Abstract
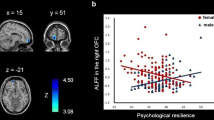
Psychological resilience reflects an individual’s ability to adapt and cope successfully in adverse environments and situations, making it a crucial trait in resisting stress-linked mental disorders and physical diseases. Although prior literature has consistently shown that males are more resilient than females, the sex-linked neuroanatomical correlates of psychological resilience are largely unknown. This study aims to explore the sex-specific relation between psychological resilience and brain gray matter volume (GMV) in adolescents via structural magnetic resonance imaging (s-MRI). A cohort of 231 healthy adolescents (121/110 females/males), aged 16 to 20 completed brain s-MRI scanning and Connor-Davidson Resilience Scale (CD-RISC) and other controlling behavioral tests. With s-MRI data, an optimized voxel-based morphometry method was used to estimate regional GMV, and a whole-brain condition-by-covariate interaction analysis was performed to identify the brain regions showing sex effects on the relation between psychological resilience and GMV. Male adolescents scored significantly higher than females on the CD-RISC. The association of psychological resilience with GMV differed between the two sex groups in the left ventrolateral prefrontal cortex extending to the adjacent anterior insula, with a positive correlation among males and a negative correlation among females. The sex-specific association between psychological resilience and GMV might be linked to sex differences in the hypothalamic–pituitary–adrenal axis and brain maturation during adolescence. This study may be novel in revealing the sex-linked neuroanatomical basis of psychological resilience, highlighting the need for a more thorough investigation of the role of sex in future studies of psychological resilience and stress-related illness.

Similar content being viewed by others
Data availability
The data and code that support the findings of present study are available from the corresponding author through reasonable request. The data and code sharing adopted by the authors comply with the requirements of the funding institute and with institutional ethics approval.
References
Fletcher D, Sarkar M (2013) Psychological resilience: a review and critique of definitions, concepts, and theory. Eur Psychol 18:12–23
Hu T, Zhang D, Wang J (2015) A meta-analysis of the trait resilience and mental health. Pers Individ Dif 76:18–27
Johnston MC, Porteous T, Crilly MA, Burton CD, Elliott A, Iversen L et al (2015) Physical disease and resilient outcomes: a systematic review of resilience definitions and study methods. Psychosomatics 56:168–180
Connor K, Zhang W (2006) Resilience: determinants, measurement, and treatment responsiveness. CNS Spectr 11:5–12
Kalin NH (2021) Trauma, resilience, anxiety disorders, and PTSD. Am J Psychiatry 178:103–105
Ghulam A, Bonaccio M, Costanzo S, Bracone F, Gianfagna F, de Gaetano G et al (2022) Psychological resilience, cardiovascular disease, and metabolic disturbances: a systematic review. Front Psychol 13:817298
Crump C, Sundquist J, Winkleby MA, Sundquist K (2016) Stress resilience and subsequent risk of type 2 diabetes in 1.5 million young men. Diabetologia 59:728–733
Wright LJ, Zautra AJ, Going S (2008) Adaptation to early knee osteoarthritis: the role of risk, resilience, and disease severity on pain and physical functioning. Ann Behav Med 36:70–80
Sturgeon JA, Zautra AJ (2010) Resilience: a new paradigm for adaptation to chronic pain. Curr Pain Headache Rep 14:105–112
Luthans F, Avolio BJ, Avey JB, Norman SM (2007) Positive psychological capital: measurement and relationship with performance and satisfaction. Pers Psychol 60:541–572
Xie X, Chen W, Lei L, Xing C, Zhang Y (2016) The relationship between personality types and prosocial behavior and aggression in Chinese adolescents. Pers Individ Dif 95:56–61
Kanai R, Rees G (2011) The structural basis of inter-individual differences in human behaviour and cognition. Nat Rev Neurosci 12:231–242
Gupta A, Love A, Kilpatrick LA, Labus JS, Bhatt R, Chang L et al (2017) Morphological brain measures of cortico-limbic inhibition related to resilience. J Neurosci Res 95:1760–1775
Kahl M, Wagner G, de la Cruz F, Köhler S, Schultz CC (2020) Resilience and cortical thickness: a MRI study. Eur Arch Psychiatry Clin Neurosci 270:533–539
Shikimoto R, Noda Y, Kida H, Nakajima S, Tsugawa S, Mimura Y et al (2021) Association between resilience and cortical thickness in the posterior cingulate cortex and the temporal pole in Japanese older people: a population-based cross-sectional study. J Psychiatr Res 142:89–100
Liu X, Lai H, Li J, Becker B, Zhao Y, Cheng B et al (2021) Gray matter structures associated with neuroticism: a meta-analysis of whole-brain voxel-based morphometry studies. Hum Brain Mapp 42:2706–2721
Hobfoll SE, Palmieri PA, Johnson RJ, Canetti-Nisim D, Hall BJ, Galea S (2009) Trajectories of resilience, resistance, and distress during ongoing terrorism: the case of Jews and Arabs in Israel. J Consult Clin Psychol 77:138–148
Bonanno GA, Galea S, Bucciarelli A, Vlahov D (2007) What predicts psychological resilience after disaster? the role of demographics, resources, and life stress. J Consult Clin Psychol 75:671–682
Stratta P, Capanna C, Patriarca S, de Cataldo S, Bonanni RL, Riccardi I et al (2013) Resilience in adolescence: Gender differences two years after the earthquake of L’Aquila. Pers Individ Dif 54:327–331
Bonanno GA, Ho SMY, Chan JCK, Kwong RSY, Cheung CKY, Wong CPY et al (2008) Psychological resilience and dysfunction among hospitalized survivors of the SARS epidemic in Hong Kong: a latent class approach. Heal Psychol 27:659–667
Rodriguez-Llanes JM, Vos F, Guha-Sapir D (2013) Measuring psychological resilience to disasters: are evidence-based indicators an achievable goal? Environ Health 12:115
Portzky M, Wagnild G, De Bacquer D, Audenaert K (2010) Psychometric evaluation of the dutch resilience scale RS-nl on 3265 healthy participants: a confirmation of the association between age and resilience found with the Swedish version. Scand J Caring Sci 24(Suppl 1):86–92
Skrove M, Romundstad P, Indredavik MS (2013) Resilience, lifestyle and symptoms of anxiety and depression in adolescence: the young-HUNT study. Soc Psychiatry Psychiatr Epidemiol 48:407–416
Bale TL, Epperson CN (2015) Sex differences and stress across the lifespan. Nat Neurosci 18:1413–1420
Hirani S, Lasiuk G, Hegadoren K (2016) The intersection of gender and resilience. J Psychiatr Ment Health Nurs 23:455–467
Oldehinkel AJ, Bouma EMC (2011) Sensitivity to the depressogenic effect of stress and HPA-axis reactivity in adolescence: a review of gender differences. Neurosci Biobehav Rev 35:1757–1770
Kilpatrick LA, Istrin JJ, Gupta A, Naliboff BD, Tillisch K, Labus JS et al (2015) Sex commonalities and differences in the relationship between resilient personality and the intrinsic connectivity of the salience and default mode networks. Biol Psychol 112:107–115
Wang S, Yang C, Zhao Y, Lai H, Zhang L, Gong Q (2019) Sex-linked neurofunctional basis of psychological resilience in late adolescence: a resting-state functional magnetic resonance imaging study. Eur Child Adolesc Psychiatry 29:1075–1087
Ashburner J (2007) A fast diffeomorphic image registration algorithm. Neuroimage 38:95–113
Pan N, Wang S, Zhao Y, Lai H, Qin K, Li J et al (2021) Brain gray matter structures associated with trait impulsivity: a systematic review and voxel-based meta-analysis. Hum Brain Mapp 42:2214–2235
Wang S, Dai J, Li J, Wang X, Chen T, Yang X et al (2018) Neuroanatomical correlates of grit: Growth mindset mediates the association between gray matter structure and trait grit in late adolescence. Hum Brain Mapp 39:1688–1699
Lai H, Kong X, Zhao Y, Pan N, Zhang X, He M et al (2022) Patterns of a structural covariance network associated with dispositional optimism during late adolescence. Neuroimage 251:119009
Oldfield RC (1971) The assessment and analysis of handedness: the Edinburgh Inventory. Neuropsychologia 9:97–113
Campbell-Sills L, Stein MB (2007) Psychometric analysis and refinement of the connor-davidson resilience scale (CD-RISC): validation of a 10-item measure of resilience. J Trauma Stress 20:1019–1028
Cheng C, Dong D, He J, Zhong X, Yao S (2020) Psychometric properties of the 10-item connor-davidson resilience scale (CD-RISC-10) in Chinese undergraduates and depressive patients. J Affect Disord 261:211–220
Kong F, Ma X, You X, Xiang Y (2018) The resilient brain: psychological resilience mediates the effect of amplitude of low-frequency fluctuations in orbitofrontal cortex on subjective well-being in young healthy adults. Soc Cogn Affect Neurosci 13:755–763
She R, Yang X, Lau MMC, Lau JTF (2020) Psychometric properties and normative data of the 10-item connor-davidson resilience scale among Chinese adolescent students in Hong Kong. Child Psychiatry Hum Dev 51:925–933
Lai H, Wang S, Zhao Y, Qiu C, Gong Q (2020) Neurostructural correlates of optimism: Gray matter density in the putamen predicts dispositional optimism in late adolescence. Hum Brain Mapp 41:1459–1471
Raven J (2000) The Raven’s progressive matrices: change and stability over culture and time. Cogn Psychol 41:1–48
Oshio A, Taku K, Hirano M, Saeed G (2018) Resilience and Big Five personality traits: a meta-analysis. Pers Individ Dif 127:54–60
DeYoung CG, Hirsh JB, Shane MS, Papademetris X, Rajeevan N, Gray JR (2010) Testing predictions from personality neuroscience. Brain structure and the big five. Psychol Sci 21:820–828
Zhang L, fang. (2011) Hardiness and the big five personality traits among chinese university students. Learn Individ Differ 21:109–113
Zhang M, Han J, Shi J, Ding H, Wang K, Kang C et al (2018) Personality traits as possible mediators in the relationship between childhood trauma and depressive symptoms in Chinese adolescents. J Psychiatr Res 103:150–155
Hayasaka S, Phan KL, Liberzon I, Worsley KJ, Nichols TE (2004) Nonstationary cluster-size inference with random field and permutation methods. Neuroimage 22:676–687
Silver M, Montana G, Nichols TE (2011) False positives in neuroimaging genetics using voxel-based morphometry data. Neuroimage 54:992–1000
Takeuchi H, Taki Y, Sekiguchi A, Nouchi R, Kotozaki Y, Nakagawa S et al (2015) Brain structures in the sciences and humanities. Brain Struct Funct 220:3295–3305
Cohen J (1988) Statistical power analysis for the behavioral sciences. Routledge, New York
Kudielka BM, Kirschbaum C (2005) Sex differences in HPA axis responses to stress: a review. Biol Psychol 69:113–132
Tamres L, Janicki D, Helgeson VS (2010) Sex differences in coping behavior: a meta-analytic review and an examination of relative coping. Personal Soc Psychol Rev 6:2–30
Bangasser DA, Valentino RJ (2014) Sex differences in stress-related psychiatric disorders: neurobiological perspectives. Front Neuroendocrinol 35:303–319
Hodes GE, Epperson CN (2019) Sex differences in vulnerability and resilience to stress across the life span. Biol Psychiatry 86:421–432
Kelly MM, Tyrka AR, Price LH, Carpenter LL (2008) Sex differences in the use of coping strategies: predictors of anxiety and depressive symptoms. Depress Anxiety 25:839–846
Friedman NP, Robbins TW (2022) The role of prefrontal cortex in cognitive control and executive function. Neuropsychopharmacol 47:72–89
Ochsner KN, Silvers JA, Buhle JT (2012) Functional imaging studies of emotion regulation: a synthetic review and evolving model of the cognitive control of emotion. Ann N Y Acad Sci 1251:E1–E24
Price JL, Drevets WC (2010) Neurocircuitry of mood disorders. Neuropsychopharmacol 35:192–216
Salvadore G, Nugent AC, Lemaitre H, Luckenbaugh DA, Tinsley R, Cannon DM et al (2011) Prefrontal cortical abnormalities in currently depressed versus currently remitted patients with major depressive disorder. Neuroimage 54:2643–2651
Strawn JR, Hamm L, Fitzgerald DA, Fitzgerald KD, Monk CS, Phan KL (2015) Neurostructural abnormalities in pediatric anxiety disorders. J Anxiety Disord 32:81–88
Pompei F, Dima D, Rubia K, Kumari V, Frangou S (2011) Dissociable functional connectivity changes during the Stroop task relating to risk, resilience and disease expression in bipolar disorder. Neuroimage 57:576–582
Asami T, Yamasue H, Hayano F, Nakamura M, Uehara K, Otsuka T et al (2009) Sexually dimorphic gray matter volume reduction in patients with panic disorder. Psychiatry Res 173:128–134
Vijayakumar N, Whittle S, Yücel M, Dennison M, Simmons J, Allen NB (2014) Thinning of the lateral prefrontal cortex during adolescence predicts emotion regulation in females. Soc Cogn Affect Neurosci 9:1845–1854
Colich NL, Williams ES, Ho TC, King LS, Humphreys KL, Price AN et al (2017) The association between early life stress and prefrontal cortex activation during implicit emotion regulation is moderated by sex in early adolescence. Dev Psychopathol 29:1851–1864
Carrion VG, Weems CF, Richert K, Hoffman BC, Reiss AL (2010) Decreased prefrontal cortical volume associated with increased bedtime cortisol in traumatized youth. Biol Psychiatry 68:491–493
Kern S, Oakes TR, Stone CK, McAuliff EM, Kirschbaum C, Davidson RJ (2008) Glucose metabolic changes in the prefrontal cortex are associated with HPA axis response to a psychosocial stressor. Psychoneuroendocrinology 33:517–529
Goel N, Workman J, Lee T, Innala L (2014) Physiol VV-C, 2014 U. sex differences in the HPA axis. Compr Physiol 4:1121–1155
Cahill L (2006) Why sex matters for neuroscience. Nat Rev Neurosci 7:477–484
Shansky RM, Glavis-Bloom C, Lerman D, McRae P, Benson C, Miller K et al (2004) Estrogen mediates sex differences in stress-induced prefrontal cortex dysfunction. Mol Psychiatry 9:531–538
du Plessis MR, Smeekens S, Cillessen AHN, Whittle S, Güroǧlu B (2018) Bullying the brain? longitudinal links between childhood peer victimization, cortisol, and adolescent brain structure. Front Psychol 9:2706
Herting MM, Gautam P, Spielberg JM, Kan E, Dahl RE, Sowell ER (2014) The role of testosterone and estradiol in brain volume changes across adolescence: a longitudinal structural MRI study. Hum Brain Mapp 35:5633–5645
Kaczkurkin AN, Raznahan A, Satterthwaite TD (2019) Sex differences in the developing brain: insights from multimodal neuroimaging. Neuropsychopharmacology 44:71–85
Group BDC (2012) Total and regional brain volumes in a population-based normative sample from 4 to 18 years: the NIH MRI study of normal brain development. Cereb Cortex 22:1–12
Gennatas ED, Avants BB, Wolf DH, Satterthwaite TD, Ruparel K, Ciric R et al (2017) Age-related effects and sex differences in gray matter density, volume, mass, and cortical thickness from childhood to young adulthood. J Neurosci 37:5065–5073
Menon V (2011) Large-scale brain networks and psychopathology: a unifying triple network model. Trends Cogn Sci 15:483–506
Kong F, Wang X, Hu S, Liu J (2015) Neural correlates of psychological resilience and their relation to life satisfaction in a sample of healthy young adults. Neuroimage 123:165–172
Sridharan D, Levitin DJ, Menon V (2008) A critical role for the right fronto-insular cortex in switching between central-executive and default-mode networks. Proc Natl Acad Sci U S A 105:12569–12574
Holt-Gosselin B, Tozzi L, Ramirez CA, Gotlib IH, Williams LM (2021) Coping strategies, neural structure, and depression and anxiety during the COVID-19 pandemic: a longitudinal study in a naturalistic sample spanning clinical diagnoses and subclinical symptoms. Biol Psychiatry Glob Open Sci 1:261–271
Lyoo IK, Kim MJ, Stoll AL, Demopulos CM, Parow AM, Dager SR et al (2004) Frontal lobe gray matter density decreases in bipolar I disorder. Biol Psychiatry 55:648–651
Shi L, Ren Z, Qiu J (2021) High thought control ability, high resilience: the effect of temporal cortex and insula connectivity. Neuroscience 472:60–67
Yan X, Brown AD, Lazar M, Cressman VL, Henn-Haase C, Neylan TC et al (2013) Spontaneous brain activity in combat related PTSD. Neurosci Lett 547:1–5
Lui S, Huang X, Chen L, Tang H, Zhang T, Li X et al (2009) High-field MRI reveals an acute impact on brain function in survivors of the magnitude 8.0 earthquake in China. Proc Natl Acad Sci U S A 106:15412–15417
Waugh CE, Wager TD, Fredrickson BL, Noll DC, Taylor SF (2008) The neural correlates of trait resilience when anticipating and recovering from threat. Soc Cogn Affect Neurosci 3:322–332
Shao R, Lau WKW, Leung MK, Lee TMC (2018) Subgenual anterior cingulate-insula resting-state connectivity as a neural correlate to trait and state stress resilience. Brain Cogn 124:73–81
Tanner JJ, Johnson AJ, Terry EL, Cardoso J, Garvan C, Staud R et al (2021) Resilience, pain, and the brain: relationships differ by sociodemographics. J Neurosci Res 99:1207–1235
Helpman L, Zhu X, Suarez-Jimenez B, Lazarov A, Monk C, Neria Y (2017) Sex differences in trauma-related psychopathology: a critical review of neuroimaging literature (2014–2017). Curr Psychiatry Rep 19:1–13
Lawson GM, Camins JS, Wisse L, Wu J, Duda JT, Cook PA et al (2017) Childhood socioeconomic status and childhood maltreatment: distinct associations with brain structure. PLoS ONE 12:e0175690
Sublette ME, Galfalvy HC, Oquendo MA, Bart CP, Schneck N, Arango V et al (2016) Relationship of recent stress to amygdala volume in depressed and healthy adults. J Affect Disord 203:136–142
Lissek S, Kaczkurkin AN, Rabin S, Geraci M, Pine DS, Grillon C (2014) Generalized anxiety disorder is associated with overgeneralization of classically conditioned fear. Biol Psychiatry 75:909–915
Lissek S, Grillon C (2012) Learning models of PTSD. Oxford Handbook, Tramatic Disord., pp 175–190
Günther V, Jahn S, Webelhorst C, Bodenschatz CM, Bujanow A, Mucha S et al (2022) Coping with anxiety: brain structural correlates of vigilance and cognitive avoidance. Front Psychiatry 13:1–8
Krohne HW, Egloff B, Varner LJ, Burns LR, Weidner G, Ellis HC (2000) The assessment of dispositional vigilance and cognitive avoidance: factorial structure, psychometric properties, and validity of the mainz coping inventory. Cognit Ther Res 24:297–311
Oswald LM, Wand GS, Kuwabara H, Wong DF, Zhu S, Brasic JR (2014) History of childhood adversity is positively associated with ventral striatal dopamine responses to amphetamine. Psychopharmacology 231:2417–2433
Wu Y, Li H, Zhou Y, Yu J, Zhang Y, Song M et al (2016) Sex-specific neural circuits of emotion regulation in the centromedial amygdala. Sci Rep 6:23112
Wang S, Zhao Y, Li J (2023) True grit and brain: Trait grit mediates the connection of DLPFC functional connectivity density to posttraumatic growth following COVID-19. J Affect Disord 325:313–320
Lin J, Li L, Pan N, Liu X, Zhang X, Suo X et al (2023) Neural correlates of neuroticism: a coordinate-based meta-analysis of resting-state functional brain imaging studies. Neurosci Biobehav R 146:105055
Mackey AP, Finn AS, Leonard JA, Jacoby-Senghor DS, West MR, Gabrieli CFO et al (2015) Neuroanatomical correlates of the income-achievement gap. Psychol Sci 26:925–933
Funding
This study was supported by the National Natural Science Foundation of China (Grant Nos. 31800963, 81621003, 81761128023, 81820108018 and 82027808), the National Key R&D Program of China (2022YFC2009900), and the Key Research and Development Program of Sichuan Province (Grant No. 2023YFS0084). The funding sources had no involvement in the study design, data collection and analysis, results interpretation, or writing of the paper.
Author information
Authors and Affiliations
Contributions
SW and QG designed the study and supervised the conduct of the study. NP, CY, XS, and SW contributed to the data collection. NP and CY performed the data analysis and results interpretation. NP and SW drafted the manuscript. AS and SH provided interpretive advice and critically revised the manuscript, which all authors reviewed and approved for publication.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethical approval
The authors assert that all procedures contributing to this work comply with the ethical standards of the relevant national and institutional committees on human experimentation and with the Helsinki Declaration of 1975, as revised in 2008.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Pan, N., Yang, C., Suo, X. et al. Sex differences in the relationship between brain gray matter volume and psychological resilience in late adolescence. Eur Child Adolesc Psychiatry 33, 1057–1066 (2024). https://doi.org/10.1007/s00787-023-02231-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00787-023-02231-7