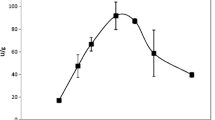
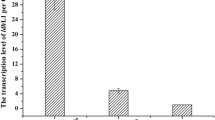
Abstract
Bacillus coagulans, a sporogenic lactic acid bacterium, grows optimally at 50–55°C and produces lactic acid as the primary fermentation product from both hexoses and pentoses. The amount of fungal cellulases required for simultaneous saccharification and fermentation (SSF) at 55°C was previously reported to be three to four times lower than for SSF at the optimum growth temperature for Saccharomyces cerevisiae of 35°C. An ethanologenic B. coagulans is expected to lower the cellulase loading and production cost of cellulosic ethanol due to SSF at 55°C. As a first step towards developing B. coagulans as an ethanologenic microbial biocatalyst, activity of the primary fermentation enzyme L-lactate dehydrogenase was removed by mutation (strain Suy27). Strain Suy27 produced ethanol as the main fermentation product from glucose during growth at pH 7.0 (0.33 g ethanol per g glucose fermented). Pyruvate dehydrogenase (PDH) and alcohol dehydrogenase (ADH) acting in series contributed to about 55% of the ethanol produced by this mutant while pyruvate formate lyase and ADH were responsible for the remainder. Due to the absence of PDH activity in B. coagulans during fermentative growth at pH 5.0, the l-ldh mutant failed to grow anaerobically at pH 5.0. Strain Suy27-13, a derivative of the l-ldh mutant strain Suy27, that produced PDH activity during anaerobic growth at pH 5.0 grew at this pH and also produced ethanol as the fermentation product (0.39 g per g glucose). These results show that construction of an ethanologenic B. coagulans requires optimal expression of PDH activity in addition to the removal of the LDH activity to support growth and ethanol production.




Similar content being viewed by others
References
Bothast RJ, Schlicher MA (2005) Biotechnological processes for conversion of corn into ethanol. Appl Microbiol Biotechnol 67:19–25
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Cripps RE, Eley K, Leak DJ, Rudd B, Taylor M, Todd M, Boakes S, Martin S, Atkinson T (2009) Metabolic engineering of Geobacillus thermoglucosidasius for high yield ethanol production. Metab Eng 11:398–408
De Clerck E, Rodriguez-Diaz M, Forsyth G, Lebbe L, Logan NA, DeVos P (2004) Polyphasic characterization of Bacillus coagulans strains, illustrating heterogeneity within this species, and emended description of the species. System Appl Microbiol 27:50–60
Demain AL (2009) Biosolutions to the energy problem. J Ind Microbiol Biotechnol 36:319–332
Demain AL, Newcomb M, Wu JH (2005) Cellulase, clostridia, and ethanol. Microbiol Mol Biol Rev 69:124–154
Department of Energy (2009) Biomass multi-year program plan. Office of the biomass program, energy efficiency and renewable energy. http://www1.eere.energy.gov/biomass/pdfs/mypp_december2009.pdf
Desai SG, Guerinot ML, Lynd LR (2004) Cloning of L-lactate dehydrogenase and elimination of lactic acid production via gene knockout in Thermoanaerobacterium saccharolyticum JW/SL-YS485. Appl Microbiol Biotechnol 65:600–605
Drago L, De Vecchi E (2009) Should Lactobacillus sporogenes and Bacillus coagulans have a future? J Chemother 21:371–377
Galbe M, Sassner P, Wingren A, Zacchi G (2007) Process engineering economics of bioethanol production. Adv Biochem Eng Biotechnol 108:303–327
Gao H, Jiang X, Pogliano K, Aronson AI (2002) The E1beta and E2 subunits of the Bacillus subtilis pyruvate dehydrogenase complex are involved in regulation of sporulation. J Bacteriol 184:2780–2788
Gauss WF, Suzuki S, Takagi M 1976. Manufacture of alcohol from cellulosic materials using plural ferments. United States patent 3,990,944
Hartley BS, Payton MA (1983) Industrial prospects for thermophiles and thermophilic enzymes. Biochem Soc Symp 48:133–146
Ingram LO, Aldrich HC, Borges AC, Causey TB, Martinez A, Morales F, Saleh A, Underwood SA, Yomano LP, York SW, Zaldivar J, Zhou S (1999) Enteric bacterial catalysts for fuel ethanol production. Biotechnol Prog 15:855–866
Iuchi S, Lin EC (1993) Adaptation of Escherichia coli to redox environments by gene expression. Mol Microbiol 9:9–15
Jarboe LR, Grabar TB, Yomanao LP, Shanmugam KT, Ingram LO (2007) Development of ethanologenic bacteria. Adv Biochem Eng Biotechnol 108:237–261
Kim Y, Ingram LO, Shanmugam KT (2007) Construction of an Escherichia coli K-12 mutant for homoethanologenic fermentation of glucose or xylose without foreign genes. Appl Environ Microbiol 73:1766–1771
Kim Y, Ingram LO, Shanmugam KT (2008) Dihydrolipoamide dehydrogenase mutation alters the NADH sensitivity of pyruvate dehydrogenase complex of Escherichia coli K-12. J Bacteriol 190:3851–3858
Leber J (2010) Economics improve for first commercial cellulosic ethanol plants. New York Times: Feb. 16, 2010
Lee JH, Patel P, Sankar P, Shanmugam KT (1985) Isolation and characterization of mutant strains of Escherichia coli altered in H2 metabolism. J Bacteriol 162:344–352
Lynd LR, Laser MS, Bransby D, Dale BE, Davison B, Hamilton R, Himmel M, Keller M, McMillan JD, Sheehan J, Wyman CE (2008) How biotech can transform biofuels. Nat Biotechnol 26:169–172
Lynd LR, Weimer PJ, van Zyl WH, Pretorius IS (2002) Microbial cellulose utilization: fundamentals and biotechnology. Microbiol Mol Biol Rev 66:506–577
Ou MS, Mohammed N, Ingram LO, Shanmugam KT (2009) Thermophilic Bacillus coagulans requires less cellulases for simultaneous saccharification and fermentation of cellulose to products than mesophilic microbial biocatalysts. Appl Biochem Biotechnol 155:379–385
Patel MA, Ou M, Ingram LO, Shanmugam KT (2005) Simultaneous saccharification and co-fermentation of crystalline cellulose and sugar cane bagasse hemicellulose hydrolysate to lactate by a thermotolerant acidophilic Bacillus sp. Biotech Prog 21:1453–1460
Patel MA, Ou MS, Harbrucker R, Aldrich HC, Buszko ML, Ingram LO, Shanmugam KT (2006) Isolation and characterization of acid-tolerant, thermophilic bacteria for effective fermentation of biomass-derived sugars to lactic acid. Appl Environ Microbiol 72:3228–3235
Payot T, Chemaly Z, Fick M (1999) Lactic acid production by Bacillus coagulans—Kinetic studies and optimization of culture medium for batch and continuous fermentations. Enz Microb Technol 24:191–199
Reents H, Munch R, Dammeyer T, Jahn D, Hartig E (2006) The Fnr regulon of Bacillus subtilis. J Bacteriol 188:1103–1112
Romero S, Merino E, Bolivar F, Gosset G, Martinez A (2007) Metabolic engineering of Bacillus subtilis for ethanol production: lactate dehydrogenase plays a key role in fermentative metabolism. Appl Environ Microbiol 73:5190–5198
Rosenberg M, Rebros M, Kristofikova L, Malatova K (2005) High temperature lactic acid production by Bacillus coagulans immobilized in LentiKats. Biotechnol Lett 27:1943–1947
Shanmugam KT, Valentine RC (1980) Nitrogen fixation (nif) mutants of Klebsiella pneumoniae. Meth Enzymol 69:47–52
Shaw AJ, Podkaminer KK, Desai SG, Bardsley JS, Rogers SR, Thorne PG, Hogsett DA, Lynd LR (2008) Metabolic engineering of a thermophilic bacterium to produce ethanol at high yield. Proc Natl Acad Sci USA 105:13769–13774
Tao H, Gonzalez R, Martinez A, Rodriguez M, Ingram LO, Preston JF, Shanmugam KT (2001) Engineering a homo-ethanol pathway in Escherichia coli: increased glycolytic flux and levels of expression of glycolytic genes during xylose fermentation. J Bacteriol 183:2979–2988
Tenenbaum DJ (2008) Food vs. fuel: diversion of crops could cause more hunger. Environ Health Perspect 116:A254–A257
Underwood SA, Zhou S, Causey TB, Yomano LP, Shanmugam KT, Ingram LO (2002) Genetic changes to optimize carbon partitioning between ethanol and biosynthesis in ethanologenic Escherichia coli. Appl Environ Microbiol 68:6263–6272
Visser J, Kester H, Jeyaseelan K, Topp R (1982) Pyruvate dehydrogenase complex from Bacillus. Methods Enzymol 89(Pt D):399–407
Wang Q, Ou MS, Kim Y, Ingram LO, Shanmugam KT (2010) Metabolic flux control at the pyruvate node in an anaerobic Escherichia coli strain with an active pyruvate dehydrogenase. Appl Environ Microbiol 76:2107–2114
Wyman CE (2007) What is (and is not) vital to advancing cellulosic ethanol. Trends Biotechnol 25:153–157
Yoshida A, Freese E (1975) Lactate dehydrogenase from Bacillus subtilis. Meth Enzymol 41:304–309
Zhou S, Iverson AG, Grayburn WS (2008) Engineering a native homoethanol pathway in Escherichia coli B for ethanol production. Biotechnol Lett 30:335–342
Acknowledgments
We thank Dr. Julie A. Maupin-Furlow for the use of BioFlo fermenter. This study was supported in part by a grant from the Department of Energy (DE-FG36-04GO14019), the State of Florida, University of Florida Agricultural Experiment Station, and the Florida Energy Systems Consortium.
Author information
Authors and Affiliations
Corresponding author
Additional information
Y. Su and M. S. Rhee contributed equally to this work.
Rights and permissions
About this article
Cite this article
Su, Y., Rhee, M.S., Ingram, L.O. et al. Physiological and fermentation properties of Bacillus coagulans and a mutant lacking fermentative lactate dehydrogenase activity. J Ind Microbiol Biotechnol 38, 441–450 (2011). https://doi.org/10.1007/s10295-010-0788-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10295-010-0788-4