Abstract
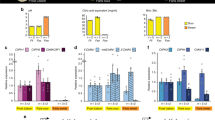
The sour taste of lemons (Citrus limon (L.) Burm.) is determined by the amount of citric acid in vacuoles of juice sac cells. Faris is a “sweet” lemon variety since it accumulates low levels of citric acid. The University of California Riverside Citrus Variety Collection includes a Faris tree that produces sweet (Faris non-acid; FNA) and sour fruit (Faris acid; FA) on different branches; it is apparently a graft chimera with layer L1 derived from Millsweet limetta and layer L2 from a standard lemon. The transcription profiles of Faris sweet lemon were compared with Faris acid lemon and Frost Lisbon (L), which is a standard sour lemon genetically indistinguishable from Faris in prior work with SSR markers. Analysis of microarray data revealed that the transcriptomes of the two sour lemon genotypes were nearly identical. In contrast, the transcriptome of Faris sweet lemon was very different from those of both sour lemons. Among about 1,000 FNA-specific, presumably pH-related genes, the homolog of Arabidopsis H+-ATPase proton pump AHA10 was not expressed in FNA, but highly expressed in FA and L. Since Arabidopsis AHA10 is involved in biosynthesis and acidification of vacuoles, the lack of expression of the AHA10 citrus homolog represents a very conspicuous molecular feature of the FNA sweet phenotype. In addition, high expression of several 2-oxoglutarate degradation-related genes in FNA suggests activation of the GABA shunt and degradation of valine and tyrosine as components of the mechanism that reduces the level of citric acid in sweet lemon.





Similar content being viewed by others
References
Ap Rees T, Hill SA (1994) Metabolic control analysis of plant metabolism. Plant Cell Environ 12:587–599
Aprile A, Federici C, Close T, De Bellis L, Cattivelli L, Roose M (2011) High and low acid lemons: origin and transcriptome comparisons. Acta Hort 892:37–42
Baxter IR, Young JC, Armstrong G, Foster N, Bogenschutz N, Cordova T, Peer WA, Hazen SP, Murphy AS, Harper JF (2005) A plasma membrane H-ATPase is required for the formation of proanthocyanidins in the seed coat endothelium of Arabidopsis thaliana. Proc Natl Acad Sci U S A 102:2649–2654
Benjamini Y, Hochberg Y (1995) Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Stat Soc 57:289–300
Bogin E, Erickson LC (1965) Activity of mitochondrial preparations obtained from Faris sweet lemon fruit. Plant Physiol 40:566–569
Bogin E, Wallace A (1966) Organic acid synthesis and accumulation in sweet and sour lemon fruit. J Amer Soc Hort Sci 89:182–194
Bown AW, Shelp BJ (1997) The metabolism and functions of γ-aminobutyric acid. Plant Physiol 115:1–5
Canel C, Bailey-Serres JN, Roose ML (1995) In vitro [14C] citrate uptake by tonoplast vesicles of acidless citrus juice cells. J Amer Soc Hort Sci 120:510–514
Canel C, Bailey-Serres JN, Roose ML (1996) Molecular characterization of the mitochondrial citrate synthase gene of an acidless pummelo. Plant Mol Biol 31:143–147
Cercos M, Soler G, Iglesias DJ, Gadea J, Forment J, Talon M (2006) Global analysis of gene expression during development and ripening of citrus fruit flesh. A proposed mechanism for citric acid utilization. Plant Mol Biol 62:513–527
Chapot H (1964) Citrons doux… ou acides? Cah Rech Agron 18:105–112
Chen JJ, Wang S-J, Tsai C-A, Lin C-J (2007) Selection of differentially expressed genes in microarray data analysis. Pharmacogenomics J 7:212–220
Czechowski T, Stitt M, Altmann T, Udvardi MK, Scheible W-R (2005) Genome-wide identification and testing of superior reference genes for transcript normalization in Arabidopsis. Plant Physiol 139:5–17
Daschner K, Coue I, Binder S (2001) The mitochondrial isovaleryl-coenzyme A dehydrogenase of Arabidopsis oxidizes intermediates of leucine and valine catabolism. Plant Physiol 126:601–612
Fatland BL, Ke J, Anderson MD, Mentzen WI, Cui LW, Allred CC, Johnston JL, Nikolau BJ, Wurtele ES (2002) Molecular characterization of the heteromeric ATP-citrate lyase that generate cytosolic acetyl-coenzyme A in Arabidopsis. Plant Physiol 130:740–756
Gaxiola RA, Palmgren MG, Schumacher K (2007) Plant proton pumps. FEBS Lett 581:2204–2214
Gulsen O, Roose M (2001) Lemons: diversity and relationships with selected citrus genotypes as measured with nuclear genome markers. J Amer Soc Hort Sci 126:309–317
Heyer LJ, Kruglyak S, Yooseph S (1999) Exploring expression data: identification and analysis of coexpressed genes. Genome Res 9:1106–1115
Irizarry RA, Bolstad BM, Collin F, Cope LM, Hobbs B, Speed TP (2003) Summaries of affymetrix genechip probe level data. Nucleic Acids Res 31(4):e15
Irizarry RA, Gautier L, Bolstad BM, Miller C with contributions from Astrand M, Cope LM, Gentleman R, Gentry J, Halling C, Huber W, MacDonald J, Rubinstein BIP, Workman C and Zhang J (2006) Affy: methods for affymetrix oligonucleotide arrays. R package version 1.12.1
Kanehisa M, Goto S, Hattori M, Aoki-Kinoshita KF, Itoh M, Kawashima S, Katayama T, Araki M, Hirakawa M (2006) From genomics to chemical genomics: new developments in KEGG. Nucleic Acids Res 34:D354–D357
Lam HM, Coschigano KT, Oliveira IC, Melo-Oliveira R, Coruzzi GM (1996) The molecular-genetics of nitrogen assimilation into amino acids in higher. Ann Rev Plant Physiol Plant Molec Biol 47:569–593
Lee PD, Sladek R, Greenwood CMT, Hudson TJ (2001) Control genes and variability: absence of ubiquitous reference transcripts in diverse mammalian expression studies. Genome Res 12:292–297
Lobyt P, Genard M, Wu BH, Soing P, Habib R (2003) Modelling citrate metabolism in fruit: responses to growth and temperature. J Exp Bot 54:2489–2501
Martinoia E, Ratajczak R (1997) Transport of organic molecules across the tonoplast. Adv Bot Res 25:365–400
Muller ML, Taiz L (2002) Regulation of the lemon-fruit V-ATPase by variable stoichiometry and organic acids. J Membr Biol 185:209–220
Muller ML, IrkensKiesecker U, Kramer D, Taiz L (1997) Purification and reconstitution of the vacuolar H+-ATPases from lemon fruits and epicotyls. J Biol Chem 272:12762–12770
Oleski N, Mahdavi P, Bennett AB (1987) Transport properties of the tomato fruit tonoplast. II. Citrate transport. Plant Physiol 84:997–1000
Plaxton WC, Podestà FE (2006) The functional organization and control of plant respiration. Crit Rev Plant Sci 25:159–198
Rea PA, Sanders D (1987) Tonoplast energization: two H+ pumps, one membrane. Physiol Plant 71:131–141
Rentsch D, Martinoia E (1991) Citrate transport into barley mesophyll vacuoles—comparison with malate-uptake activity. Planta 184:532–537
Ruepp A, Zollner A, Maier D, Albermann K, Hani J, Mokrejs M, Tetko I, Guldener U, Mannhaupt G, Munsterkotter M, Mewes HW (2004) The FunCat, a functional annotation scheme for systematic classification of proteins from whole genomes. Nucl Acids Res 32:5539–5545
Sadka A, Dahan E, Cohen L, Marsh KB (2000a) Aconitase activity and expression during the development of lemon fruit. Physiol Plant 108:255–262
Sadka A, Dahan E, Or E, Cohen L (2000b) NADP+-isocitrate dehydrogenase gene expression and isozyme activity during citrus fruit development. Plant Sci 158:173–181
Sadka A, Dahan E, Or E, Roose ML, Marsh KB, Cohen L (2001) Comparative analysis of citrate synthase gene structure, transcript level and enzymatic activity in acidless and acid-containing citrus varieties. Austral J Plant Physiol 28:383–390
Shimada T, Nakano R, Shulaev V, Sadka A, Blumwald E (2006) Vacuolar citrate/H+ symporter of citrus juice cells. Planta 224:472–480
Subramanian S, Sivaraman C (1984) Bacterial citrate lyase. J Biosci 6:379–401
Suzuki T, Higgins PJ, Crawford DR (2000) Control selection for RNA quantitation. Biotechniques 29:332–337
Thellin O, Zorzi W, Lakaye B, De Borman B, Coumans B, Henne G, Grisar T, Igout A, Heinen E (1999) Housekeeping genes as internal standards: use and limits. J Biotechnol 75:197–200
Ting SV, Attaway JA (1971) Citrus fruits. In: Hulme AC (ed) The biochemistry of fruits and their products, Vol. 2. Academic, London, pp 107–169
Tucker GA (1993) Introduction. In: Seymour G, Taylor J, Tucker G (eds) Biochemistry of fruit ripening. Chapman and Hall, London, pp 1–37
Yeung KY, Ruzzo WL (2001) Principal component analysis for clustering gene expression data. Bioinformatics 17:763–774
Acknowledgments
The authors would like to thank Ottilia Bier for bringing to our attention the presence of sour fruit on the Faris lemon trees, Ray Fenton for isolating the RNA, Jan Svensson for cooperation on identification of reference transcripts, and Barbara Walter for labeling the RNA and processing the gene chips. Avi Sadka provided helpful advice and comments on the results.
Author information
Authors and Affiliations
Corresponding author
Additional information
Financial Source: California Citrus Research Board, University of California Industry-University Cooperative Research Program, Affymetrix Inc. Italian Ministry of Agriculture special project “CITRUSTART”
Electronic supplementary materials
Below is the link to the electronic supplementary material.
Figure S1
All QT clusters. Three genotypes are plotted on the x-axis, young samples on the left and mature samples on the right. The relative expression level (data normalized to the median for each probe set) is plotted on the y-axis. The lines represent the average expression of all probe sets of a cluster. The cluster “unclassified” contain the 1,202 probe sets that did not satisfy either of the two selected thresholds. FNA Faris non-acid, FA Faris acid, L Frost Lisbon (JPEG 140 kb)
Figure S2
Branched-chain amino acids catabolism pathway. Differentially expressed genes are in solid line boxes. Not differentially expressed genes are in dotted line boxes. BCAT branched-chain amino acid transaminase, BDKD branched-chain keto-acid dehydrogenase, BDH 3-hydroxyisobutyrate dehydrogenase (JPEG 121 kb)
Table S1
Summary of all differentially expressed genes. In column A are listed the probe set IDs. “Comparisons” columns show the results of t test analysis. An empty cell means a non-significant result. For each probe set the relative gene ontology (GO) and Arabidopsis best match are shown (“Annotation” columns). Columns R to W report expression levels and columns X to AA the fold changes in sweet vs sour lemons (XLS 2208 kb)
Table S2
Functional classification of the cluster 1 genes using FunCat (Ruepp et al. 2004). Each category is sorted by increasing p value. Categories with p value lower than 0.05 (red) were considered over-represented in cluster 1 relative to observed Arabidopsis genome frequencies (XLS 91 kb)
Table S3
Functional classification of the cluster 2 genes using FunCat (Ruepp et al. 2004). Each category is sorted by increasing p value. Categories with p value lower than 0.05 (red) were considered over-represented in cluster 2 relative to observed Arabidopsis genome frequencies (XLS 90 kb)
Table S4
List of the probe sets that were not expressed in FNA samples and were expressed in FA and L samples. We report the probe set ID, the relative QT cluster, gene ontologies, Arabidopsis best match and expression level (XLS 38 kb)
Table S5
List of all the probe sets represented on Affymetrix Citrus GeneChip® related to glycolysis, acetyl-CoA synthesis, TCA cycle, citrate catabolism, glutamate synthesis, GABA shunt, tyrosine catabolism, branched-chain amino acid catabolism, and proton pumps. The last columns show the relative expression levels and fold changes in sweet vs sour lemons (XLS 125 kb)
Rights and permissions
About this article
Cite this article
Aprile, A., Federici, C., Close, T.J. et al. Expression of the H+-ATPase AHA10 proton pump is associated with citric acid accumulation in lemon juice sac cells. Funct Integr Genomics 11, 551–563 (2011). https://doi.org/10.1007/s10142-011-0226-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10142-011-0226-3