Abstract
Objectives
The aims of this study were to compare the in vitro cytokine response of gingival fibroblasts (GF’s) from healthy and inflamed human gingival tissues and to assess whether GF’s from inflamed gingivae are capable of mounting a secondary inflammatory response after exposure to P. gingivalis LPS.
Materials and methods
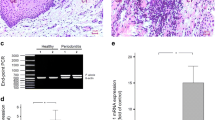
GF’s were obtained from healthy donors and periodontitis patients and cultured in vitro. Cells were exposed to P. gingivalis LPS for 24h before measurement of MCP-1, GRO, IL-6, IL-8 and VEGF using a bead-based multiplex assay. Statistical comparisons were made between LPS-exposed GF’s and unstimulated cells as well as the two patient groups by two-way ANOVA.
Results
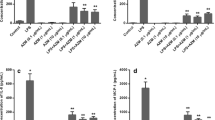
GF’s exposed to P. gingivalis LPS significantly increased their production of MCP-1, GRO, IL-6, IL-8 and VEGF compared to unstimulated cells. GF’s isolated from inflamed tissue from periodontitis patients demonstrated consistently less cytokine production after exposure to P. gingivalis LPS, most notably for GRO and IL-6.
Conclusions
The current study demonstrates that GF’s play an active role in the inflammatory response in periodontal disease by producing a number of chemokines and cytokines. Furthermore, inflamed GF’s may be compromised in their ability to mount an adequate secondary immune response in relation to chemokine/cytokine production.
Clinical relevance
The compromised inflammatory cytokine response of inflamed human gingival fibroblasts to P. gingivalis LPS may impact on their ability to recruit and activate inflammatory cells while maintaining persistent inflammation, a key feature of periodontal disease.


Similar content being viewed by others
References
Socransky SS, Haffajee AD, Cugini MA, Smith C, Kent RL Jr (1998) Microbial complexes in subgingival plaque. J Clin Periodontol 25:134–144
Takeuchi Y, Umeda M, Sakamoto M, Benno Y, Huang Y, Ishikawa I (2001) Treponema socranskii, Treponema denticola, and Porphyromonas gingivalis are associated with severity of periodontal tissue destruction. J Periodontol 72:1354–1363
Fitzsimmons TR, Sanders AE, Slade GD, Bartold PM (2009) Biomarkers of periodontal inflammation in the Australian adult population. Aust Dent J 54:115–122
D'Aiuto F, Parkar M, Andreou G, Suvan J, Brett PM, Ready D, Tonetti MS (2004) Periodontitis and systemic inflammation: control of the local infection is associated with a reduction in serum inflammatory markers. J Dent Res 83:156–160
Noack B, Genco RJ, Trevisan M, Grossi S, Zambon JJ, De Nardin E (2001) Periodontal infections contribute to elevated systemic C-reactive protein level. J Periodontol 72:1221–1227
Moutsopoulos NM, Madianos PN (2006) Low-grade inflammation in chronic infectious diseases: paradigm of periodontal infections. Ann N Y Acad Sci 1088:251–264
Bartold PM, Millar SJ (1988) Effect of lipopolysaccharide on proteoglycan synthesis by adult human gingival fibroblasts in vitro. Infect Immun 56:2149–2155
Wilson M (1995) Biological activities of lipopolysaccharides from oral bacteria and their relevance to the pathogenesis of chronic periodontitis. Sci Prog 78(Pt 1):19–34
Wang PL, Azuma Y, Shinohara M, Ohura K (2000) Toll-like receptor 4-mediated signal pathway induced by Porphyromonas gingivalis lipopolysaccharide in human gingival fibroblasts. Biochem Biophys Res Commun 273:1161–1167
Hatakeyama J, Tamai R, Sugiyama A, Akashi S, Sugawara S, Takada H (2003) Contrasting responses of human gingival and periodontal ligament fibroblasts to bacterial cell-surface components through the CD14/toll-like receptor system. Oral Microbiol Immunol 18:14–23
Wang PL, Sato K, Oido M, Fujii T, Kowashi Y, Shinohara M, Ohura K, Tani H, Kuboki Y (1998) Involvement of CD14 on human gingival fibroblasts in Porphyromonas gingivalis lipopolysaccharide-mediated interleukin-6 secretion. Arch Oral Biol 43:687–694
Wright SD, Ramos RA, Tobias PS, Ulevitch RJ, Mathison JC (1990) CD14, a receptor for complexes of lipopolysaccharide (LPS) and LPS binding protein. Science 249:1431–1433
Almasri A, Wisithphrom K, Windsor LJ, Olson B (2007) Nicotine and lipopolysaccharide affect cytokine expression from gingival fibroblasts. J Periodontol 78:533–541
Yamazaki K, Ikarashi F, Aoyagi T, Takahashi K, Nakajima T, Hara K, Seymour GJ (1992) Direct and indirect effects of Porphyromonas gingivalis lipopolysaccharide on interleukin-6 production by human gingival fibroblasts. Oral Microbiol Immunol 7:218–224
Herath TD, Wang Y, Seneviratne CJ, Darveau RP, Wang CY, Jin L (2013) The expression and regulation of matrix metalloproteinase-3 is critically modulated by Porphyromonas gingivalis lipopolysaccharide with heterogeneous lipid A structures in human gingival fibroblasts. BMC Microbiol 13:73
Jonsson D, Amisten S, Bratthall G, Holm A, Nilsson BO (2009) LPS induces GROalpha chemokine production via NF-kappaB in oral fibroblasts. Inflamm Res 58:791–796
Morandini AC, Sipert CR, Gasparoto TH, Greghi SL, Passanezi E, Rezende ML, Sant'ana AP, Campanelli AP, Garlet GP, Santos CF (2010) Differential production of macrophage inflammatory protein-1alpha, stromal-derived factor-1, and IL-6 by human cultured periodontal ligament and gingival fibroblasts challenged with lipopolysaccharide from P. gingivalis. J Periodontol 81:310–317
Belibasakis GN, Bostanci N, Hashim A, Johansson A, Aduse-Opoku J, Curtis MA, Hughes FJ (2007) Regulation of RANKL and OPG gene expression in human gingival fibroblasts and periodontal ligament cells by Porphyromonas gingivalis: a putative role of the Arg-gingipains. Microb Pathog 43:46–53
El-Awady AR, Messer RL, Gamal AY, Sharawy MM, Wenger KH, Lapp CA (2010) Periodontal ligament fibroblasts sustain destructive immune modulators of chronic periodontitis. J Periodontol 81:1324–1335
Sun Y, Li H, Sun MJ, Zheng YY, Gong DJ, Xu Y (2014) Endotoxin tolerance induced by lipopolysaccharides derived from Porphyromonas gingivalis and Escherichia coli: alternations in toll-like receptor 2 and 4 signaling pathway. Inflammation 37:268–276
Ara T, Kurata K, Hirai K, Uchihashi T, Uematsu T, Imamura Y, Furusawa K, Kurihara S, Wang PL (2009) Human gingival fibroblasts are critical in sustaining inflammation in periodontal disease. J Periodontal Res 44:21–27
Ogawa T, Ozaki A, Shimauchi H, Uchida H (1997) Hyporesponsiveness of inflamed human gingival fibroblasts from patients with chronic periodontal diseases against cell surface components of Porphyromonas gingivalis. FEMS Immunol Med Microbiol 18:17–30
Pena OM, Pistolic J, Raj D, Fjell CD, Hancock RE (2011) Endotoxin tolerance represents a distinctive state of alternative polarization (M2) in human mononuclear cells. J Immunol 186:7243–7254
Page RC, Eke PI (2007) Case definitions for use in population-based surveillance of periodontitis. J Periodontol 78:1387–1399
Kang W, Hu Z, Ge S (2016) Healthy and inflamed gingival fibroblasts differ in their inflammatory response to Porphyromonas gingivalis lipopolysaccharide. Inflammation 39:1842–1852
Wada N, Wang B, Lin NH, Laslett AL, Gronthos S, Bartold PM (2011) Induced pluripotent stem cell lines derived from human gingival fibroblasts and periodontal ligament fibroblasts. J Periodontal Res 46:438–447
Steffen MJ, Holt SC, Ebersole JL (2000) Porphyromonas gingivalis induction of mediator and cytokine secretion by human gingival fibroblasts. Oral Microbiol Immunol 15:172–180
Doyle CJ, Fitzsimmons TR, Marchant C, Dharmapatni AA, Hirsch R, Bartold PM (2014) Azithromycin suppresses P. gingivalis LPS-induced pro-inflammatory cytokine and chemokine production by human gingival fibroblasts in vitro. Clin Oral Investig 19(2):221–227
Suthin K, Matsushita K, Machigashira M, Tatsuyama S, Imamura T, Torii M, Izumi Y (2003) Enhanced expression of vascular endothelial growth factor by periodontal pathogens in gingival fibroblasts. J Periodontal Res 38:90–96
Yu X, Graves DT (1995) Fibroblasts, mononuclear phagocytes, and endothelial cells express monocyte chemoattractant protein-1 (MCP-1) in inflamed human gingiva. J Periodontol 66:80–88
Bartold PM, Haynes DR (1991) Interleukin-6 production by human gingival fibroblasts. J Periodontal Res 26:339–345
Pradeep AR, Daisy H, Hadge P (2009) Serum levels of monocyte chemoattractant protein-1 in periodontal health and disease. Cytokine 47:77–81
Giannopoulou C, Kamma JJ, Mombelli A (2003) Effect of inflammation, smoking and stress on gingival crevicular fluid cytokine level. J Clin Periodontol 30:145–153
Teles R, Sakellari D, Teles F, Konstantinidis A, Kent R, Socransky S, Haffajee A (2009) Relationships among gingival crevicular fluid biomarkers, clinical parameters of periodontal disease, and the subgingival microbiota. J Periodontol 81:89–98
Scheres N, Crielaard W (2013) Gingival fibroblast responsiveness is differentially affected by Porphyromonas gingivalis: implications for the pathogenesis of periodontitis. Mol Oral Microbiol 28:204–218
Bozkurt SB, Hakki SS, Hakki EE, Durak Y, Kantarci A (2017) Porphyromonas gingivalis lipopolysaccharide induces a pro-inflammatory human gingival fibroblast phenotype. Inflammation 40:144–153
Zaric S, Shelburne C, Darveau R, Quinn DJ, Weldon S, Taggart CC, Coulter WA (2010) Impaired immune tolerance to Porphyromonas gingivalis lipopolysaccharide promotes neutrophil migration and decreased apoptosis. Infect Immun 78:4151–4156
Pap T, Müller-Ladner U, Gay RE, Gay S (2000) Fibroblast biology: role of synovial fibroblasts in the pathogenesis of rheumatoid arthritis. Arthritis Research & Therapy 2:361
Brouty-Boyé D, Pottin-Clémenceau C, Doucet C, Jasmin C, Azzarone B (2000) Chemokines and CD40 expression in human fibroblasts. Eur J Immunol 30:914–919
Slany A, Meshcheryakova A, Beer A, Ankersmit HJ, Paulitschke V, Gerner C (2014) Plasticity of fibroblasts demonstrated by tissue-specific and function-related proteome profiling. Clin Proteomics 11:41
Jang JY, Song IS, Baek KJ, Choi Y, Ji S (2016) Immunologic characteristics of human gingival fibroblasts in response to oral bacteria. J Periodontal Res 52:447–457
Stathopoulou PG, Benakanakere MR, Galicia JC, Kinane DF (2009) The host cytokine response to Porphyromonas gingivalis is modified by gingipains. Oral Microbiol Immunol 24:11–17
Muthukuru M, Jotwani R, Cutler CW (2005) Oral mucosal endotoxin tolerance induction in chronic periodontitis. Infect Immun 73:687–694
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Funding
This study was supported with internal funding from the University of Adelaide.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Fitzsimmons, T.R., Ge, S. & Bartold, P.M. Compromised inflammatory cytokine response to P. gingivalis LPS by fibroblasts from inflamed human gingiva. Clin Oral Invest 22, 919–927 (2018). https://doi.org/10.1007/s00784-017-2171-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-017-2171-6