Abstract
Background
Despite the fact that rituximab (RTX)-associated adverse events may be relatively frequent in younger patients, recent studies have reported RTX as a suitable first-line steroid-sparing agent for maintaining remission in children with steroid-dependent nephrotic syndrome (SDNS). However, the impact of age at RTX initiation on the long-term outcome remains unknown in this cohort.
Methods
We retrospectively reviewed the clinical course of 61 patients with complicated SDNS who received a single dose of RTX (375 mg/m2) followed by maintenance immunosuppressive agents (IS) from January 2008 to March 2021. In patients who achieved > 12 months of prednisolone-free remission, IS tapering within 6 months was tried to achieve. The primary endpoint was the probability of achieving long-term treatment-free remission at the last follow-up.
Results
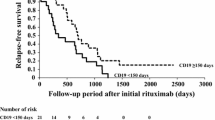
After RTX initiation, 52 patients (85.2%) relapsed after a median of 665 days, and 44 patients (72.1%) received additional RTX doses (total, 226 infusions). At the last follow-up (median observation period, 8.3 years; median age, 18.3 years), 16 patients (26.2%) achieved long-term remission. Multivariate analysis showed that older age at RTX initiation was the independent predictive factor for achieving long-term remission (odds ratio, 1.25; p < 0.05). The proportion of those who achieved long-term remission was significantly higher in patients aged ≥ 13.5 years than in those aged < 13.5 years at RTX initiation (52.6 vs 14.3%, p < 0.05). Persistent severe hypogammaglobulinemia did not develop in older children (≥ 13.5 years) at RTX initiation.
Conclusion
For older children with complicated SDNS, RTX appeared to be a suitable disease-modifying therapy without persistent adverse events.



Similar content being viewed by others
Data availability
The datasets of the current study are available from the corresponding author on reasonable request.
References
Benz K, Dötsch J, Rascher W, Stachel D. Change of the course of steroid-dependent nephrotic syndrome after rituximab therapy. Pediatr Nephrol. 2004;19:794–7.
Guigonis V, Dallocchio A, Baudouin V, Dehennault M, Hachon-Le Camus C, Afanetti M, Groothoff J, Llanas B, Niaudet P, Nivet H, Raynaud N, Taque S, Ronco P, Bouissou F. Rituximab treatment for severe steroid- or cyclosporine-dependent nephrotic syndrome: a multicentric series of 22 cases. Pediatr Nephrol. 2008;23:1269–79.
Kamei K, Ito S, Nozu K, Fujinaga S, Nakayama M, Sako M, Saito M, Yoneko M, Iijima K. Single dose of rituximab for refractory steroid-dependent nephrotic syndrome in children. Pediatr Nephrol. 2009;24:1321–2138.
Fujinaga S, Hirano D, Nishizaki N, Kamei K, Ito S, Ohtomo Y, Shimizu T, Kaneko K. Single infusion of rituximab for persistent steroid-dependent minimal-change nephrotic syndrome after long-term cyclosporine. Pediatr Nephrol. 2010;25:539–44.
Ito S, Kamei K, Ogura M, Sato M, Fujimaru T, Ishikawa T, Udagawa T, Iijima K. Maintenance therapy with mycophenolate mofetil after rituximab in pediatric patients with steroid-dependent nephrotic syndrome. Pediatr Nephrol. 2011;26:1823–918.
Fujinaga S, Someya T, Watanabe T, Ito A, Ohtomo Y, Shimizu T, Kaneko K. Cyclosporine versus mycophenolate mofetil for maintenance of remission of steroid-dependent nephrotic syndrome after a single infusion of rituximab. Eur J Pediatr. 2013;172:513–8.
Iijima K, Sako M, Nozu K, Mori R, Tuchida N, Kamei K, Miura K, Aya K, Nakanishi K, Ohtomo Y, Takahashi S, Tanaka R, Kaito H, Nakamura H, Ishikura K, Ito S, Ohashi Y, Rituximab for Childhood-onset Refractory Nephrotic Syndrome (RCRNS) Study Group. Rituximab for childhood-onset, complicated, frequently relapsing nephrotic syndrome or steroid-dependent nephrotic syndrome: a multicentre, double-blind, randomised, placebo-controlled trial. Lancet. 2014;384:1273–81.
Ravani P, Ponticelli A, Siciliano C, Fornoni A, Magnasco A, Sica F, Bodria M, Caridi G, Wei C, Belingheri M, Ghio L, Merscher-Gomez S, Edefonti A, Pasini A, Montini G, Murtas C, Wang X, Muruve D, Vaglio A, Martorana D, Pani A, Scolari F, Reiser J, Ghiggeri GM. Rituximab is a safe and effective long-term treatment for children with steroid and calcineurin inhibitor-dependent idiopathic nephrotic syndrome. Kidney Int. 2013;84:1025–33.
Fujinaga S, Hirano D. Risk factors for early relapse during maintenance therapy after a single infusion of rituximab in children with steroid-dependent nephrotic syndrome. Pediatr Nephrol. 2013;29:491–2.
Sinha A, Bhatia D, Gulati A, Rawat M, Dinda AK, Hari P, Bagga A. Efficacy and safety of rituximab in children with difficult-to-treat nephrotic syndrome. Nephrol Dial Transplant. 2015;30:96–106.
Kamei K, Ogura M, Sato M, Sako M, Iijima K, Ito S. Risk factors for relapse and long-term outcome in steroid-dependent nephrotic syndrome treated with rituximab. Pediatr Nephrol. 2016;31:89–95.
Chan EY, Webb H, Yu E, Ghiggeri GM, Kemper MJ, Ma AL, Yamamura T, Sinha A, Bagga A, Hogan J, Dossier C, Vivarelli M, Liu ID, Kamei K, Ishikura K, Saini P, Tullus K. Both the rituximab dose and maintenance immunosuppression in steroid-dependent/frequently-relapsing nephrotic syndrome have important effects on outcomes. Kidney Int. 2020;97:393–401.
Chan EY, Yu ELM, Angeletti A, Arslan Z, Basu B, Boyer O, Chan CY, Colucci M, Dorval G, Dossier C, Drovandi S, Ghiggeri GM, Gipson DS, Hamada R, Hogan J, Ishikura K, Kamei K, Kemper MJ, Ma AL, Parekh RS, Radhakrishnan S, Saini P, Shen Q, Sinha R, Subun C, Teo S, Vivarelli M, Webb H, Xu H, Yap HK, Tullus K. Long-term efficacy and safety of repeated rituximab to maintain remission in idiopathic childhood nephrotic syndrome: an international study. J Am Soc Nephrol. 2020;33:1193–207.
Ravani P, Rossi R, Bonanni A, Quinn RR, Sica F, Bodria M, Pasini A, Montini G, Edefonti A, Belingheri M, De Giovanni D, Barbano G, Degl’Innocenti L, Scolari F, Murer L, Reiser J, Fornoni A, Ghiggeri GM. Rituximab in children with steroid-dependent nephrotic syndrome: a multicenter, open-label, noninferiority, randomized controlled trial. J Am Soc Nephrol. 2015;26:2259–66.
Webb H, Jaureguiberry G, Dufek S, Tullus K, Bockenhauer D. Cyclophosphamide and rituximab in frequently relapsing/steroid-dependent nephrotic syndrome. Pediatr Nephrol. 2016;31:589–94.
Basu B, Sander A, Roy B, Preussler S, Barua S, Mahapatra TKS, Schaefer F. Efficacy of rituximab vs tacrolimus in pediatric corticosteroid-dependent nephrotic syndrome: a randomized clinical trial. JAMA Pediatr. 2018;172:757–64.
Kari JA, Alhasan KA, Albanna AS, Safdar OY, Shalaby MA, Böckenhauer D, El-Desoky SM. Rituximab versus cyclophosphamide as first steroid-sparing agent in childhood frequently relapsing and steroid-dependent nephrotic syndrome. Pediatr Nephrol. 2020;35:1445–53.
Ravani P, Lugani F, Pisani I, Bodria M, Piaggio G, Bartolomeo D, Prunotto M, Ghiggeri GM. Rituximab for very low dose steroid-dependent nephrotic syndrome in children: a randomized controlled study. Pediatr Nephrol. 2020;35:1437–44.
Wang L, Zhu J, Xia M, Hua R, Deng F. Comparison of rituximab, cyclophosphamide, and tacrolimus as first steroid-sparing agents for complicated relapsing/steroid-dependent nephrotic syndrome in children: an evaluation of the health-related quality of life. Arch Med Sci. 2022;18:275–8.
Ravani P, Lugani F, Drovandi S, Caridi G, Angeletti A, Ghiggeri GM. Rituximab vs low-dose mycophenolate mofetil in recurrence of steroid-dependent nephrotic syndrome in children and young adults: a randomized clinical trial. JAMA Pediatr. 2021;175:631–2.
Fujinaga S, Tomii Y. Profound effect of post-rituximab mycophenolate mofetil administration for persistent hypogammaglobulinemia in young children with steroid-dependent nephrotic syndrome. Clin Exp Nephrol. 2020;24:386–7.
Parmentier C, Delbet JD, Decramer S, Boyer O, Hogan J, Ulinski T. Immunoglobulin serum levels in rituximab-treated patients with steroid-dependent nephrotic syndrome. Pediatr Nephrol. 2020;35:455–62.
Kamei K, Takahashi M, Fuyama M, Saida K, Machida H, Sato M, Ogura M, Ito S. Rituximab-associated agranulocytosis in children with refractory idiopathic nephrotic syndrome: case series and review of literature. Nephrol Dial Transplant. 2015;30:91–6.
Fujinaga S, Tomii Y. Predictors of rituximab-related neutropenia in Japanese children with steroid-dependent nephrotic syndrome. Pediatr Nephrol. 2019;34:1303–4.
Fujinaga S, Hirano D, Mizutani A, Sakuraya K, Yamada A, Sakurai S, Shimizu T. Predictors of relapse and long-term outcome in children with steroid-dependent nephrotic syndrome after rituximab treatment. Clin Exp Nephrol. 2017;21:671–6.
Ishikura K, Matsumoto S, Sako M, Tsuruga K, Nakanishi K, Kamei K, Saito H, Fujinaga S, Hamasaki Y, Chikamoto H, Ohtsuka Y, Komatsu Y, Ohta T, Nagai T, Kaito H, Kondo S, Ikezumi Y, Tanaka S, Kaku Y, Iijima K, Japanese Society for Pediatric Nephrology; Japanese Society for Pediatric Nephrology. Clinical practice guideline for pediatric idiopathic nephrotic syndrome 2013: medical therapy. Clin Exp Nephrol. 2015;19:6–33.
Onuki Y, Fujinaga S. Long-term prognosis of rituximab-induced hypogammaglobulinemia in children with complicated steroid-dependent nephrotic syndrome: impact of multiple rituximab courses. Pediatr Nephrol. 2023;38:615–6.
Trompeter RS, Lloyd BW, Hicks J, White RH, Cameron JS. Long-term outcome for children with minimal-change nephrotic syndrome. Lancet. 1985;16:368–70.
Lewis MA, Baildom EM, Davis N, Houston IB, Postlethwaite RJ. Nephrotic syndrome: from toddlers to twenties. Lancet. 1989;4:255–9.
Fakhouri F, Bocquet N, Taupin P, Presne C, Gagnadoux MF, Landais P, Lesavre P, Chauveau D, Knebelmann B, Broyer M, Grünfeld JP, Niaudet P. Steroid-sensitive nephrotic syndrome: from childhood to adulthood. Am J Kidney Dis. 2003;41:550–7.
Rüth EM, Kemper MJ, Leumann EP, Laube GF, Neuhaus TJ. Children with steroid-sensitive nephrotic syndrome come of age: long-term outcome. J Pediatr. 2005;147:202–7.
Ishikura K, Yoshikawa N, Nakazato H, Sasaki S, Nakanishi K, Matsuyama T, Ito S, Hamasaki Y, Yata N, Ando T, Iijima K, Honda M; Japanese Study Group of Renal Disease in Children. Morbidity in children with frequently relapsing nephrosis: 10-year follow-up of a randomized controlled trial. Pediatr Nephrol. 2015;30:459–68.
Mathew G, Sinha A, Ahmed A, Grewal N, Khandelwal P, Hari P, Bagga A. Efficacy of rituximab versus tacrolimus in difficult-to-treat steroid-sensitive nephrotic syndrome: an open-label pilot randomized controlled trial. Pediatr Nephrol. 2022;37:3117–26.
Sinha A, Mathew G, Arushi A, Govindarajan S, Ghanapriya K, Grewal N, Rai K, Brijwal M, Kalluru SL, Tewari P, Misra A, Khandelwal P, Hari P, Bagga A. Sequential rituximab therapy sustains remission of nephrotic syndrome but carries high risk of adverse effects. Nephrol Dial Transplant. 2022. https://doi.org/10.1093/ndt/gfac228.
Vester U, Kranz B, Zimmermann S, Hoyer PF. Cyclophosphamide in steroid-sensitive nephrotic syndrome: outcome and outlook. Pediatr Nephrol. 2023;18:661–4.
Azib S, Macher MA, Kwon T, Dechartres A, Alberti C, Loirat C, Deschênes G, Baudouin V. Cyclophosphamide in steroid-dependent nephrotic syndrome. Pediatr Nephrol. 2011;26:927–32.
Sandhu J, Bhat D, Dhooria GS, Pooni PA, Bhargava S, Kakkar S, Gill KS. Oral cyclophosphamide therapy in 100 children with steroid-sensitive nephrotic syndrome: experience from a developing country. Pediatr Nephrol. 2011;36:2759–67.
Acknowledgements
The authors would like to thank MARUZEN-YUSHODO Co., Ltd. for the English language editing.
Funding
SF has received Grants-in-Aid for research of Saitama Children's Medical Center (2021-B-3).
Author information
Authors and Affiliations
Contributions
RO and SF designed the study, drafted the manuscript, were responsible for the data integrity and analysis results. DH provided statistical support. All authors critically reviewed and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
Ito received grants from Zenyaku Kogyo Co., Ltd. and lectures fees from Chugai Pharmaceutical Co., Ltd.
Ethical approval
All procedures performed in this study involving human participants were in accordance with the ethical standards set by the ethics committee of Saitama Children’s Medical Center in which the study was conducted (approval number, 2022–03-010) and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
The Institutional Review Board waived the requirement for informed consent due to the retrospective nature of this study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Ohyama, R., Fujinaga, S., Sakuraya, K. et al. Predictive factors of long-term disease remission after rituximab administration in patients with childhood-onset complicated steroid-dependent nephrotic syndrome: a single-center retrospective study. Clin Exp Nephrol 27, 865–872 (2023). https://doi.org/10.1007/s10157-023-02374-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10157-023-02374-6