Abstract
Purpose
Rapid automated CT volumetry of pulmonary contusion may predict progression to Acute Respiratory Distress Syndrome (ARDS) and help guide early clinical management in at-risk trauma patients. This study aims to train and validate state-of-the-art deep learning models to quantify pulmonary contusion as a percentage of total lung volume (Lung Contusion Index, or auto-LCI) and assess the relationship between auto-LCI and relevant clinical outcomes.
Methods
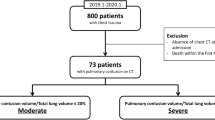
302 adult patients (age ≥ 18) with pulmonary contusion were retrospectively identified from reports between 2016 and 2021. nnU-Net was trained on manual contusion and whole-lung segmentations. Point-of-care candidate variables for multivariate regression included oxygen saturation, heart rate, and systolic blood pressure on admission. Logistic regression was used to assess ARDS risk, and Cox proportional hazards models were used to determine differences in ICU length of stay and mechanical ventilation time.
Results
Mean Volume Similarity Index and mean Dice scores were 0.82 and 0.67. Interclass correlation coefficient and Pearson r between ground-truth and predicted volumes were 0.90 and 0.91. 38 (14%) patients developed ARDS. In bivariate analysis, auto-LCI was associated with ARDS (p < 0.001), ICU admission (p < 0.001), and need for mechanical ventilation (p < 0.001). In multivariate analyses, auto-LCI was associated with ARDS (p = 0.04), longer length of stay in the ICU (p = 0.02) and longer time on mechanical ventilation (p = 0.04). AUC of multivariate regression to predict ARDS using auto-LCI and clinical variables was 0.70 while AUC using auto-LCI alone was 0.68.
Conclusion
Increasing auto-LCI values corresponded with increased risk of ARDS, longer ICU admissions, and longer periods of mechanical ventilation.


Similar content being viewed by others
Data availability
Data supporting the findings of this study are available upon request to the corresponding author.
References
Ahmad Ganie F, Lone H, Nabi Lone G, Lateef Wani M, Singh S, Majeed Dar A et al (2023) Lung contusion: a clinico-pathological entity with unpredictable clinical course. Bull Emerg Trauma 1:7–16
Cohn SM, DuBose JJ (2010) Pulmonary contusion: an update on recent advances in clinical management. World J Surg 34(8):1959–1970. https://doi.org/10.1007/S00268-010-0599-9
Chong WH, Saha BK, Austin A, Chopra A (2021) The significance of subpleural sparing in ct chest: a state-of-the-art review. Am J Med Sci 361:427–435. https://doi.org/10.1016/J.AMJMS.2021.01.008
Donnelly LF, Klosterman LA (1997) Subpleural sparing: a CT finding of lung contusion in children. Radiology 204:385–387. https://doi.org/10.1148/RADIOLOGY.204.2.9240524
Miller PR, Croce MA, Bee TK, Qaisi WG, Smith CP, Collins GL et al (2001) ARDS after pulmonary contusion: accurate measurement of contusion volume identifies high-risk patients. J Trauma 51:223–230. https://doi.org/10.1097/00005373-200108000-00003
Bellani G, Laffey JG, Pham T, Fan E, Brochard L, Esteban A et al (2016) Epidemiology, patterns of care, and mortality for patients with acute respiratory distress syndrome in intensive care units in 50 countries. JAMA 315:788–800. https://doi.org/10.1001/JAMA.2016.0291
Villar J, Blanco J, Añón JM, Santos-Bouza A, Blanch L, Ambrós A et al (2011) The ALIEN study: incidence and outcome of acute respiratory distress syndrome in the era of lung protective ventilation. Intensive Care Med 37:1932–1941. https://doi.org/10.1007/S00134-011-2380-4
Sayed MS, Elmeslmany KA, Elsawy AS, Mohamed NA (2022) The validity of quantifying pulmonary contusion extent by lung ultrasound score for predicting ARDS in blunt thoracic trauma. Crit Care Res Pract 2022:1. https://doi.org/10.1155/2022/3124966
Zingg SW, Millar DA, Goodman MD, Pritts TA, Janowak CF (2021) The association between pulmonary contusion severity and respiratory failure. Respir Care 66:1665–1672. https://doi.org/10.4187/RESPCARE.09145
Choi J, Tennakoon L, You JG, Kaghazchi A, Forrester JD, Spain DA (2021) Pulmonary contusions in patients with rib fractures: The need to better classify a common injury. Am J Surg 221:211–215. https://doi.org/10.1016/j.amjsurg.2020.07.022
Choi J, Mavrommati K, Li NY, Patil A, Chen K, Hindin DI et al (2022) Scalable deep learning algorithm to compute percent pulmonary contusion among patients with rib fractures. J Trauma Acute Care Surg 93:461. https://doi.org/10.1097/TA.0000000000003619
Dreizin D, Zhou Y, Zhang Y, Tirada N, Yuille AL (2020) Performance of a deep learning algorithm for automated segmentation and quantification of traumatic pelvic hematomas on CT. J Digit Imaging 33:243–251. https://doi.org/10.1007/S10278-019-00207-1
Zhou Y, Dreizin D, Wang Y, Liu F, Shen W, Yuille AL (2022) External attention assisted multi-phase splenic vascular injury segmentation with limited data. IEEE Trans Med Imaging 41:1346–1357. https://doi.org/10.1109/TMI.2021.3139637
Dreizin D, Zhou Y, Chen T, Li G, Yuille AL, McLenithan A et al (2020) Deep learning-based quantitative visualization and measurement of extraperitoneal hematoma volumes in patients with pelvic fractures: Potential role in personalized forecasting and decision support. J Trauma Acute Care Surg 88:425–433. https://doi.org/10.1097/TA.0000000000002566
Dreizin D, Zhou Y, Fu S, Wang Y, Li G, Champ K et al (2020) a multiscale deep learning method for quantitative visualization of traumatic hemoperitoneum at CT: assessment of feasibility and comparison with subjective categorical estimation. Radiol Artif Intell 2:1–9. https://doi.org/10.1148/RYAI.2020190220
Dreizin D, Chen T, Liang Y, Zhou Y, Paes F, Wang Y et al (2021) Added value of deep learning-based liver parenchymal CT volumetry for predicting major arterial injury after blunt hepatic trauma: a decision tree analysis. Abdom Radiol (NY) 46:2556–2566. https://doi.org/10.1007/S00261-020-02892-X
Chen H, Unberath M, Dreizin D (2023) Toward automated interpretable AAST grading for blunt splenic injury. Emerg Radiol 30:41–50. https://doi.org/10.1007/S10140-022-02099-1
Dreizin D, Nixon B, Hu J, Albert B, Yan C, Yang G et al (2022) A pilot study of deep learning-based CT volumetry for traumatic hemothorax. Emerg Radiol 29:995. https://doi.org/10.1007/S10140-022-02087-5
Zhou Y, Dreizin D, Li Y, Zhang Z, Wang Y, Yuille A (2019) Multi-scale attentional network for multi-focal segmentation of active bleed after pelvic fractures. Lecture Notes Comput Sci (Including Subseries Lecture Notes in Artif Intell Lecture Notes Bioinformatics) 11861 LNCS:461–9. https://doi.org/10.1007/978-3-030-32692-0_53/COVER
Roth HR, Xu Z, Tor-Díez C, Sanchez Jacob R, Zember J, Molto J et al (2022) Rapid artificial intelligence solutions in a pandemic—The COVID-19–20 Lung CT Lesion Segmentation Challenge. Med Image Anal 82:102605. https://doi.org/10.1016/j.media.2022.102605
Lessmann N, Sánchez CI, Beenen L, Boulogne LH, Brink M, Calli E et al (2021) Automated assessment of COVID-19 reporting and data system and chest CT severity scores in patients suspected of having COVID-19 using artificial intelligence. Radiology 298:E18-28. https://doi.org/10.1148/RADIOL.2020202439/ASSET/IMAGES/LARGE/RADIOL.2020202439.FIG6.JPEG
Fedorov A, Beichel R, Kalpathy-Cramer J, Finet J, Fillion-Robin JC, Pujol S et al (2012) 3D Slicer as an image computing platform for the Quantitative Imaging Network. Magn Reson Imaging 30:1323–1341. https://doi.org/10.1016/j.mri.2012.05.001
Loftis KL, Price J, Gillich PJ (2018) Evolution of the Abbreviated Injury Scale: 1990–2015. Traffic Inj Prev 19:S109–S113. https://doi.org/10.1080/15389588.2018.1512747
Baker SP, O’Neill B, Haddon W, Long WB (1974) The injury severity score: a method for describing patients with multiple injuries and evaluating emergency care. J Trauma 14:187–196. https://doi.org/10.1097/00005373-197403000-00001
Ferguson ND, Fan E, Camporota L, Antonelli M, Anzueto A, Beale R et al (2012) The Berlin definition of ARDS: an expanded rationale, justification, and supplementary material. Intensive Care Med 38:1573–1582. https://doi.org/10.1007/S00134-012-2682-1
Isensee F, Jaeger PF, Kohl SAA, Petersen J, Maier-Hein KH (2020) nnU-Net: a self-configuring method for deep learning-based biomedical image segmentation. Nat Methods 18(2):203–11. https://doi.org/10.1038/s41592-020-01008-z
Litjens G, Kooi T, Bejnordi BE, Setio AAA, Ciompi F, Ghafoorian M et al (2017) A survey on deep learning in medical image analysis. Med Image Anal 42:60–88. https://doi.org/10.1016/J.MEDIA.2017.07.005
Aerts HJWL, Velazquez ER, Leijenaar RTH, Parmar C, Grossmann P, Cavalho S et al (2014) Decoding tumour phenotype by noninvasive imaging using a quantitative radiomics approach. Nat Commun 5:1–9. https://doi.org/10.1038/ncomms5006
Zou KH, Tuncali K, Silverman SG (2003) Correlation and simple linear regression. Radiology 227:617–622. https://doi.org/10.1148/RADIOL.2273011499
Dreizin D, Munera F (2012) Blunt polytrauma: evaluation with 64-section whole-body CT angiography. Radiographics 32:609–632. https://doi.org/10.1148/RG.323115099
Sangster GP, González-Beicos A, Carbo AI, Heldmann MG, Ibrahim H, Carrascosa P et al (2007) Blunt traumatic injuries of the lung parenchyma, pleura, thoracic wall, and intrathoracic airways: multidetector computer tomography imaging findings. Emerg Radiol 14:297–310. https://doi.org/10.1007/S10140-007-0651-8
Clark GC, Schecter WP, Trunkey DD (1988) Variables affecting outcome in blunt chest trauma: flail chest vs. pulmonary contusion. J Trauma 28:298–304. https://doi.org/10.1097/00005373-198803000-00004
Navarrete-Navarro P, Rodriguez A, Reynolds N, West R, Habashi N, Rivera R et al (2001) Acute respiratory distress syndrome among trauma patients: trends in ICU mortality, risk factors, complications and resource utilization. Intensive Care Med 27:1133–1140. https://doi.org/10.1007/S001340100955
Hudson LD, Milberg JA, Anardi D, Maunder RJ (1995) Clinical risks for development of the acute respiratory distress syndrome. Am J Respir Crit Care Med 151:293–301. https://doi.org/10.1164/AJRCCM.151.2.7842182
Hoyt DB, Simons RK, Winchell RJ, Cushman J, Hollingsworth-Fridlund P, Holbrook T et al (1993) A risk analysis of pulmonary complications following major trauma. J Trauma 35:524–531. https://doi.org/10.1097/00005373-199310000-00005
Bellani G, Pham T, Laffey JG (2020) Missed or delayed diagnosis of ARDS: a common and serious problem. Intensive Care Med 46:1180–1183. https://doi.org/10.1007/S00134-020-06035-0/FIGURES/1
Yadav H, Thompson BT, Gajic O (2017) Is acute respiratory distress syndrome a preventable disease? Am J Respir Crit Care Med 195:725–736. https://doi.org/10.1164/RCCM.201609-1767CI/SUPPL_FILE/DISCLOSURES.PDF
Dreizin D, Staziaki PV, Khatri GD, Beckmann NM, Feng Z, Liang Y et al (2023) Artificial intelligence CAD tools in trauma imaging: a scoping review from the American Society of Emergency Radiology (ASER) AI/ML Expert Panel. Emerg Radiol 30:251. https://doi.org/10.1007/S10140-023-02120-1
Agrawal A, Khatri GD, Khurana B, Sodickson AD, Liang Y, Dreizin D (2023) A survey of ASER members on artificial intelligence in emergency radiology: trends, perceptions, and expectations. Emerg Radiol 30:267. https://doi.org/10.1007/S10140-023-02121-0
Guo Y, Liu Y, Georgiou T, Lew MS (2018) A review of semantic segmentation using deep neural networks. Int J Multimed Inf Retr 7:87–93. https://doi.org/10.1007/S13735-017-0141-Z/FIGURES/3
Röhrich S, Hofmanninger J, Negrin L, Langs G, Prosch H (2021) Radiomics score predicts acute respiratory distress syndrome based on the initial CT scan after trauma. Eur Radiol 31:5443–5453. https://doi.org/10.1007/S00330-020-07635-6/TABLES/5
Roth HR, Xu Z, Diez CT, Jacob RS, Zember J, Molto J, et al (2021) Rapid artificial intelligence solutions in a pandemic - the Covid-19–20 lung CT lesion segmentation challenge. Res Sq. https://doi.org/10.21203/RS.3.RS-571332/V1
Zhang L, LaBelle W, Unberath M, Chen H, Hu J, Li G, et al (2023) A vendor-agnostic, PACS integrated, and DICOM-compatible software-server pipeline for testing segmentation algorithms within the clinical radiology workflow. Res Sq. https://doi.org/10.21203/RS.3.RS-2837634/V1
Oikonomou A, Prassopoulos P (2011) CT imaging of blunt chest trauma. Insights Imaging 2:281. https://doi.org/10.1007/S13244-011-0072-9
Dreizin D, Rosales R, Li G, Syed H, Chen R (2021) Volumetric markers of body composition may improve personalized prediction of major arterial bleeding after pelvic fracture: a secondary analysis of the Baltimore CT prediction model cohort. Can Assoc Radiol J 72:854–861. https://doi.org/10.1177/0846537120952508
Acknowledgements
NIH K08 EB027141-01A1 (PI: David Dreizin, MD)
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare that are relevant to the content of this article.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sarkar, N., Zhang, L., Campbell, P. et al. Pulmonary contusion: automated deep learning-based quantitative visualization. Emerg Radiol 30, 435–441 (2023). https://doi.org/10.1007/s10140-023-02149-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10140-023-02149-2