinical monitoring of photodynamic therapy effects. Cancer Res 68:8590–8596
Chen Y, Zheng W, Li Y, Zhong J, Ji J, Shen P (2008) Apoptosis induced by methylene-blue-mediated photodynamic therapy in melanomas and the involvement of mitochondrial dysfunction revealed by proteomics. Cancer Sci 99:2019–2027
Henderson BW, Waldow SM, Mang TS, Potter WR, Malone PB, Dougherty TJ (1985) Tumor destruction and kinetics of tumor cell death in two experimental mouse tumors following photodynamic therapy. Cancer Res 45:572–576
Oleinick NL, Morris RL, Belichenko I (2002) The role of apoptosis in response to photodynamic therapy: what, where, why, and how. Photochem Photobiol Sci 1:1–21
Buytaert E, Callewaert G, Hendrickx N et al (2006) Role of endoplasmic reticulum depletion and multidomain proapoptotic BAX and BAK proteins in shaping cell death after hypericin-mediated photodynamic therapy. FASEB J 20:756–758
Buytaert E, Callewaert G, Vandenheede JR, Agostinis P (2006) Deficiency in apoptotic effectors Bax and Bak reveals an autophagic cell death pathway initiated by photodamage to the endoplasmic reticulum. Autophagy 2:238–240
Kessel D, Castelli M (2001) Evidence that bcl-2 is the target of three photosensitizers that induce a rapid apoptotic response. Photochem Photobiol 74:318–322
Puthalakath H, O’Reilly LA, Gunn P et al (2007) ER stress triggers apoptosis by activating BH3-only protein Bim. Cell 129:1337–1349
Vanlangenakker N, Vanden Berghe T, Krysko DV, Festjens N, Vandenabeele P (2008) Molecular mechanisms and pathophysiology of necrotic cell death. Curr Mol Med 8:207–220
Martinet W, Agostinis P, Vanhoecke B, Dewaele M, De Meyer GR (2009) Autophagy in disease: a double-edged sword with therapeutic potential. Clin Sci (Lond) 116:697–712
Seshadri M, Spernyak JA, Mazurchuk R et al (2005) Tumor vascular response to photodynamic therapy and the antivascular agent 5, 6-dimethylxanthenone-4-acetic acid: implications for combination therapy. Clin Cancer Res 11:4241–4250
Ferrario A, von Tiehl KF, Rucker N, Schwarz MA, Gill PS, Gomer CJ (2000) Antiangiogenic treatment enhances photodynamic therapy responsiveness in a mouse mammary carcinoma. Cancer Res 60:4066–4069
Korbelik M, Krosl G, Krosl J, Dougherty GJ (1996) The role of host lymphoid populations in the response of mouse EMT6 tumor to photodynamic therapy. Cancer Res 56:5647–5652
Sitnik TM, Hampton JA, Henderson BW (1998) Reduction of tumour oxygenation during and after photodynamic therapy in vivo: effects of fluence rate. Br J Cancer 77:1386–1394
Foster TH, Murant RS, Bryant RG, Knox RS, Gibson SL, Hilf R (1991) Oxygen consumption and diffusion effects in photodynamic therapy. Radiat Res 126:296–303
Henderson BW, Busch TM, Vaughan LA et al (2000) Photofrin photodynamic therapy can significantly deplete or preserve oxygenation in human basal cell carcinomas during treatment, depending on fluence rate. Cancer Res 60:525–529
Busch TM, Hahn SM, Evans SM, Koch CJ (2000) Depletion of tumor oxygenation during photodynamic therapy: detection by the hypoxia marker EF3 [2-(2-nitroimidazol-1[H]-yl)-N-(3, 3, 3-trifluoropropyl)acetamide]. Cancer Res 60:2636–2642
Bertout JA, Patel SA, Simon MC (2008) The impact of O2 availability on human cancer. Nat Rev Cancer 8:967–975
Mitra S, Cassar SE, Niles DJ, Puskas JA, Frelinger JG, Foster TH (2006) Photodynamic therapy mediates the oxygen-independent activation of hypoxia-inducible factor 1alpha. Mol Cancer Ther 5:3268–3274
Bil J, Wlodarski P, Winiarska M et al (2009) Photodynamic therapy-driven induction of suicide cytosine deaminase gene. Cancer Lett. doi:10.1016/j.canlet.2009.09.012
Nizet V, Johnson RS (2009) Interdependence of hypoxic and innate immune responses. Nat Rev Immunol 9:609–617
Peyssonnaux C, Datta V, Cramer T et al (2005) HIF-1alpha expression regulates the bactericidal capacity of phagocytes. J Clin Invest 115:1806–1815
Kuhlicke J, Frick JS, Morote-Garcia JC, Rosenberger P, Eltzschig HK (2007) Hypoxia inducible factor (HIF)-1 coordinates induction of Toll-like receptors TLR2 and TLR6 during hypoxia. PLoS One 2:e1364
Jantsch J, Chakravortty D, Turza N et al (2008) Hypoxia and hypoxia-inducible factor-1 alpha modulate lipopolysaccharide-induced dendritic cell activation and function. J Immunol 180:4697–4705
Qiang YG, Yow CM, Huang Z (2008) Combination of photodynamic therapy and immunomodulation: current status and future trends. Med Res Rev 28:632–644
Canti G, Lattuada D, Nicolin A, Taroni P, Valentini G, Cubeddu R (1994) Antitumor immunity induced by photodynamic therapy with aluminum disulfonated phthalocyanines and laser light. Anticancer Drugs 5:443–447
Korbelik M, Dougherty GJ (1999) Photodynamic therapy-mediated immune response against subcutaneous mouse tumors. Cancer Res 59:1941–1946
Chen WR, Singhal AK, Liu H, Nordquist RE (2001) Antitumor immunity induced by laser immunotherapy and its adoptive transfer. Cancer Res 61:459–461
Abdel-Hady ES, Martin-Hirsch P, Duggan-Keen M et al (2001) Immunological and viral factors associated with the response of vulval intraepithelial neoplasia to photodynamic therapy. Cancer Res 61:192–196
Wnag S, Keltner L, Winship J, Lee W (2009) A phase 1/2 safety and efficacy study of intratumoral light-activated drug therapy using talaporfin sodium in patients with inoperable hepatocellular carcinoma. J Clin Oncol 27(suppl.): abstract e15684
Ortner MA, Liebetruth J, Schreiber S et al (1998) Photodynamic therapy of nonresectable cholangiocarcinoma. Gastroenterology 114:536–542
Dragieva G, Prinz BM, Hafner J et al (2004) A randomized controlled clinical trial of topical photodynamic therapy with methyl aminolaevulinate in the treatment of actinic keratoses in transplant recipients. Br J Dermatol 151:196–200
de Graaf YG, Kennedy C, Wolterbeek R, Collen AF, Willemze R, Bouwes Bavinck JN (2006) Photodynamic therapy does not prevent cutaneous squamous-cell carcinoma in organ-transplant recipients: results of a randomized-controlled trial. J Invest Dermatol 126:569–574
Yom SS, Busch TM, Friedberg JS et al (2003) Elevated serum cytokine levels in mesothelioma patients who have undergone pleurectomy or extrapleural pneumonectomy and adjuvant intraoperative photodynamic therapy. Photochem Photobiol 78:75–81
Nseyo UO, Whalen RK, Duncan MR, Berman B, Lundahl SL (1990) Urinary cytokines following photodynamic therapy for bladder cancer. A preliminary report. Urology 36:167–171
Thong PS, Ong KW, Goh NS et al (2007) Photodynamic-therapy-activated immune response against distant untreated tumours in recurrent angiosarcoma. Lancet Oncol 8:950–952
Kabingu E, Oseroff AR, Wilding GE, Gollnick SO (2009) Enhanced systemic immune reactivity to a Basal cell carcinoma associated antigen following photodynamic therapy. Clin Cancer Res 15:4460–4466
Evans D (2005) Suppression of the acute-phase response as a biological mechanism for the placebo effect. Med Hypotheses 64:1–7
Jolles CJ, Ott MJ, Straight RC, Lynch DH (1988) Systemic immunosuppression induced by peritoneal photodynamic therapy. Am J Obstet Gynecol 158:1446–1453
Cecic I, Stott B, Korbelik M (2006) Acute phase response-associated systemic neutrophil mobilization in mice bearing tumors treated by photodynamic therapy. Int Immunopharmacol 6:1259–1266
Fingar VH, Wieman TJ, Wiehle SA, Cerrito PB (1992) The role of microvascular damage in photodynamic therapy: the effect of treatment on vessel constriction, permeability, and leukocyte adhesion. Cancer Res 52:4914–4921
Sun J, Cecic I, Parkins CS, Korbelik M (2002) Neutrophils as inflammatory and immune effectors in photodynamic therapy-treated mouse SCCVII tumours. Photochem Photobiol Sci 1:690–695
Korbelik M, Cecic I (1999) Contribution of myeloid and lymphoid host cells to the curative outcome of mouse sarcoma treatment by photodynamic therapy. Cancer Lett 137:91–98
de Vree WJ, Essers MC, Koster JF, Sluiter W (1997) Role of interleukin 1 and granulocyte colony-stimulating factor in photofrin-based photodynamic therapy of rat rhabdomyosarcoma tumors. Cancer Res 57:2555–2558
Fingar VH, Siegel KA, Wieman TJ, Doak KW (1993) The effects of thromboxane inhibitors on the microvascular and tumor response to photodynamic therapy. Photochem Photobiol 58:393–399
Cecic I, Korbelik M (2002) Mediators of peripheral blood neutrophilia induced by photodynamic therapy of solid tumors. Cancer Lett 183:43–51
de Vree WJ, Essers MC, de Bruijn HS, Star WM, Koster JF, Sluiter W (1996) Evidence for an important role of neutrophils in the efficacy of photodynamic therapy in vivo. Cancer Res 56:2908–2911
Krosl G, Korbelik M, Krosl J, Dougherty GJ (1996) Potentiation of photodynamic therapy-elicited antitumor response by localized treatment with granulocyte-macrophage colony-stimulating factor. Cancer Res 56:3281–3286
De Vree WJ, de Bruijn HS, Kraak-Slee G, Koster JF, Sluiter W (2001) The effect of thrombocytopenia on the efficacy of Photofrin-based photodynamic therapy in vivo. Proc SPIE Int Soc Opt Eng 4146:79–85
Kousis PC, Henderson BW, Maier PG, Gollnick SO (2007) Photodynamic therapy enhancement of antitumor immunity is regulated by neutrophils. Cancer Res 67:10501–10510
Yang D, de la Rosa G, Tewary P, Oppenheim JJ (2009) Alarmins link neutrophils and dendritic cells. Trends Immunol 30:531–537
Di Carlo E, Forni G, Lollini P, Colombo MP, Modesti A, Musiani P (2001) The intriguing role of polymorphonuclear neutrophils in antitumor reactions. Blood 97:339–345
Lim HW (1989) Role mediators of inflammation and cells in porphyrin-induced phototoxicity. SPIE Conf Proc 1065:28–33
Korbelik M (2009) Complement upregulation in photodynamic therapy-treated tumors: role of Toll-like receptor pathway and NF kappa B. Cancer Lett 281:232–238
Korbelik M, Stott B, Sun J, Cecic I (2004) Localized production of complement proteins in tumors treated by photodynamic therapy. Mol Immunol 41:260
Korbelik M, Cecic I (2008) Complement activation cascade and its regulation: relevance for the response of solid tumors to photodynamic therapy. J Photochem Photobiol B 93:53–59
Ferrario A, Fisher AM, Rucker N, Gomer CJ (2005) Celecoxib and NS-398 enhance photodynamic therapy by increasing in vitro apoptosis and decreasing in vivo inflammatory and angiogenic factors. Cancer Res 65:9473–9478
Ferrario A, Chantrain CF, von Tiehl K et al (2004) The matrix metalloproteinase inhibitor prinomastat enhances photodynamic therapy responsiveness in a mouse tumor model. Cancer Res 64:2328–2332
Galli SJ, Nakae S, Tsai M (2005) Mast cells in the development of adaptive immune responses. Nat Immunol 6:135–142
Hume DA (2008) Macrophages as APC and the dendritic cell myth. J Immunol 181:5829–5835
Gollnick SO, Vaughan L, Henderson BW (2002) Generation of effective antitumor vaccines using photodynamic therapy. Cancer Res 62:1604–1608
Jalili A, Makowski M, Switaj T et al (2004) Effective photoimmunotherapy of murine colon carcinoma induced by the combination of photodynamic therapy and dendritic cells. Clin Cancer Res 10:4498–4508
Gollnick SO, Owczarczak B, Maier P (2006) Photodynamic therapy and anti-tumor immunity. Lasers Surg Med 38:509–515
Buytaert E, Matroule JY, Durinck S et al (2008) Molecular effectors and modulators of hypericin-mediated cell death in bladder cancer cells. Oncogene 27:1916–1929
Kocanova S, Buytaert E, Matroule JY et al (2007) Induction of heme-oxygenase 1 requires the p38MAPK and PI3K pathways and suppresses apoptotic cell death following hypericin-mediated photodynamic therapy. Apoptosis 12:731–741
Hyatt G, Melamed R, Park R et al (2006) Gene expression microarrays: glimpses of the immunological genome. Nat Immunol 7:686–691
Garg AD, Nowis D, Golab J, Vandenabeele P, Krysko DV, Agostinis P (2010) Immunogenic cell death, DAMPs and anticancer therapeutics: an emerging amalgamation. Biochim Biophys Acta 1805:53–71
Eruslanov E, Kaliberov S, Daurkin I et al (2009) Altered expression of 15-hydroxyprostaglandin dehydrogenase in tumor-infiltrated CD11b myeloid cells: a mechanism for immune evasion in cancer. J Immunol 182:7548–7557
Grivennikov S, Karin M (2008) Autocrine IL-6 signaling: a key event in tumorigenesis? Cancer Cell 13:7–9
Sansone P, Storci G, Tavolari S et al (2007) IL-6 triggers malignant features in mammospheres from human ductal breast carcinoma and normal mammary gland. J Clin Invest 117:3988–4002
Sato Y, Goto Y, Narita N, Hoon DS (2009) Cancer cells expressing toll-like receptors and the tumor microenvironment. Cancer Microenviron 2(Suppl 1):205–214
Szczepanski M, Stelmachowska M, Stryczynski L et al (2007) Assessment of expression of toll-like receptors 2, 3 and 4 in laryngeal carcinoma. Eur Arch Otorhinolaryngol 264:525–530
Goto Y, Arigami T, Kitago M et al (2008) Activation of Toll-like receptors 2, 3, and 4 on human melanoma cells induces inflammatory factors. Mol Cancer Ther 7:3642–3653
Bianchi ME (2007) DAMPs, PAMPs and alarmins: all we need to know about danger. J Leukoc Biol 81:1–5
Nicaise V, Roux M, Zipfel C (2009) Recent advances in PAMP-triggered immunity against bacteria: pattern recognition receptors watch over and raise the alarm. Plant Physiol 150:1638–1647
Rubartelli A, Lotze MT (2007) Inside, outside, upside down: damage-associated molecular-pattern molecules (DAMPs) and redox. Trends Immunol 28:429–436
Elliott MR, Chekeni FB, Trampont PC et al (2009) Nucleotides released by apoptotic cells act as a find-me signal to promote phagocytic clearance. Nature 461:282–286
Lanneau D, Brunet M, Frisan E, Solary E, Fontenay M, Garrido C (2008) Heat shock proteins: essential proteins for apoptosis regulation. J Cell Mol Med 12:743–761
Schmitt E, Gehrmann M, Brunet M, Multhoff G, Garrido C (2007) Intracellular and extracellular functions of heat shock proteins: repercussions in cancer therapy. J Leukoc Biol 81:15–27
Korbelik M, Sun J, Cecic I (2005) Photodynamic therapy-induced cell surface expression and release of heat shock proteins: relevance for tumor response. Cancer Res 65:1018–1026
Korbelik M, Sun JH (2006) Photodynamic therapy-generated vaccine for cancer therapy. Cancer Immunol Immunother 55:900–909
Zhou F, Xing D, Chen WR (2008) Dynamics and mechanism of HSP70 translocation induced by photodynamic therapy treatment. Cancer Lett 264:135–144
Calderwood SK, Mambula SS, Gray PJ Jr (2007) Extracellular heat shock proteins in cell signaling and immunity. Ann N Y Acad Sci 1113:28–39
Lancaster GI, Febbraio MA (2005) Exosome-dependent trafficking of HSP70: a novel secretory pathway for cellular stress proteins. J Biol Chem 280:23349–23355
Gastpar R, Gehrmann M, Bausero MA et al (2005) Heat shock protein 70 surface-positive tumor exosomes stimulate migratory and cytolytic activity of natural killer cells. Cancer Res 65:5238–5247
Doody AD, Kovalchin JT, Mihalyo MA, Hagymasi AT, Drake CG, Adler AJ (2004) Glycoprotein 96 can chaperone both MHC class I- and class II-restricted epitopes for in vivo presentation, but selectively primes CD8+ T cell effector function. J Immunol 172:6087–6092
Singh-Jasuja H, Toes RE, Spee P et al (2000) Cross-presentation of glycoprotein 96-associated antigens on major histocompatibility complex class I molecules requires receptor-mediated endocytosis. J Exp Med 191:1965–1974
Tsutsumi S, Neckers L (2007) Extracellular heat shock protein 90: a role for a molecular chaperone in cell motility and cancer metastasis. Cancer Sci 98:1536–1539
Asea A, Kraeft SK, Kurt-Jones EA et al (2000) HSP70 stimulates cytokine production through a CD14-dependant pathway, demonstrating its dual role as a chaperone and cytokine. Nat Med 6:435–442
Wang Y, Kelly CG, Singh M et al (2002) Stimulation of Th1-polarizing cytokines, C-C chemokines, maturation of dendritic cells, and adjuvant function by the peptide binding fragment of heat shock protein 70. J Immunol 169:2422–2429
Lehner T, Wang Y, Whittall T, McGowan E, Kelly CG, Singh M (2004) Functional domains of HSP70 stimulate generation of cytokines and chemokines, maturation of dendritic cells and adjuvanticity. Biochem Soc Trans 32:629–632
Kuppner MC, Gastpar R, Gelwer S et al (2001) The role of heat shock protein (hsp70) in dendritic cell maturation: hsp70 induces the maturation of immature dendritic cells but reduces DC differentiation from monocyte precursors. Eur J Immunol 31:1602–1609
Vega VL, Rodriguez-Silva M, Frey T et al (2008) Hsp70 translocates into the plasma membrane after stress and is released into the extracellular environment in a membrane-associated form that activates macrophages. J Immunol 180:4299–4307
Multhoff G (2007) Heat shock protein 70 (Hsp70): membrane location, export and immunological relevance. Methods 43:229–237
Sidera K, Patsavoudi E (2008) Extracellular HSP90: conquering the cell surface. Cell Cycle 7:1564–1568
Kazama H, Ricci JE, Herndon JM, Hoppe G, Green DR, Ferguson TA (2008) Induction of immunological tolerance by apoptotic cells requires caspase-dependent oxidation of high-mobility group box-1 protein. Immunity 29:21–32
Eleouet S, Carre J, Vonarx V, Heyman D, Lajat Y, Patrice T (1997) Delta-aminolevulinic acid-induced fluorescence in normal human lymphocytes. J Photochem Photobiol B 41:22–29
Hryhorenko EA, Oseroff AR, Morgan J, Rittenhouse-Diakun K (1998) Antigen specific and nonspecific modulation of the immune response by aminolevulinic acid based photodynamic therapy. Immunopharmacology 40:231–240
Hunt DW, Jiang H, Granville DJ, Chan AH, Leong S, Levy JG (1999) Consequences of the photodynamic treatment of resting and activated peripheral T lymphocytes. Immunopharmacology 41:31–44
Obochi MO, Canaan AJ, Jain AK, Richter AM, Levy JG (1995) Targeting activated lymphocytes with photodynamic therapy: susceptibility of mitogen-stimulated splenic lymphocytes to benzoporphyrin derivative (BPD) photosensitization. Photochem Photobiol 62:169–175
Hunt DW, Jiang HJ, Levy JG, Chan AH (1995) Sensitivity of activated murine peritoneal macrophages to photodynamic killing with benzoporphyrin derivative. Photochem Photobiol 61:417–421
Krutmann J, Athar M, Mendel DB et al (1989) Inhibition of the high affinity Fc receptor (Fc gamma RI) on human monocytes by porphyrin photosensitization is highly specific and mediated by the generation of superoxide radicals. J Biol Chem 264:11407–11413
Ostrand-Rosenberg S, Sinha P (2009) Myeloid-derived suppressor cells: linking inflammation and cancer. J Immunol 182:4499–4506
Gabrilovich DI, Nagaraj S (2009) Myeloid-derived suppressor cells as regulators of the immune system. Nat Rev Immunol 9:162–174
Wang HY, Wang RF (2007) Regulatory T cells and cancer. Curr Opin Immunol 19:217–223
Fingar VH, Taber SW, Haydon PS, Harrison LT, Kempf SJ, Wieman TJ (2000) Vascular damage after photodynamic therapy of solid tumors: a view and comparison of effect in pre-clinical and clinical models at the University of Louisville. In Vivo 14:93–100
Krammer B (2001) Vascular effects of photodynamic therapy. Anticancer Res 21:4271–4277
Cecic I, Serrano K, Gyongyossy-Issa M, Korbelik M (2005) Characteristics of complement activation in mice bearing Lewis lung carcinomas treated by photodynamic therapy. Cancer Lett 225:215–223
Henderson BW, Donovan JM (1989) Release of prostaglandin E2 from cells by photodynamic treatment in vitro. Cancer Res 49:6896–6900
Matroule JY, Bonizzi G, Morliere P et al (1999) Pyropheophorbide-a methyl ester-mediated photosensitization activates transcription factor NF-kappaB through the interleukin-1 receptor-dependent signaling pathway. J Biol Chem 274:2988–3000
Kick G, Messer G, Goetz A, Plewig G, Kind P (1995) Photodynamic therapy induces expression of interleukin 6 by activation of AP-1 but not NF-kappa B DNA binding. Cancer Res 55:2373–2379
Granville DJ, Carthy CM, Jiang H et al (2000) Nuclear factor-kappaB activation by the photochemotherapeutic agent verteporfin. Blood 95:256–262
Rubin B (2009) Natural immunity has significant impact on immune responses against cancer. Scand J Immunol 69:275–290
Musser DA, Oseroff AR (2001) Characteristics of the immunosuppression induced by cutaneous photodynamic therapy: persistence, antigen specificity and cell type involved. Photochem Photobiol 73:518–524
Musser DA, Camacho SH, Manderscheid PA, Oseroff AR (1999) The anatomic site of photodynamic therapy is a determinant for immunosuppression in a murine model. Photochem Photobiol 69:222–225
Simkin GO, King DE, Levy JG, Chan AH, Hunt DW (1997) Inhibition of contact hypersensitivity with different analogs of benzoporphyrin derivative. Immunopharmacology 37:221–230
Gollnick SO, Liu X, Owczarczak B, Musser DA, Henderson BW (1997) Altered expression of interleukin 6 and interleukin 10 as a result of photodynamic therapy in vivo. Cancer Res 57:3904–3909
Gollnick SO, Lee BY, Vaughan L, Owczarczak B, Henderson BW (2001) Activation of the IL-10 gene promoter following photodynamic therapy of murine keratinocytes. Photochem Photobiol 73:170–177
Pestka S, Krause CD, Sarkar D, Walter MR, Shi Y, Fisher PB (2004) Interleukin-10 and related cytokines and receptors. Annu Rev Immunol 22:929–979
Simkin GO, Tao JS, Levy JG, Hunt DW (2000) IL-10 contributes to the inhibition of contact hypersensitivity in mice treated with photodynamic therapy. J Immunol 164:2457–2462
Gollnick SO, Musser DA, Oseroff AR, Vaughan L, Owczarczak B, Henderson BW (2001) IL-10 does not play a role in cutaneous Photofrin photodynamic therapy-induced suppression of the contact hypersensitivity response. Photochem Photobiol 74:811–816
Yusuf N, Katiyar SK, Elmets CA (2008) The immunosuppressive effects of phthalocyanine photodynamic therapy in mice are mediated by CD4+ and CD8+ T cells and can be adoptively transferred to naive recipients. Photochem Photobiol 84:366–370
Lynch DH, Haddad S, King VJ, Ott MJ, Straight RC, Jolles CJ (1989) Systemic immunosuppression induced by photodynamic therapy (PDT) is adoptively transferred by macrophages. Photochem Photobiol 49:453–458
Zhang H, Ma W, Li Y (2009) Generation of effective vaccines against liver cancer by using photodynamic therapy. Lasers Med Sci 24:549–552
Gollnick SO, Brackett CM (2009) Enhancement of anti-tumor immunity by photodynamic therapy. Immunol Res. doi:10.1007/s12026-009-8119-4
Further reading
Maleckar JR, Sherman LA (1987) The composition of the T cell receptor repertoire in nude mice. J Immunol 138:3873–3876
de Winther MPJ, Heeringa P (2003) Bone marrow transplantations to study gene function in hematopoietic cells. In: Hofker MH, van Deursen J (eds) Methods in molecular biology, vol 209: transgenic mouse: methods and protocols. Humana Press, Totowa, p 281
Zhu C, Roth DB (1995) Characterization of coding ends in thymocytes of scid mice: implications for the mechanism of V(D)J recombination. Immunity 2:101–112
Potter M (1985) The Balb/C mouse—genetics and immunology—preface. Curr Top Microbiol Immunol 122:R5–R7
Levine S, Sowinski R (1973) Experimental allergic encephalomyelitis in inbred and outbred mice. J Immunol 110:139–143
Keetch DW, Humphrey P, Ratliff TL (1994) Development of a mouse model for nonbacterial prostatitis. J Urol 152:247–250
Vladutiu AO, Rose NR (1971) Autoimmune murine thyroiditis relation to histocompatibility (H-2) type. Science 174:1137–1139
Beck JA, Lloyd S, Hafezparast M et al (2000) Genealogies of mouse inbred strains. Nat Genet 24:23–25
Festing MF, Blackmore DK (1971) Life span of specified-pathogen-free (MRC category 4) mice and rats. Lab Anim 5:179–192
Russell ES, Neufeld EF, Higgins CT (1951) Comparison of normal blood picture of young adults from 18 inbred strains of mice. Proc Soc Exp Biol Med 78:761–766
Tokunaga Y, Hiramine C, Itoh M, Mukasa A, Hojo K (1993) Genetic susceptibility to the induction of murine experimental autoimmune orchitis (EAO) without adjuvant. I. Comparison of pathology, delayed type hypersensitivity, and antibody. Clin Immunol Immunopathol 66:239–247
Stinson SF (1990) Spontaneous tumors in Fischer rats. In: Stinson SF, Schuller HM, Reznik G (eds) Atlas of tumor pathology of the Fischer rat. CRC Press, Boca Raton, FL, pp 1–18
Zupko I, Kamuhabwa AR, D’Hallewin MA, Baert L, De Witte PA (2001) In vivo photodynamic activity of hypericin in transitional cell carcinoma bladder tumors. Int J Oncol 18:1099–1105
Hamilton JA (2008) Colony-stimulating factors in inflammation and autoimmunity. Nat Rev Immunol 8:533–544
Dranoff G (2004) Cytokines in cancer pathogenesis and cancer therapy. Nat Rev Cancer 4:11–22
McInnes IB, Schett G (2007) Cytokines in the pathogenesis of rheumatoid arthritis. Nat Rev Immunol 7:429–442
Gupta P, Su ZZ, Lebedeva IV et al (2006) mda-7/IL-24: multifunctional cancer-specific apoptosis-inducing cytokine. Pharmacol Ther 111:596–628
Cheuk AT, Mufti GJ, Guinn BA (2004) Role of 4-1BB:4-1BB ligand in cancer immunotherapy. Cancer Gene Ther 11:215–226
Wolf I, O’Kelly J, Rubinek T et al (2006) 15-Hydroxyprostaglandin dehydrogenase is a tumor suppressor of human breast cancer. Cancer Res 66:7818–7823
Balkwill F (1998) The molecular and cellular biology of the chemokines. J Viral Hepat 5:1–14
Feijoo E, Alfaro C, Mazzolini G et al (2005) Dendritic cells delivered inside human carcinomas are sequestered by interleukin-8. Int J Cancer 116:275–281
Li T, Zhong J, Chen Y et al (2006) Expression of chemokine-like factor 1 is upregulated during T lymphocyte activation. Life Sci 79:519–524
Doyen V, Rubio M, Braun D et al (2003) Thrombospondin 1 is an autocrine negative regulator of human dendritic cell activation. J Exp Med 198:1277–1283
Cvetanovic M, Ucker DS (2004) Innate immune discrimination of apoptotic cells: repression of proinflammatory macrophage transcription is coupled directly to specific recognition. J Immunol 172:880–889
Proskuryakov SY, Gabai VL, Konoplyannikov AG, Zamulaeva IA, Kolesnikova AI (2005) Immunology of apoptosis and necrosis. Biochemistry (Mosc) 70:1310–1320
Fadok VA, Bratton DL, Konowal A, Freed PW, Westcott JY, Henson PM (1998) Macrophages that have ingested apoptotic cells in vitro inhibit proinflammatory cytokine production through autocrine/paracrine mechanisms involving TGF-beta, PGE2, and PAF. J Clin Invest 101:890–898
Li MO, Sanjabi S, Flavell RA (2006) Transforming growth factor-beta controls development, homeostasis, and tolerance of T cells by regulatory T cell-dependent and -independent mechanisms. Immunity 25:455–471
Murakami M, Naraba H, Tanioka T et al (2000) Regulation of prostaglandin E2 biosynthesis by inducible membrane-associated prostaglandin E2 synthase that acts in concert with cyclooxygenase-2. J Biol Chem 275:32783–32792
Liu J, Miwa T, Hilliard B et al (2005) The complement inhibitory protein DAF (CD55) suppresses T cell immunity in vivo. J Exp Med 201:567–577
Krispin A, Bledi Y, Atallah M et al (2006) Apoptotic cell thrombospondin-1 and heparin-binding domain lead to dendritic-cell phagocytic and tolerizing states. Blood 108:3580–3589
Norata GD, Marchesi P, Pirillo A et al (2008) Long pentraxin 3, a key component of innate immunity, is modulated by high-density lipoproteins in endothelial cells. Arterioscler Thromb Vasc Biol 28:925–931
Rovere P, Peri G, Fazzini F et al (2000) The long pentraxin PTX3 binds to apoptotic cells and regulates their clearance by antigen-presenting dendritic cells. Blood 96:4300–4306
He X, Han B, Liu M (2007) Long pentraxin 3 in pulmonary infection and acute lung injury. Am J Physiol Lung Cell Mol Physiol 292:L1039–L1049
Zitvogel L, Tesniere A, Kroemer G (2006) Cancer despite immunosurveillance: immunoselection and immunosubversion. Nat Rev Immunol 6:715–727
Mellor AL, Munn DH (2008) Creating immune privilege: active local suppression that benefits friends, but protects foes. Nat Rev Immunol 8:74–80
Rabinovich GA, Gabrilovich D, Sotomayor EM (2007) Immunosuppressive strategies that are mediated by tumor cells. Annu Rev Immunol 25:267–296
Chen BJ, Cui X, Liu C, Chao NJ (2002) Prevention of graft-versus-host disease while preserving graft-versus-leukemia effect after selective depletion of host-reactive T cells by photodynamic cell purging process. Blood 99:3083–3088
Greinix HT, Volc-Platzer B, Kalhs P et al (2000) Extracorporeal photochemotherapy in the treatment of severe steroid-refractory acute graft-versus-host disease: a pilot study. Blood 96:2426–2431
McKenna KE, Whittaker S, Rhodes LE et al (2006) Evidence-based practice of photopheresis 1987–2001: a report of a workshop of the British Photodermatology Group and the U.K. Skin Lymphoma Group. Br J Dermatol 154:7–20
Greinix HT, Volc-Platzer B, Rabitsch W et al (1998) Successful use of extracorporeal photochemotherapy in the treatment of severe acute and chronic graft-versus-host disease. Blood 92:3098–3104
Kanold J, Messina C, Halle P et al (2005) Update on extracorporeal photochemotherapy for graft-versus-host disease treatment. Bone Marrow Transplant 35(Suppl 1):S69–S71
Calore E, Calo A, Tridello G et al (2008) Extracorporeal photochemotherapy may improve outcome in children with acute GVHD. Bone Marrow Transplant 42:421–425
Schleyer V, Radakovic-Fijan S, Karrer S et al (2006) Disappointing results and low tolerability of photodynamic therapy with topical 5-aminolaevulinic acid in psoriasis. A randomized, double-blind phase I/II study. J Eur Acad Dermatol Venereol 20:823–828
Radakovic-Fijan S, Blecha-Thalhammer U, Schleyer V et al (2005) Topical aminolaevulinic acid-based photodynamic therapy as a treatment option for psoriasis? Results of a randomized, observer-blinded study. Br J Dermatol 152:279–283
Smits T, Kleinpenning MM, van Erp PE, van de Kerkhof PC, Gerritsen MJ (2006) A placebo-controlled randomized study on the clinical effectiveness, immunohistochemical changes and protoporphyrin IX accumulation in fractionated 5-aminolaevulinic acid-photodynamic therapy in patients with psoriasis. Br J Dermatol 155:429–436
Fransson J, Ros AM (2005) Clinical and immunohistochemical evaluation of psoriatic plaques treated with topical 5-aminolaevulinic acid photodynamic therapy. Photodermatol Photoimmunol Photomed 21:326–332
Bissonnette R, Tremblay JF, Juzenas P, Boushira M, Lui H (2002) Systemic photodynamic therapy with aminolevulinic acid induces apoptosis in lesional T lymphocytes of psoriatic plaques. J Invest Dermatol 119:77–83
Robinson DJ, Collins P, Stringer MR et al (1999) Improved response of plaque psoriasis after multiple treatments with topical 5-aminolaevulinic acid photodynamic therapy. Acta Derm Venereol 79:451–455
Kim JY, Kang HY, Lee ES, Kim YC (2007) Topical 5-aminolaevulinic acid photodynamic therapy for intractable palmoplantar psoriasis. J Dermatol 34:37–40
Salah M, Samy N, Fadel M (2009) Methylene blue mediated photodynamic therapy for resistant plaque psoriasis. J Drugs Dermatol 8:42–49
Fernandez-Guarino M, Harto A, Sanchez-Ronco M, Garcia-Morales I, Jaen P (2009) Pulsed dye laser vs. photodynamic therapy in the treatment of refractory nail psoriasis: a comparative pilot study. J Eur Acad Dermatol Venereol 23:891–895
Ratkay LG, Waterfield JD, Hunt DW (2000) Photodynamic therapy in immune (non-oncological) disorders: focus on benzoporphyrin derivatives. BioDrugs 14:127–135
Torikai E, Kageyama Y, Kohno E et al (2008) Photodynamic therapy using talaporfin sodium for synovial membrane from rheumatoid arthritis patients and collagen-induced arthritis rats. Clin Rheumatol 27:751–761
Gabriel D, Busso N, So A, van den Bergh H, Gurny R, Lange N (2009) Thrombin-sensitive photodynamic agents: a novel strategy for selective synovectomy in rheumatoid arthritis. J Control Release 138:225–234
Hansch A, Frey O, Gajda M et al (2008) Photodynamic treatment as a novel approach in the therapy of arthritic joints. Lasers Surg Med 40:265–272
Trauner KB, Gandour-Edwards R, Bamberg M, Shortkroff S, Sledge C, Hasan T (1998) Photodynamic synovectomy using benzoporphyrin derivative in an antigen-induced arthritis model for rheumatoid arthritis. Photochem Photobiol 67:133–139
Ratkay LG, Chowdhary RK, Neyndorff HC, Tonzetich J, Waterfield JD, Levy JG (1994) Photodynamic therapy; a comparison with other immunomodulatory treatments of adjuvant-enhanced arthritis in MRL-lpr mice. Clin Exp Immunol 95:373–377
Szeimies RM, Landthaler M, Karrer S (2002) Non-oncologic indications for ALA-PDT. J Dermatolog Treat 13(Suppl 1):S13–S18
Taub AF (2007) Photodynamic therapy: other uses. Dermatol Clin 25:101–109
Babilas P, Landthaler M, Szeimies RM (2006) Photodynamic therapy in dermatology. Eur J Dermatol 16:340–348
Takahashi H, Komatsu S, Ibe M et al (2006) ATX-S10(Na)-PDT shows more potent effect on collagen metabolism of human normal and scleroderma dermal fibroblasts than ALA-PDT. Arch Dermatol Res 298:257–263
Bissonnette R, Shapiro J, Zeng H, McLean DI, Lui H (2000) Topical photodynamic therapy with 5-aminolaevulinic acid does not induce hair regrowth in patients with extensive alopecia areata. Br J Dermatol 143:1032–1035
Dall’Amico R, Livi U, Milano A et al (1995) Extracorporeal photochemotherapy as adjuvant treatment of heart transplant recipients with recurrent rejection. Transplantation 60:45–49
Rose EA, Barr ML, Xu H et al (1992) Photochemotherapy in human heart transplant recipients at high risk for fatal rejection. J Heart Lung Transplant 11:746–750
Meiser BM, Kur F, Reichenspurner H et al (1994) Reduction of the incidence of rejection by adjunct immunosuppression with photochemotherapy after heart transplantation. Transplantation 57:563–568
Barr ML, Baker CJ, Schenkel FA et al (2000) Prophylactic photopheresis and chronic rejection: effects on graft intimal hyperplasia in cardiac transplantation. Clin Transplant 14:162–166
Ludvigsson J, Samuelsson U, Ernerudh J, Johansson C, Stenhammar L, Berlin G (2001) Photopheresis at onset of type 1 diabetes: a randomised, double blind, placebo controlled trial. Arch Dis Child 85:149–154
Cavaletti G, Perseghin P, Dassi M et al (2006) Extracorporeal photochemotherapy: a safety and tolerability pilot study with preliminary efficacy results in refractory relapsing-remitting multiple sclerosis. Neurol Sci 27:24–32
Besnier DP, Chabannes D, Mussini JM, Dupas B, Esnault VL (2002) Extracorporeal photochemotherapy for secondary chronic progressive multiple sclerosis: a pilot study. Photodermatol Photoimmunol Photomed 18:36–41
Acknowledgements
P.A. and A.D.G. are supported by the grants from the K.U.Leuven (OT49/06) and F.W.O. Flanderen (G.0661.09). This paper presents research results of the IAP6/18, funded by the Interuniversity Attraction Poles Programme, initiated by the Belgian State, Science Policy Office. J.G. and D.N. are supported by grants N40112331/2736 and R0504303 from Ministry of Science and Higher Education in Poland; the European Union within European Regional Development Fund through Innovative Economy grant POIG.01.01.02-00-008/08 and by the Foundation for Polish Science Team Programme co-financed by the EU European Regional Development Fund. J.G. is a recipient of the Mistrz Award from the Foundation for Polish Science. Lastly, all figures were produced using Servier Medical Art (www.servier.com) for which the authors would like to acknowledge Servier.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Garg, A.D., Nowis, D., Golab, J. et al. Photodynamic therapy: illuminating the road from cell death towards anti-tumour immunity. Apoptosis 15, 1050–1071 (2010). https://doi.org/10.1007/s10495-010-0479-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10495-010-0479-7