Abstract
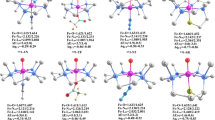
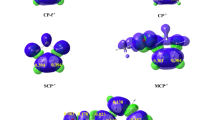
The geometric structures and mechanisms for hydrogen abstraction from cyclohexane for four high-valent complexes, [FeIV(O)(TMC)(NCMe)]2+ (where TMC is 1,4,8,11-tetramethyl-1,4,8,11-tetraazacyclotetradecane; 1-NCMe), the inverted isomer [FeIV(NCMe)(TMC)(O)]2+ (2-NCMe), [RuIV(O)(TMC)(NCMe)]2+ (the ruthenium analogue of 1-NCMe; 3-NCMe), and the inverted isomer [RuIV(NCMe)(TMC)(O)]2+ (4-NCMe), were investigated using density functional theory. The axial NCMe ligand was found to be sterically more hindered in 2-NCMe than in 1-NCMe, which is in accord with the calculated results that the Fe–Laxial distance is longer in the former. Both 1-NCMe and 2-NCMe are capable of hydrogen abstraction from cyclohexane via two-state reactivity patterns. In contrast, 3-NCMe and 4-NCMe react with cyclohexane by a single-state mechanism. The reaction pathways computed reveal that 2-NCMe is more reactive than 1-NCMe, in agreement with experimental results, whereas the reactivity of 3-NCMe and 4-NCMe shows little dependence on whether the oxo unit is syn or anti to the four N-methyl groups. Our analysis shows that along the reaction pathway for 2-NCMe in the triplet spin state, the NCMe ligand moves away from the iron center, and therefore the energy of the \( \sigma_{z}^{* 2} \) (α-spin) orbital decreases and an electron is transferred to this orbital. Finally, we calculated the kinetic isotope effect and investigated the relationship between this effect and reaction barriers.







Similar content being viewed by others
References
Que L Jr, Ho RYN (1996) Chem Rev 96:2607–2624
Costas M, Mehn MP, Jensen MP, Que L Jr (2004) Chem Rev 104:939–986
Solomon EI, Brunold TC, Davis MI, Kemsley JN, Lee S-K, Lehnert N, Neese F, Skulan AJ, Yang Y-S, Zhou J (2000) Chem Rev 100:235–350
Bernasconi L, Louwerse MJ, Baerends EJ (2007) Eur J Inorg Chem 3023–3033
Shan XP, Que L Jr (2006) J Inorg Biochem 100:421–433
Que L Jr (2007) Acc Chem Res 40:493–500
Rohde J-U, In J-H, Lim MH, Brennessel WW, Bukowski MR, Stubna A, Münck E, Nam W, Que L Jr (2003) Science 299:1037–1039
Lim MH, Rohde J-U, Stubna A, Bukowski MR, Costas M, Ho RYN, Münck E, Nam W, Que L Jr (2003) Proc Natl Acad Sci USA 100:3665–3670
Balland V, Charlot M-F, Banse F, Girerd J-J, Mattioli TA, Bill E, Bartoli J-F, Battioni P, Mansuy D (2004) Eur J Inorg Chem 301–308
Klinker EJ, Kaizer J, Brennessel WW, Woodrum NL, Cramer CJ, Que L Jr (2005) Angew Chem Int Ed 44:3690–3694
Grapperhaus CA, Mienert B, Bill E, Weyhermüller T, Wieghardt K (2000) Inorg Chem 39:5306–5317
Martinho M, Banse F, Bartoli J-F, Mattioli TA, Battioni P, Horner O, Bourcier S, Girerd J-J (2005) Inorg Chem 44:9592–9596
Bukowski MR, Koehntop KD, Stubna A, Bominaar EL, Halfen JA, Münck E, Nam W, Que L Jr (2005) Science 310:1000–1002
Ray K, England J, Fiedler AT, Martinho M, Münck E, Que L Jr (2008) Angew Chem Int Ed 47:8068–8071
Nam W (2007) Acc Chem Res 40:522–531
Jackson TA, Rohde J-U, Seo MS, Sastri CV, DeHont R, Stubna A, Ohta T, Kitagawa T, Münck E, Nam W, Que L Jr (2008) J Am Chem Soc 130:12394–12407
Sastri CV, Park MJ, Ohta T, Jackson TA, Stubna A, Seo MS, Lee J, Kim J, Kitagawa T, Münck E, Que L Jr, Nam W (2005) J Am Chem Soc 127:12494–12495
Hirao H, Kumar D, Que L Jr, Shaik S (2006) J Am Chem Soc 128:8590–8606
Decker A, Rohde J-U, Klinker EJ, Wong SD, Que L Jr, Solomon EI (2007) J Am Chem Soc 129:15983–15996
Decker A, Clay MD, Solomon EI (2006) J Inorg Biochem 100:697–706
Decker A, Rohde J-U, Que L Jr, Solomon EI (2004) J Am Chem Soc 126:5378–5379
Decker A, Solomon EI (2005) Angew Chem Int Ed 44:2252–2255
Decker A, Solomon EI (2005) Curr Opin Chem Biol 9:152–163
Wang Y, Wang Y, Han K-L (2008) Chem J Chin Univ 2469–2473
Hirao H, Que L Jr, Nam W, Shaik S (2008) Chem Eur J 14:1740–1756
Sastri CV, Lee J, Oh K, Lee YJ, Lee J, Jackson TA, Ray K, Hirao H, Shin W, Halfen JA, Kim J, Que L Jr, Shaik S, Nam W (2007) Proc Natl Acad Sci USA 104:19181–19186
Rohde J-U, Que L Jr (2005) Angew Chem Int Ed 44:2255–2258
Dhuri SN, Seo MS, Lee Y-M, Hirao H, Wang Y, Nam W, Shaik S (2008) Angew Chem Int Ed 47:3356–3359
Che C-M, Wong K-Y, Mak TCW (1985) J Chem Soc Chem Commun 546–548
Sharma PK, de Visser SP, Ogliaro F, Shaik S (2003) J Am Chem Soc 125:2291–2300
Ogliaro F, de Visser SP, Groves JT, Shaik S (2001) Angew Chem Int Ed 40:2874–2878
Gross Z, Ini S (1999) Inorg Chem 38:1446–1449
Groves JT, Roman JS (1995) J Am Chem Soc 117:5594–5595
Groves JT, Quinn R (1985) J Am Chem Soc 107:5790–5792
Frisch MJ et al (2004) Gaussian 03, revision D.01. Gaussian, Wallingford
Becke AD (1992) J Chem Phys 96:2155–2160
Becke AD (1992) J Chem Phys 97:9173–9177
Becke AD (1993) J Chem Phys 98:5648–5652
Lee C, Yang W, Parr RG (1988) Phys Rev B 37:785–789
Hay JP, Wadt WR (1985) J Chem Phys 82:299–310
Friesner RA, Murphy RB, Beachy MD, Ringnalda MN, Pollard WT, Dunietz BD, Cao YX (1999) J Phys Chem A 103:1913–1928
de Visser SP (2006) J Am Chem Soc 128:9813–9824
de Visser SP (2006) J Am Chem Soc 128:15809–15818
Wang Y, Wang Y, Han K-L (2009) J Biol Inorg Chem 14:533–545
Scott AP, Radom L (1996) J Phys Chem 100:16502–16513
Melander L, Saunders WH Jr (1987) In: Reaction rates of isotopic molecules, chap 2. Krieger, Malabar
Handy NC, Cohen AJ (2001) Mol Phys 99:403–412
Perdew JP, Burke K, Ernzerhof M (1996) Phys Rev Lett 77:3865–3868
Perdew JP, Burke K, Ernzerhof M (1997) Phys Rev Lett 78:1396
Michel C, Baerends EJ (2009) Inorg Chem 48:3628–3638
Neese F (2006) J Inorg Biochem 100:716–726
SchÖneboom JC, Neese F, Thiel W (2005) J Am Chem Soc 127:5840–5853
Shaik S, Hirao H, Kumar D (2007) Acc Chem Res 40:532–542
Kumar D, Hirao H, Que L Jr, Shaik S (2005) J Am Chem Soc 127:8026–8027
Shaik S, Danovich D, Fiedler A, Schröder D, Schwarz H (1995) Helv Chem Acta 78:1393–1407
Schröder D, Shaik S, Schwarz H (2000) Acc Chem Res 33:139–145
Shaik S, Kumar D, de Visser SP (2008) J Am Chem Soc 130:10128–10140
Sharma PK, de Visser SP, Shaik S (2003) J Am Chem Soc 125:8698–8699
Godfrey E, Porro CS, de Visser SP (2008) J Phys Chem A 112:2464–2468
Aluri S, de Visser SP (2007) J Am Chem Soc 129:14846–14847
de Visser SP, Oh K, Han A-R, Nam W (2007) Inorg Chem 46:4632–4641
de Visser SP, Nam W (2008) J Phys Chem A 112:12887–12895
Acknowledgments
This work was supported by NKBRSF (2007CB815202) and NSFC (20833008).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wang, Y., Han, K. Steric hindrance effect of the equatorial ligand on Fe(IV)O and Ru(IV)O complexes: a density functional study. J Biol Inorg Chem 15, 351–359 (2010). https://doi.org/10.1007/s00775-009-0607-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00775-009-0607-4