Abstract
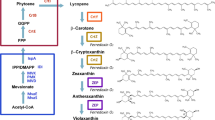
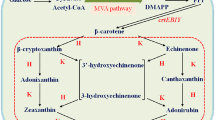
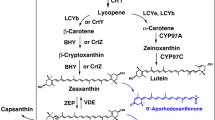
Zeaxanthin is a high-value carotenoid that is used in nutraceuticals, cosmetics, food, and animal feed industries. Zeaxanthin is chemically synthesized or purified from microorganisms as a natural product; however, increasing demand requires development of alternative sources such as heterologous biosynthesis by recombinant bacteria. For this purpose, we molecularly engineered Escherichia coli to optimize the synthesis of zeaxanthin from lycopene using fusion protein-mediated substrate channeling as well as by the introduction of tunable intergenic regions. The tunable intergenic regions approach was more efficient compared with protein fusion for coordinating expression of lycopene β-cyclase gene crtY and β-carotene 3-hydroxylase gene crtZ. The influence of the substrate channeling effect suggests that the reaction catalyzed by CrtZ is the rate-limiting step in zeaxanthin biosynthesis. Then Pantoea ananatis, Pantoea agglomerans and Haematococcus pluvialis crtZ were compared. Because P. ananatis crtZ is superior to that of P. agglomerans or H. pluvialis for zeaxanthin production, we used it to generate a recombinant strain of E. coli BETA-1 containing pZSPBA-2(P37-crtZPAN) that produced higher amounts of zeaxanthin (11.95 ± 0.21 mg/g dry cell weight) than other engineered E. coli strains described in the literature.




Similar content being viewed by others
References
Albermann Trachtmann N, Sprenger GA (2010) A simple and reliable method to conduct and monitor expression cassette integration into the Escherichia coli chromosome. Biotechnol J 5(1):32–38
Albrecht M, Misawa N, Sandmann G (1999) Metabolic engineering of the terpenoid biosynthetic pathway of Escherichia coli for production of the carotenoids β-carotene and zeaxanthin. Biotechnol Lett 21(9):791–795
Asker D, Beppu T, Ueda K (2007) Mesoflavibacter zeaxanthinifaciens gen. nov., sp. nov., a novel zeaxanthin-producing marine bacterium of the family Flavobacteriaceae. Syst Appl Microbiol 30(4):291–296
Asker D, Beppu T, Ueda K (2007) Zeaxanthinibacter enoshimensis gen. nov., sp. nov., a novel zeaxanthin-producing marine bacterium of the family Flavobacteriaceae, isolated from seawater off Enoshima Island, Japan. Int J Syst Evol Microbiol 57(4):837–843
BCC R (2011) The Global Market for Carotenoids. http://www.marketresearch.com/BCC-Research-v374/Global-Carotenoids-6558319
Berry A, Janssens D, Humbelin M, Jore JPM, Hoste B, Cleenwerck I, Vancanneyt M, Bretzek W, Mayer AF, Lopez-Ulibarri R, Shanmugam B, Swings J, Pasamontes L (2003) Paracoccus zeaxanthinifaciens sp nov., a zeaxanthin-producing bacterium. Int J Syst Evol Microbiol 53:231–238
Beuttler H, Hoffmann J, Jeske M, Hauer B, Schmid RD, Altenbuchner J, Urlacher VB (2011) Biosynthesis of zeaxanthin in recombinant Pseudomonas putida. Appl Microbiol Biotechnol 89(4):1137–1147
Bhosale P, Larson AJ, Bernstein PS (2004) Factorial analysis of tricarboxylic acid cycle intermediates for optimization of zeaxanthin production from Flavobacterium multivorum. J Appl Microbiol 96(3):623–629
Chen YY, Shen HJ, Cui YY, Chen SG, Weng ZM, Zhao M, Liu JZ (2013) Chromosomal evolution of Escherichia coli for the efficient production of lycopene. BMC Biotechnol 13:6
Choi SK, Matsuda S, Hoshino T, Peng X, Misawa N (2006) Characterization of bacterial beta-carotene 3,3′-hydroxylases, CrtZ, and P450 in astaxanthin biosynthetic pathway and adonirubin production by gene combination in Escherichia coli. Appl Microbiol Biotechnol 72(6):1238–1246
Cui YY, Ling C, Zhang YY, Huang J, Liu JZ (2014) Production of shikimic acid from E. coli through chemically inducible chromosomal evolution and cofactor metabolic engineering. Microb Cell Factories 13:21
Fraser PD, Miura Y, Misawa N (1997) In vitro characterization of astaxanthin biosynthetic enzymes. J Biol Chem 272(10):6128–6135
Gierhart DL (1995) Zeaxanthin-containing compositions produced by flavobacterium multivorum. U.S. patient 5427783
Guzman LM, Belin D, Carson MJ, Beckwith J (1995) Tight regulation, modulation, and high-level expression by vectors containing the arabinose PBAD promoter. J Bacteriol 177(14):4121–4130
Haldimann A, Wanner BL (2001) Conditional-replication, integration, excision, and retrieval plasmid-host systems for gene structure-function studies of bacteria. J Bacteriol 183(21):6384–6393
Huang JC, Zhong YJ, Liu J, Sandmann G, Chen F (2013) Metabolic engineering of tomato for high-yield production of astaxanthin. Metab Eng 17:59–67
Huang MT, Chen YY, Liu JZ (2014) Chromosomal engineering of Escherichia coli for the efficient production of coenzyme Q10. Chin J Chem Eng 22(5):559–569
Huang X, Holden HM, Raushel FM (2001) Channeling of substrates and intermediates in enzyme-catalyzed reactions. Ann Rev Biochem 70:149–180
Jin ES, Feth B, Melis A (2003) A mutant of the green alga Dunaliella salina constitutively accumulates zeaxanthin under all growth conditions. Biotechnol Bioeng 81(1):115–124
Kirchner O, Tauch A (2003) Tools for genetic engineering in the amino acid-producing bacterium Corynebacterium glutamicum. J Biotechnol 104(1–3):287–299
Lagarde D, Beuf L, Vermaas M (2000) Increased production of zeaxanthin and other pigments by application of genetic engineering techniques to Synechocystis sp strain PCC 6803. Appl Environ Microbiol 66(1):64–72
Lee TS, Krupa RA, Zhang FZ, Hajimorad M, Holtz WJ, Prasad N, Lee SK, Keasling JD (2011) BglBrick vectors and datasheets: a synthetic biology platform for gene expression. J Biol Eng 5:1–14
Lutz R, Bujard H (1997) Independent and tight regulation of transcriptional units in Escherichia coli via the LacR/O, the TetR/O and AraC/I1-I2 regulatory elements. Nucleic Acids Res 25(6):1203–1210
Misawa N, Nakagawa M, Kobayashi K, Yamano S, Nakamura K, Harashima K (1990) Elucidation of the Erwinia uredovora carotenoid biosynthetic pathway by functional analysis of gene products expressed in Escherichia coli. J Bacteriol 172(12):6704–6712
Moeller SM, Jacques PF, Blumberg JB (2000) The potential role of dietary xanthophylls in cataract and age-related macular degeneration. J Am Coll Nutr 5:522–527
Mohamed I, Mearns AS, Fraser K, Hodgson R (2013) Biological production of zeaxanthin and carotenoid biosynthesis control. U.S. patient 8361743
Nelis JH, De Leenheer PA (1991) Microbial sources of carotenoid pigments used in foods and feeds. J Appl Bacteriol 70:181–191
Nishino H, Murakoshi M, Tokuda H, Satomi Y (2009) Cancer prevention by carotenoids. Arch Biochem Biophys 483(2):165–168
Nishizaki T, Tsuge K, Itaya M, Doi N, Yanagawa H (2007) Metabolic engineering of carotenoid biosynthesis in Escherichia coli by ordered gene assembly in Bacillus subtilis. Appl Environ Microbiol 73(4):1355–1361
Pfleger BF, Pitera DJ, Smolke CD, Keasling JD (2006) Combinatorial engineering of intergenic regions in operons tunes expression of multiple genes. Nat Biotechnol 24(8):1027–1032
Prabhu S, Rekha PD, Young CC, Hameed A, Lin SY, Arun AB (2013) Zeaxanthin production by novel marine isolates from coastal sand of India and its antioxidant properties. Appl Biochem Biotechnol 171(4):817–831
Puigbò P, Guzmán E, Romeu A, Garcia-Vallvé S (2007) OPTIMIZER: a web server for optimizing the codon usage of DNA sequences. Nucleic Acids Res 35:W126–W131
Ruther A, Misawa N, Boger P, Sandmann G (1997) Production of zeaxanthin in Escherichia coli transformed with different carotenogenic plasmids. Appl Microbiol Biotechnol 48(2):162–167
Sajilata MG, Singhal RS, Kamat MY (2008) The carotenoid pigment zeaxanthin—a review. Compr Rev Food Sci F 7(1):29–49
Seo SW, Yang JS, Kim I, Yang J, Min BE, Kim S, Jung GY (2013) Predictive design of mRNA translation initiation region to control prokaryotic translation efficiency. Metab Eng 15:67–74
Shepherd D and Daseek J (1976) Production of zeaxanthin. U.S. patent 3951742
Singh D, Puri M, Wilkens S, Mathur AS, Tuli DK, Barrow CJ (2013) Characterization of a new zeaxanthin producing strain of Chlorella saccharophila isolated from New Zealand marine waters. Bioresour Technol 143:308–314
Snodderly DM (1995) Evidence for protection against age-related macular degeneration by carotenoids and antioxidant vitamins. Am J Clin Nutr 62(6 Suppl):1448S–1461S
Thawornwiriyanun P, Tanasupawat S, Dechsakulwatana C, Techkarnjanaruk S, Suntornsuk W (2012) Identification of newly zeaxanthin-producing bacteria isolated from sponges in the gulf of Thailand and their zeaxanthin production. Appl Biochem Biotechnol 167(8):2357–2368
Wang C, Yoon SH, Jang HJ, Chung YR, Kim JY, Choi ES, Kim SW (2011) Metabolic engineering of Escherichia coli for α-farnesene production. Metab Eng 13(6):648–655
Xu P, Vansiri A, Bhan N, Koffas MAG (2012) ePathBrick: a synthetic biology platform for engineering metabolic pathways in E. coli. ACS Synth Biol 1(7):256–266
Zhang YH (2011) Substrate channeling and enzyme complexes for biotechnological applications. Biotechnol Adv 29(6):715–725
Acknowledgments
We are grateful to the National Natural Science Foundation of China (Grant No. 30970089, 21276289), the Natural Science Foundation of Guangdong Province (No. S2011010001396), and the Key Project of the Scientific and Technical Innovation of Higher Education of Guangdong Province (2012CXZD0002) for financial support.
Conflict of interest
The authors declare that they have no conflict of interests.
Author information
Authors and Affiliations
Corresponding author
Additional information
Special Issue: Metabolic Engineering.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Li, XR., Tian, GQ., Shen, HJ. et al. Metabolic engineering of Escherichia coli to produce zeaxanthin. J Ind Microbiol Biotechnol 42, 627–636 (2015). https://doi.org/10.1007/s10295-014-1565-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10295-014-1565-6