Abstract
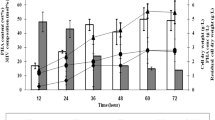
The extreme haloarchaea Haloferax mediterranei accumulates poly(3-hydroxybutyrate-co-3-hydroxyvalerate) (PHBV) without the need for specific precursors. In this study, growth kinetics and PHBV synthesis were characterised under nitrogen-excess and nitrogen-limiting conditions in ammonium and, for the first time, nitrate. With excess nitrogen, ammonium and nitrate cultures generated 10.7 g/L biomass containing 4.6 wt% PHBV and 5.6 g/L biomass with 9.3 wt% PHBV, respectively. Copolymer composition varied with the nitrogen source used: PHBV from ammonium cultures had 16.9 mol% 3-hydroxyvalerate (HV), while PHBV from nitrate cultures contained 12.5 mol% HV. Nitrogen limitation was achieved with carbon-to-nitrogen (C/N) molar ratios of 25 or higher. Nitrogen limitation reduced biomass generation and polymer concentration, but polymer accumulation increased to 6.6 and 9.4% for ammonium and nitrate, respectively, with C/N 42. PHBV composition was also affected and cultures with lower C/N ratios produced richer HV polymers. Copolymer formation was not a uniform process: HV was only detected after a minimum accumulation of 0.45 g/L PHB and lasted for a maximum of 48 h. The understanding of copolymer synthesis and the influence of culture conditions such as the nitrogen source will help in designing novel strategies for the production of PHBV with more regular structure and material properties.





Similar content being viewed by others
References
Bhattacharyya A, Saha J, Haldar S, Bhowmic A, Mukhopadhyay UK, Mukherjee J (2014) Production of poly-3-(hydroxybutyrate-co-hydroxyvalerate) by Haloferax mediterranei using rice-based ethanol stillage with simultaneous recovery and re-use of medium salts. Extremophiles 18:463–470. doi:10.1007/s00792-013-0622-9
Bonete MJ, Martinez-Espinosa RM, Pire C, Zafrilla B, Richardson DJ (2008) Nitrogen metabolism in haloarchaea. Saline Syst 4:9. doi:10.1186/1746-1448-4-9
Cabello P, Roldan MD, Moreno-Vivian C (2004) Nitrate reduction and the nitrogen cycle in archaea. Microbiology 150:3527–3546. doi:10.1099/mic.0.27303-0
Cavaillé L, Grousseau E, Pocquet M, Lepeuple A-S, Uribelarrea J-L, Hernandez-Raquet G, Paul E (2013) Polyhydroxybutyrate production by direct use of waste activated sludge in phosphorus-limited fed-batch culture. Biores Technol 149:301–309. doi:10.1016/j.biortech.2013.09.044
Chen CW, Don T-M, Yen H-F (2006) Enzymatic extruded starch as a carbon source for the production of poly(3-hydroxybutyrate-co-3-hydroxyvalerate) by Haloferax mediterranei. Process Biochem 41:2289–2296. doi:10.1016/j.procbio.2006.05.026
Cui Y-W, Zhang H-Y, Ji S-Y, Wang Z-W (2016) Kinetic analysis of the temperature effect on polyhydroxyalkanoate production by Haloferax mediterranei in synthetic molasses wastewater. J Polym Environ. doi:10.1007/s10924-016-0807-2
Don TM, Chen CW, Chan TH (2006) Preparation and characterization of poly(hydroxyalkanoate) from the fermentation of Haloferax mediterranei. J Biomater Sci Polym Ed 17:1425–1438
Esclapez J, Bravo-Barrales G, Bautista V, Pire C, Camacho M, Bonete MJ (2014) Effects of nitrogen sources on the nitrate assimilation in Haloferax mediterranei: growth kinetics and transcriptomic analysis. FEMS Microbiol Lett 350:168–174. doi:10.1111/1574-6968.12325
Fernandez-Castillo R, Rodriguez-Valera F, Gonzalez-Ramos J, Ruiz-Berraquero F (1986) Accumulation of poly(beta-hydroxybutyrate) by Halobacteria. Appl Environ Microbiol 51:214–216
Han J, Li M, Hou J, Wu L, Zhou J, Xiang H (2010) Comparison of four phaC genes from Haloferax mediterranei and their function in different PHBV copolymer biosyntheses in Haloarcula hispanica. Saline Syst 6:9. doi:10.1186/1746-1448-6-9
Han J et al (2013) Multiple propionyl coenzyme A-supplying pathways for production of the bioplastic poly(3-hydroxybutyrate-co-3-hydroxyvalerate) in Haloferax mediterranei. Appl Environ Microbiol 79:2922–2931. doi:10.1128/aem.03915-12
Han J, Wu LP, Hou J, Zhao D, Xiang H (2015) Biosynthesis, characterization, and hemostasis potential of tailor-made poly(3-hydroxybutyrate-co-3-hydroxyvalerate) produced by Haloferax mediterranei. Biomacromolecules 16:578–588. doi:10.1021/bm5016267
Hermann-Krauss C, Koller M, Muhr A, Fasl H, Stelzer F, Braunegg G (2013) Archaeal production of polyhydroxyalkanoate (PHA) co- and terpolyesters from biodiesel industry-derived by-products. Archaea 2013:10. doi:10.1155/2013/129268
Hou J, Xiang H, Han J (2015) Propionyl coenzyme A (propionyl-CoA) carboxylase in Haloferax mediterranei: indispensability for propionyl-CoA assimilation and impacts on global metabolism. Appl Environ Microbiol 81:794–804. doi:10.1128/AEM.03167-14
Koller M, Hesse P, Bona R, Kutschera C, Atlic A, Braunegg G (2007) Potential of various archae- and eubacterial strains as industrial polyhydroxyalkanoate producers from whey. Macromol Biosci 7:218–226. doi:10.1002/mabi.200600211
Koller M et al (2008) Polyhydroxyalkanoate production from whey by Pseudomonas hydrogenovora. Biores Technol 99:4854–4863. doi:10.1016/j.biortech.2007.09.049
Laycock B et al (2014a) Thermal properties and crystallization behavior of fractionated blocky and random polyhydroxyalkanoate copolymers from mixed microbial cultures. J Appl Polym Sci. doi:10.1002/app.40836
Laycock B, Halley P, Pratt S, Werker A, Lant P (2014b) The chemomechanical properties of microbial polyhydroxyalkanoates. Prog Polym Sci 39:397–442. doi:10.1016/j.progpolymsci.2013.06.008
Lee W-H, Loo C-Y, Nomura CT, Sudesh K (2008) Biosynthesis of polyhydroxyalkanoate copolymers from mixtures of plant oils and 3-hydroxyvalerate precursors. Biores Technol 99:6844–6851. doi:10.1016/j.biortech.2008.01.051
Lillo JG, Rodriguez-Valera F (1990) Effects of culture conditions on poly(β-hydroxybutyric acid) production by Haloferax mediterranei. Appl Environ Microbiol 56:2517–2521
Lledó B, Martı́nez-Espinosa RM, Marhuenda-Egea FC, Bonete MJ (2004) Respiratory nitrate reductase from haloarchaeon Haloferax mediterranei: biochemical and genetic analysis. Biochim Biophys Acta (BBA) Gen Subj 1674:50–59. doi:10.1016/j.bbagen.2004.05.007
Lorantfy B, Seyer B, Herwig C (2014) Stoichiometric and kinetic analysis of extreme halophilic Archaea on various substrates in a corrosion resistant bioreactor. New Biotechnol 31:80–89. doi:10.1016/j.nbt.2013.08.003
Lu Q, Han J, Zhou L, Zhou J, Xiang H (2008) Genetic and biochemical characterization of the poly(3-hydroxybutyrate-co-3-hydroxyvalerate) synthase in Haloferax mediterranei. J Bacteriol 190:4173–4180. doi:10.1128/JB.00134-08
Martinez-Espinosa RM, Marhuenda-Egea FC, Bonete MJ (2001) Assimilatory nitrate reductase from the haloarchaeon Haloferax mediterranei: purification and characterisation. FEMS Microbiol Lett 204:381–385
Martínez-Espinosa RM, Lledó B, Marhuenda-Egea FC, Bonete MJ (2007) The effect of ammonium on assimilatory nitrate reduction in the haloarchaeon Haloferax mediterranei. Extremophiles 11:759–767. doi:10.1007/s00792-007-0095-9
Meng D-C, Shi Z-Y, Wu L-P, Zhou Q, Wu Q, Chen J-C, Chen G-Q (2012) Production and characterization of poly(3-hydroxypropionate-co-4-hydroxybutyrate) with fully controllable structures by recombinant Escherichia coli containing an engineered pathway. Metab Eng 14:317–324. doi:10.1016/j.ymben.2012.04.003
Obruca S, Marova I, Snajdar O, Mravcova L, Svoboda Z (2010) Production of poly(3-hydroxybutyrate-co-3-hydroxyvalerate) by Cupriavidus necator from waste rapeseed oil using propanol as a precursor of 3-hydroxyvalerate. Biotechnol Lett 32:1925–1932. doi:10.1007/s10529-010-0376-8
Oren A (2010) Industrial and environmental applications of halophilic microorganisms. Environ Technol 31:825–834. doi:10.1080/09593330903370026
Oren A, Gurevich P (1994) Production of d-lactate, acetate, and pyruvate from glycerol in communities of halophilic archaea in the Dead Sea and in saltern crystallizer ponds. FEMS Microbiol Ecol 14:147–155. doi:10.1111/j.1574-6941.1994.tb00101.x
Pais J, Serafim LS, Freitas F, Reis MAM (2016) Conversion of cheese whey into poly(3-hydroxybutyrate-co-3-hydroxyvalerate) by Haloferax mediterranei. New Biotechnol 33:224–230. doi:10.1016/j.nbt.2015.06.001
Poli A, Di Donato P, Abbamondi GR, Nicolaus B (2011) Synthesis, production, and biotechnological applications of exopolysaccharides and polyhydroxyalkanoates by archaea. Archaea 2011:693253. doi:10.1155/2011/693253
Quillaguamán J, Delgado O, Mattiasson B, Hatti-Kaul R (2006) Poly(β-hydroxybutyrate) production by a moderate halophile, Halomonas boliviensis LC1. Enzyme and Microb Technol 38:148–154. doi:10.1016/j.enzmictec.2005.05.013
Quillaguamán J, Guzmán H, Van-Thuoc D, Hatti-Kaul R (2010) Synthesis and production of polyhydroxyalkanoates by halophiles: current potential and future prospects. Appl Microbiol Biotechnol 85:1687–1696. doi:10.1007/s00253-009-2397-6
Riedel SL, Bader J, Brigham CJ, Budde CF, Yusof ZAM, Rha C, Sinskey AJ (2012) Production of poly(3-hydroxybutyrate-co-3-hydroxyhexanoate) by Ralstonia eutropha in high cell density palm oil fermentations. Biotechnol Bioeng 109:74–83. doi:10.1002/bit.23283
Rodriguez-Valera F, Ruiz-Berraquero F, Ramos-Cormenzana A (1980) Short communication isolation of extremely halophilic bacteria able to grow in defined inorganic media with single carbon sources. Microbiology 119:535–538. doi:10.1099/00221287-119-2-535
Singh M, Kumar P, Ray S, Kalia VC (2015) Challenges and opportunities for customizing polyhydroxyalkanoates. Indian J Microbiol 55(3):235–249. doi:10.1007/s12088-015-0528-6
Sudesh K, Abe H, Doiu Y (2000) Synthesis, structure and properties of polyhydroxyalkanoates: biological polyesters. Prog Polym Sci 25:1503–1555. doi:10.1016/S0079-6700(00)00035-6
Tan D, Wu Q, Chen J-C, Chen G-Q (2014) Engineering Halomonas TD01 for the low-cost production of polyhydroxyalkanoates. Metab Eng 26:34–47. doi:10.1016/j.ymben.2014.09.001
Torregrosa-Crespo J et al (2016) Anaerobic metabolism in Haloferax genus: denitrification as case of study. Adv Microb Physiol 68:41–85. doi:10.1016/bs.ampbs.2016.02.001
Urtuvia V, Villegas P, González M, Seeger M (2014) Bacterial production of the biodegradable plastics polyhydroxyalkanoates. Int J Biol Macromol 70:208–213. doi:10.1016/j.ijbiomac.2014.06.001
Valappil SP, Rai R, Bucke C, Roy I (2008) Polyhydroxyalkanoate biosynthesis in Bacillus cereus SPV under varied limiting conditions and an insight into the biosynthetic genes involved. J Appl Microbiol 104:1624–1635. doi:10.1111/j.1365-2672.2007.03678.x
Wang Y, Yin J, Chen GQ (2014) Polyhydroxyalkanoates, challenges and opportunities. Curr Opin Biotechnol 30:59–65. doi:10.1016/j.copbio.2014.06.001
Yin J, Chen JC, Wu Q, Chen GQ (2015) Halophiles, coming stars for industrial biotechnology. Biotechnol Adv 33:1433–1442. doi:10.1016/j.biotechadv.2014.10.008
Acknowledgements
This work was funded by the Biotechnology and Biological Sciences Research Council (BBSRC) Grant BB/J014478/1.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by H. Atomi.
Rights and permissions
About this article
Cite this article
Ferre-Guell, A., Winterburn, J. Production of the copolymer poly(3-hydroxybutyrate-co-3-hydroxyvalerate) with varied composition using different nitrogen sources with Haloferax mediterranei . Extremophiles 21, 1037–1047 (2017). https://doi.org/10.1007/s00792-017-0964-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00792-017-0964-9