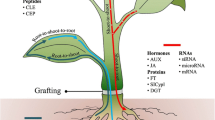
Abstract
Long-distance signaling via phloem tissues is an important mechanism for inter-organ communication. Such communication allows plants to integrate environmental information into physiological and developmental responses. Grafting has provided persuasive evidence of long-distance signaling involved in various processes, including flowering, tuberization, nodulation, shoot branching, post-transcriptional gene silencing, and disease resistance. A micro-grafting technique to generate two-shoot grafts is available for young seedlings of Arabidopsis thaliana and was adapted for use in the study of flowering. Histological analysis using transgenic plants expressing β-glucuronidase (GUS) in phloem tissues showed that phloem continuity between a stock and a scion was established between 7 and 10 days after grafting. Experiments using tracer dyes and enhanced green fluorescent protein (EGFP) showed that the phloem connection was functional and capable of effecting macromolecular transmission. Successful grafts can be obtained at high frequency (10–30%) and selected after 2–3 weeks of post-surgery growth. This method was applied successfully to the study of flowering, one of the important events regulated by long-distance signaling. This grafting technique will facilitate the study of the long-distance action of genes involved in various aspects of growth and development, and in transport of signal molecules.






Similar content being viewed by others
References
Abe M, Kobayashi Y, Yamamoto S, Daimon Y, Yamaguchi A, Ikeda Y, Ichinoki H, Notaguchi M, Goto K, Araki T (2005) FD, a bZIP protein mediating signals from the floral pathway integrator FT at the shoot apex. Science 309:1052–1056
An H, Roussot C, Suárez-López P, Corbesier L, Vincent C, Piñeiro M, Hepworth S, Mouradov A, Justin S, Turnbull C, Coupland G (2004) CONSTANS acts in the phloem to regulate a systemic signal that induces photoperiodic flowering of Arabidopsis. Development 131:3615–3626
Araki T (2001) Transition from vegetative to reproductive phase. Curr Opin Plant Biol 4:63–68
Asahina M, Iwai H, Kikuchi A, Yamaguchi S, Kamiya Y, Kamada H, Satoh S (2002) Gibberellin produced in the cotyledon is required for cell division during tissue reunion in the cortex of cut cucumber and tomato hypocotyls. Plant Physiol 129:201–210
Ayre BG, Turgeon R (2004) Graft transmission of a floral stimulant derived from CONSTANS. Plant Physiol 13:2271–2278
Bainbridge K, Bennett T, Turnbull C, Leyser O (2006) Grafting. Methods Mol Biol 323:39–44
Beveridge C (2006) Axillary bud outgrowth: sending a message. Curr Opin Plant Biol 9:35–40
Booker J, Chatfield S, Leyser O (2003) Auxin acts in xylem-associated or medullary cells to mediate apical dominance. Plant Cell 15:495–507
Brosnan C, Mitter N, Christie M, Smith N, Waterhouse P, Carroll B (2007) Nuclear gene silencing directs reception of long-distance mRNA silencing in Arabidopsis. Proc Natl Acad Sci USA 104:14741–14746
Chen A, Komives E, Schroeder J (2006) An improved grafting technique for mature Arabidopsis plants demonstrates long-distance shoot-to-root transport of phytochelatins in Arabidopsis. Plant Physiol 141:108–120
Clough S, Bent A (1998) Floral dip: a simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J 16:735–743
Corbesier L, Vincent C, Jang S, Fornara F, Fan Q, Searle I, Giakountis A, Farrona S, Gissot L, Turnbull C, Coupland G (2007) FT protein movement contributes to long-distance signaling in floral induction of Arabidopsis. Science 316:1030–1033
Flaishman M, Loginovsky K, Golobowich S, Lev-Yadun S (2008) Arabidopsis thaliana as a model system for graft union development in homografts and heterografts. J Plant Growth Regul 27:231–239
Fusaro A, Matthew L, Smith N, Curtin S, Dedic-Hagan J, Ellacott G, Watson J, Wang M, Brosnan C, Carroll B, Waterhouse P (2006) RNA interference-inducing hairpin RNAs in plants act through the viral defence pathway. EMBO Rep 7:1168–1175
Gomez-Roldan V, Fermas S, Brewer P, Puech-Pages V, Dun E, Pillot J, Letisse F, Matusova R, Danoun S, Portais J, Bouwmeester H, Becard G, Beveridge C, Rameau C, Rochange S (2008) Strigolactone inhibition of shoot branching. Nature 455:189–194
Hartmann HT, Kester DE, Davies FT Jr (1990) Plant propagation: principles and practices, 5th edn. Prentice-Hall, Englewood Cliffs, NJ, pp 305–348
Haywood V, Yu T, Huang N, Lucas W (2005) Phloem long-distance trafficking of GIBBERELLIC ACID-INSENSITIVE RNA regulates leaf development. Plant J 42:49–68
Hempel FD, Weigel D, Alejandra Mandel M, Ditta G, Zambryski PC, Feldman LJ, Yanofsky MF (1997) Floral determination and expression of floral regulatory genes in Arabidopsis. Development 124:3845–3853
Imlau A, Truernit E, Sauer N (1999) Cell-to-cell and long-distance trafficking of the green fluorescent protein in the phloem and symplastic unloading of the protein into sink tissues. Plant Cell 11:309–322
Jackson S (1999) Multiple signaling pathways control tuber induction in potato. Plant Physiol 119:1–8
Jaeger KE, Wigge PA (2007) FT protein acts as a long-range signal in Arabidopsis. Curr Biol 17:1050–1054
Kim M, Canio W, Kessler S, Sinha N (2001) Developmental changes due to long-distance movement of a homeobox fusion transcript in tomato. Science 293:287–289
Kobayashi Y, Kaya H, Goto K, Iwabuchi M, Araki T (1999) A pair of related genes with antagonistic roles in mediating flowering signals. Science 286:1960–1962
Lifschitz E, Eviatar T, Rozman A, Shalit A, Goldshmidt A, Amsellem Z, Alvarez J, Eshed Y (2006) The tomato FT ortholog triggers systemic signals that regulate growth and flowering and substitute for diverse environmental stimuli. Proc Natl Acad Sci USA 103:6398–6403
Lin M, Belanger H, Lee Y, Varkonyi-Gasic E, Taoka K, Miura E, Xoconostle-Cázares B, Gendler K, Jorgensen RA, Phinney B, Lough TJ, Lucas WJ (2007) FLOWERING LOCUS T protein may act as the long-distance florigenic signal in the Cucurbits. Plant Cell 19:1488–1506
Lin S, Chiang S, Lin W, Chen J, Tseng C, Wu P, Chiou T (2008) Regulatory network of microRNA399 and PHO2 by systemic signaling. Plant Physiol 147:732–746
Lough T, Lucas W (2006) Integrative plant biology: role of phloem long-distance macromolecular trafficking. Annu Rev Plant Biol 57:203–232
Mathieu J, Warthmann N, Küttner F, Schmid M (2007) Export of FT protein from phloem companion cells is sufficient for floral induction in Arabidopsis. Curr Biol 17:1055–1060
Michaels S, Himelblau E, Kim S, Schomburg F, Amasino R (2005) Integration of flowering signals in winter-annual Arabidopsis. Plant Physiol 137:149–156
Notaguchi M, Abe M, Kimura T, Daimon Y, Kobayashi T, Yamaguchi A, Tomita Y, Dohi K, Mori M, Araki T (2008) Long-distance, graft-transmissible action of Arabidopsis FLOWERING LOCUS T protein to promote flowering. Plant Cell Physiol 49:1645–1658
Oka-Kira E, Kawaguchi M (2006) Long-distance signaling to control root nodule number. Curr Opin Plant Biol 9:496–502
Oparka KJ, Duckett CM, Prior DAM, Fisher DB (1994) Real-time imaging of phloem unloading in the root tip of Arabidopsis. Plant J 6:759–766
Palauqui J, Elmayan T, Pollien J, Vaucheret H (1997) Systemic acquired silencing: transgene-specific post-transcriptional silencing is transmitted by grafting from silenced stocks to non-silenced scions. EMBO J 16:4738–4745
Pant B, Buhtz A, Kehr J, Scheible W (2008) MicroRNA399 is a long-distance signal for the regulation of plant phosphate homeostasis. Plant J 53:731–738
Rhee SY, Somerville CR (1995) Flat-surface grafting in Arabidopsis thaliana. Plant Mol Biol Rep 13:118–123
Rus A, Baxter I, Muthukumar B, Gustin J, Lahner B, Yakubova E, Salt DE (2006) Natural variants of AtHKT1 enhance Na+ accumulation in two wild populations of Arabidopsis. PLoS Genet 2:1964–1973
Simpson G, Dean C (2002) Arabidopsis, the Rosetta stone of flowering time? Science 296:285–289
Stoddard FL, McCully ME (1980) Effects of excision of stock and scion on the formation of the graft union in Coleus: a histological study. Bot Gaz 141:401–412
Suárez-López P (2005) Long-range signalling in plant reproductive development. Int J Dev Biol 49:761–771
Sugaya S, Hayakawa K, Handa T, Ucimiya H (1989) Cell-specific expression of the rolC gene of the TL-DNA of Ri plasmid in transgenic tobacco plants. Plant Cell Physiol 30:649–653
Tamaki S, Matsuo S, Wong H, Yokoi S, Shimamoto K (2007) Hd3a protein is a mobile flowering signal in rice. Science 316:1033–1036
Tsukaya N, Naito S, Rédei G, Komeda Y (1993) A new class of mutations in Arabidopsis thaliana, acaulis1, affecting the development of both inflorescences and leaves. Development 118:751–764
Turnbull C, Booker J, Leyser H (2002) Micrografting techniques for testing long-distance signalling in Arabidopsis. Plant J 32:255–262
Umehara M, Hanada A, Yoshida S, Akiyama K, Arite T, Takeda-Kamiya N, Magome H, Kamiya Y, Shirasu K, Yoneyama K, Kyozuka J, Yamaguchi S (2008) Inhibition of shoot branching by new terpenoid plant hormones. Nature 455:195–200
Weller JL, Reid JB, Taylor SA, Murfet IC (1997) The genetic control of flowering in pea. Trends Plant Sci 2:412–418
Wigge P, Kim M, Jaeger K, Busch W, Schmid M, Lohmann J, Weigel D (2005) Integration of spatial and temporal information during floral induction in Arabidopsis. Science 309:1056–1059
Wright KM, Oparka KJ (1996) The fluorescent probe HPTS as a phloem-mobile, symplastic tracer: an evaluation using confocal laser scanning microscopy. J Exp Bot 47:439–445
Xia Y, Suzuki H, Borevitz J, Blount J, Guo Z, Patel K, Dixon R, Lamb C (2004) An extracellular aspartic protease functions in Arabidopsis disease resistance signaling. EMBO J 23:980–988
Yamaguchi A, Kobayashi Y, Goto K, Abe M, Araki T (2005) TWIN SISTER OF FT (TSF) acts as a floral pathway integrator redundantly with FT. Plant Cell Physiol 46:1175–1189
Zeevaart JAD (1976) Physiology of flower formation. Annu Rev Plant Physiol 27:321–348
Acknowledgments
We thank Mr. M. Kobayashi for plant material, Drs. M. Kawai and H. Uchimiya for a plasmid, and Dr. S. Hata for instruments. This work was supported by grants from the Ministry of Education, Culture, Sport, Science and Technology of Japan (to T.A. and M.A.), the CREST program of the Japan Science and Technology Agency (to T.A.), and the PROBRAIN program of the Bio-oriented Technology Research Advancement Institution, Japan (to M.A. and T.A.), and the Mitsubishi Foundation (to T.A.).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Notaguchi, M., Daimon, Y., Abe, M. et al. Adaptation of a seedling micro-grafting technique to the study of long-distance signaling in flowering of Arabidopsis thaliana . J Plant Res 122, 201–214 (2009). https://doi.org/10.1007/s10265-008-0209-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10265-008-0209-1