Abstract
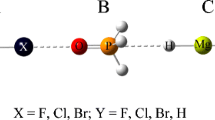
The pnicogen and halogen bonding interactions in the PH2X---BrCl(X = H, F, OH, OCH3 and CH3) complexes have been studied at the MP2/aug-cc-pVTZ level. Analysis of interaction energies shows that the pnicogen-bonded structures are less stable than the corresponding halogen-bonded structures. The pnicogen and halogen bonds were also studied by conceptual DFT reactivity indices. Noncovalent interaction (NCI) and SAPT analysis reveals that the dispersion interactions dominate the pnicogen-bonded complexes of PH2X---BrCl in nature, while the halogen-bonded complexes are dominantly electrostatic energy.

It is found that the local softness s+ or s−on the basic center P of PH2X is related to the interaction energies (ΔECP) of halogen- or pnicogen-bonded complexes.







Similar content being viewed by others
References
Metrangolo P, Carcenac Y, Lahtinen M, Pilati T, Rissanen K, Vij A, Resnati G (2009) Science 323:1461–1464
Metrangolo P, Resnati G (2008) Halogen bonding in crystal engineering. Springer, Berlin
Legon AC (2010) Phys Chem Chem Phys 12:7736–7747
Zahn S, Frank R, Hey-Hawkins E, Kirchner B (2011) Chem Eur J 17:6034–6038
Scheiner SA (2011) J Chem Phys 134:094315–094323
Del Bene JE, Alkorta I, Sánchez-Sanz G, Elguero J (2011) Chem Phys Lett 512:184–187
Politzer P, Murray J, Clark T (2013) Phys Chem Chem Phys 15:11178–11189
Murray JS, Lane P, Politzer P (2007) Int J Quantum Chem 107:2286–2292
Politzer P, Murray J, Clark T (2015) J Mol Model 21(3):1–10
Clark T, Heßelmann A (2018) Phys Chem Chem Phys https://doi.org/10.1039/c8cp03079k
Pearson RG (1973) Hard and soft acids and bases. Dowen, Hutchinson and Ross, Stroudsberg
Peterson KA, Figgen D, Goll E, Stoll H, Dolg M (2003) J Chem Phys 119:11113–11123
Frisch MJ, Trucks GW, Schlegel HB et al (2004) GAUSSIAN 03, revision B03. Gaussian, Inc, Wallingford
Boys SF (1970) Mol Phys 19:553–566
Reed AE, Curtiss LA, Weinhold F (1988) Chem Rev 88:899–926
Bulat FA, Toro-Labbe A, Brinck T, Murray JS, Politzer P (2010) J Mol Model 16:1679–1691
Popelier PLA (2000) Atoms in molecules: an introduction. Prentice Hall, Harlow
Popelier PLA (2000) Atoms in molecules: an introduction. Prentice Hall, Harlow
Keith TA (2011) AIMAll, version 10.05.04. aim.tkgristmill.com
Lu T, Chen FW (2012) J Comput Chem 33:580–592
Humphrey W, Dalke A, Schulten K (1996) J Mol Graph 14:33–38
Jeziorski B, Moszynski R, Szalewicz K (1994) Chem. Rev. 94:1887–1930
Jeziorski B, Moszynski R, Szalewicz K (1994) Chem. Rev. 94:1887–1930
Bukowski R, Cencek W, Jankowski P, Jeziorski B, Jeziorska M, Kucharski SA, Misquitta AJ, Moszynski R, Patkowski K, Rybak S, Szalewicz K, Williams HL, Wormer PES (2012) SAPT2012: An ab initio program for many-body symmetryadapted perturbation theory calculations of intermolecular interaction energies. Sequential and parallel versions. University of Delaware, Newark; University of Warsaw, Warsaw
Rozas I, Alkorta I, Elguero J (2000) J Am Chem Soc 122:11154–11161
Bader RFW (1990) Atoms in molecules: a quantum theory. Oxford University Press, Oxford
Politzer P, Murray JS, Clark T (2010) Phys Chem Chem Phys 12:7748–7757
Johnson ER, Keinan S, Mori-Sanchez P, Contreras-Garcia J, Cohen AJ, Yang W (2010) J Am Chem Soc 132:6498–6506
Gao M, Li QZ, Li HB, Li WZ, Chen JB (2015) RSC Adv 5:12488–12497
Riley KE, Hobza P (2008) J Chem Theory Comput 4:232–242
Lee CT, Yang WT, Parr RG (1988) J Mol Struct THEOCHEM 40:305–313
Yang WT, Parr RG (1985) Proc Natl Acad Sci U S A 82:6723–6726
Ayers PW, Levy M (2000) Theor Chem Accounts 103:353–360
Acknowledgments
This study was supported by grants from the National Science Foundations of China (21203135). This research is also supported by the first class subject of Zhejiang chemical engineering and Technology (Taizhou University).
Part of the calculations in this research were performed at the ScGrid of Supercomputing Center.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wu, J., Yan, H., Zhong, A. et al. Theoretical and conceptual DFT study of pnicogen- and halogen-bonded complexes of PH2X---BrCl. J Mol Model 25, 28 (2019). https://doi.org/10.1007/s00894-018-3905-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00894-018-3905-3