Abstract
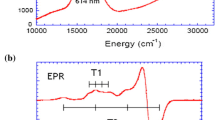
The copper content of recombinant CotA laccase from Bacillus subtilis produced by Escherichia coli cells is shown to be strongly dependent on the presence of copper and oxygen in the culture media. In copper-supplemented media, a switch from aerobic to microaerobic conditions leads to the synthesis of a recombinant holoenzyme, while the maintenance of aerobic conditions results in the synthesis of a copper-depleted population of proteins. Strikingly, cells grown under microaerobic conditions accumulate up to 80-fold more copper than aerobically grown cells. In vitro copper incorporation into apoenzymes was monitored by optical and electron paramagnetic resonance (EPR) spectroscopy. This analysis reveals that copper incorporation into CotA laccase is a sequential process, with the type 1 copper center being the first to be reconstituted, followed by the type 2 and the type 3 copper centers. The copper reconstitution of holoCotA derivatives depleted in vitro with EDTA results in the complete recovery of the native conformation as monitored by spectroscopic, kinetic and thermal stability analysis. However, the reconstitution of copper to apo forms produced in cultures under aerobic and copper-deficient conditions resulted in incomplete recovery of biochemical properties of the holoenzyme. EPR and resonance Raman data indicate that, presumably, folding in the presence of copper is indispensable for the correct structure of the trinuclear copper-containing site.






Similar content being viewed by others
References
Solomon EI, Sundaram UM, Machonkin TE (1996) Chem Rev 96:2563–2605
Stoj CS, Kosman DJ (2005) In: King RB (ed) Encyclopedia of inorganic chemistry, vol II, 2nd edn. Wiley, New York, pp 1134–1159
Lindley PF (2001) In: Bertini I, Sigel A, Sigel H (eds) Handbook on metalloproteins. Dekker, New York, pp 763–811
Blair DF, Campbell GW, Schoonover JR, Chan SI, Gray HB, Malmstrom BG, Pecht I, Swanson BI, Wooddruff WH, Cho WK, English AM, Fry HA, Lum V, Norton KA (1985) J Am Chem Soc 107:5755–5766
Martins LO, Soares CM, Pereira MM, Teixeira M, Jones GH, Henriques AO (2002) J Biol Chem 277:18849–18859
Hullo M-F, Moszer I, Danchin A, Martin-Verstraete I (2001) J Bacteriol 183:5426–5430
Donovan W, Zheng L, Sandman K, Losick R (1987) J Mol Biol 196:1–10
Enguita FJ, Martins LO, Henriques AO, Carrondo MA (2003) J Biol Chem 278:19416–25
Bento I, Martins LO, Gato GL, Carrondo MA, Lindley PF (2005) Dalton Trans 21:3507– 3513
Durão P, Bento I, Fernandes AT, Melo EP, Lindley PF, Martins LO (2006) J Biol Inorg Chem 11:514–526
Davis-Kaplan SR, Askwith CC, Bengtzen AC, Radisky D, Kaplan J (1998) Proc Natl Acad Sci USA 95:13641–13645
Blackburn NJ, Ralle M, Hassett R, Kosman DJ (2000) Biochemistry 39:2316–2324
Palmer AE, Szilagyi RK, Cherry JR, Jones A, Xu F, Solomon EI (2003) Inorg Chem 42:4006–4017
Hellman NE, Kono S, Mancini GM, Hoogeboom AJ, de Jong GJ, Gitlin JD (2002) J Biol Chem 277:46632–46638
Galli I, Musci G, di Patti MCB (2004) J Biol Inorg Chem 9:90–95
Xu F (1999) In: Flickinger MC, Drewn SW (eds) Encyclopedia of bioprocess technology: fermentation, biocatalysis and bioseparation. Wiley, New York, pp 1545–1554
Brenner AJ, Harris ED (1995) Anal Biochem 226:80–84
Bradford MM (1976) Anal Biochem 72:248–254
Aasa R, Väangard VT (1975) J Magnet Reson 19:308–315
Sanchez-Amat A, Lucas-Elio P, Fernández E, Garcia-Borrón JC, Solano F (2001) Biochim Biophys Acta 1547:104–116
Grass G, Rensing C (2003) FEMS Microbiol Rev 27:197–213
Changela A, Chen K, Xue Y, Holschen J, Outten CE, O’Halloran TV, Mondragón A (2003) Science 301:1383–1387
Finney LA, O’Halloran TV (2003) Science 300:931–936
Outten FW, Huffman DL, Hale JA, O’Halloran TV (2001) J Biol Chem 276:30670–30677
Macomber L, Rensing C, Imlay JA (2007) J Bacterial 189:1616–1626
Beswick PH, Hall GH, Hook AJ, Little K, McBride DCH, Lott KAK (1976) Chem Biol Interact 14:347–356
Partdridge JD, Sanguinetti G, Dibden D, Roberts RE, Poole RK, Green J (2007) J Biol Chem 282:11230–11237
Green MT (2006) J Am Chem Soc 128:1902–1906
Palmer AE, Randall DW, Xu F, Solomon EI (1999) J Am Chem Soc 121:7138–7149
Machokin TE, Quintanar L, Palmer AE, Hassett R, Severance S, Kosman DJ, Solomon EI (2001) J Am Chem Soc 123:5507–5517
Kataoka K, Kitagawa R, Inoue M, Naruse D, Sakurai T, Huang H-W (2005) Biochemistry 44:7004–7012
Volkin DB, Klibanov AM (1989) In: Creighton TE (ed) Minimizing protein inactivation protein function. A practical approach. IRL, Oxford, pp 1–24
Savini I, D’Alessio S, Giartosio A, Morpurgo L, Avigliano L (1990) Eur J Biochem 190:491–495
di Patti MCB, Musci G, Giartosio A, D’Alessio S, Calabrese L (1990) J Biol Chem 265:21016–21022
Vassall KA, Stathopulos PB, Rumfeldt JAO, Lepock JR, Meiering EM (2006) Biochemistry 45:7366–7379
Agostinelli E, Cervoni L, Giartosio A, Morpurgo L (1995) Biochem J 306:697–702
Ragusa S, Cambria MT, Pierfederici F, Scirè A, Bertoli E, Tanfani F, Cambria A (2002) Biochim Biophys Acta 1601:155–162
Koroleva OV, Stepanova EV, Binukov VI, Timofeev VP, Pfeil W (2001) Biochimic Biophys Acta 1547:397–407
Milardi D, Grasso DM, Verbeet MP, Canters GW, La Rosa C (2003) Arch Biochem Biophys 414:121–127
Acknowledgements
This work was supported by POCI/BIO/57083/2004 and FP6-2004-NMP-NI-4/026456 project grants. P.F. Lindley and A. Sanchez Amat are acknowledged for their useful suggestions. We thank P. Jackson for correcting the English. Z. Chen holds a Post-doc fellowship (SFRH/BPD/27104/2006) and A.T. Fernandes a PhD fellowship (SFRH/BPD/31444/2006).
Author information
Authors and Affiliations
Corresponding author
Additional information
Paulo Durão and Zhenjia Chen contributed equally to this work.
Rights and permissions
About this article
Cite this article
Durão, P., Chen, Z., Fernandes, A.T. et al. Copper incorporation into recombinant CotA laccase from Bacillus subtilis: characterization of fully copper loaded enzymes. J Biol Inorg Chem 13, 183–193 (2008). https://doi.org/10.1007/s00775-007-0312-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00775-007-0312-0