Abstract
A highly sensitive electrochemical sensor is described for the determination of H2O2. It is based on based on the use of polyaniline that was generated in-situ and within 1 min on a glassy carbon electrode (GCE) with the aid of the Fe(II)/H2O2 system. Initially, a 2-dimensional composite was prepared from graphene oxide and polyamidoamine dendrimer through covalent interaction. It was employed as a carrier for Fe(II) ions. Then, the nanocomposite was drop-coated onto the surface of the GCE. When exposed to H2O2, the Fe(II) on the GCE is converted to Fe(III), and free hydroxy radicals are formed. The Fe(III) ions and the hydroxy radicals catalyze the oxidation of aniline to produce electroactive polyaniline on the GCE. The resulting sensor, best operated at a working potential as low as 50 mV (vs. SCE) which excludes interference by dissolved oxygen, has a linear response in the 500 nM to 2 mM H2O2 concentration range, and the detection limit is 180 nM. The sensor was successfully applied to the determination of H2O2 in spiked milk and fetal bovine serum samples.

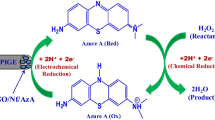
Schematic presentation of a sensitive electrochemical sensor employed for detection of H2O2 in sophisticated matrices by using graphene oxide-PAMAM dendrimer as initiator container and Fe2+/H2O2 system as signal enhancer.





Similar content being viewed by others
References
Chen S, Yuan R, Chai Y, Hu F (2013) Electrochemical sensing of hydrogen peroxide using metal nanoparticles: a review. Microchim Acta 180:15–32
Yan Z, Zhao J, Qin L, Mu F, Wang P, Feng X (2013) Non-enzymatic hydrogen peroxide sensor based on a gold electrode modified with granular cuprous oxide nanowires. Microchim Acta 180:145–150
Cui X, Wu S, Li Y, Wan G (2015) Sensing hydrogen peroxide using a glassy carbon electrode modified with in-situ electrodeposited platinum-gold bimetallic nanoclusters on a graphene surface. Microchim Acta 182:265–272
Chang H-C, Ho J-AA (2015) Gold nanocluster-assisted fluorescent detection for hydrogen peroxide and cholesterol based on the inner filter effect of gold nanoparticles. Anal Chem 87:10362–10367
Li M, Gao H, Wang X, Wang Y, Qi H, Zhang C (2017) A fluorine-doped tin oxide electrode modified with gold nanoparticles for electrochemiluminescent determination of hydrogen peroxide released by living cells. Microchim Acta 184:603–610
Xiang Z, Wang Y, Ju P, Zhang D (2016) Optical determination of hydrogen peroxide by exploiting the peroxidase-like activity of AgVO3 nanobelts. Microchim Acta 183:457–463
Gimeno P, Bousquet C, Lassu N, Maggio A-F, Civade C, Brenier C, Lempereur L (2015) High-performance liquid chromatography method for the determination of hydrogen peroxide present or released in teeth bleaching kits and hair cosmetic products. J Pharmaceut Biomed 107:386–393
Pan N, Li-Ying W, Wu L-L, Peng C-F, Xie Z-J (2017) Colorimetric determination of cysteine by exploiting its inhibitory action on the peroxidase-like activity of Au@Pt core-shell nanohybrids. Microchim Acta 184:65–72
Pauliukaite R, Hočevar SB, Hutton EA, Ogorevc B (2008) Novel electrochemical microsensor for hydrogen peroxide based on iron-ruthenium hexacyanoferrate modified carbon fiber electrode. Electroanalysis 20:47–53
Zhu C, Yang G, Li H, Du D, Lin Y (2015) Electrochemical sensors and biosensors based on nanomaterials and nanostructures. Anal Chem 87:230–249
Wang Y, Wang Z, Rui Y, Li M (2015) Horseradish peroxidase immobilization on carbon nanodots/cofe layered double hydroxides: direct electrochemistry and hydrogen peroxide sensing. Biosens Bioelectron 64:57–62
Tang J, Tang D (2015) Non-enzymatic electrochemical immunoassay using noble metal nanoparticles: a review. Microchim Acta 182:2077–2089
Zhang Y, Huang B, Yu F, Yuan Q, Gu M, Ji J, Zhang Y, Li Y (2018) 3D nitrogen-doped graphite foam@prussian blue: an electrochemical sensing platform for highly sensitive determination of H2O2 and glucose. Microchim Acta 185:86
Chen X, Wu G, Cai Z, Oyama M, Chen X (2014) Advances in enzyme-free electrochemical sensors for hydrogen peroxide, glucose, and uric acid. Microchim Acta 181:689–705
Jin J, Wu W, Min H, Wu H, Wang S, Ding Y, Yang S (2017) A glassy carbon electrode modified with FeS nanosheets as a highly sensitive amperometric sensor for hydrogen peroxide. Microchim Acta 184:1389–1396
Butwong N, Zhou L, Ng-eontae W, Burakham R, Moore E, Srijaranai S, Luong JHT, Glennon JD (2014) A sensitive nonenzymatic hydrogen peroxide sensor using cadmium oxide nanoparticles/multiwall carbon nanotube modified glassy carbon electrode. J Electroanal Chem 717-718:41–46
Li Y, Lu Q, Wu S, Wang L, Shi X (2013) Hydrogen peroxide sensing using ultrathin platinum-coated gold nanoparticles with core@shell structure. Biosens Bioelectron 41:576–581
Lai J, Yi Y, Zhu P, Shen J, Wu K, Zhang L, Liu J (2016) Polyaniline-based glucose biosensor: a review. J Electroanal Chem 782:138–153
Sen T, Mishra S, Shimpi NG (2016) Synthesis and sensing applications of polyaniline nanocomposites: a review. RSC Adv 6:42196–42222
Li H, Chang J, Hou T, Li F (2017) HRP-mimicking DNAzyme-catalyzed in situ generation of polyaniline to assist signal amplification for ultrasensitive surface plasmon resonance biosensing. Anal Chem 89:673–680
Zhang X, Shen J, Ma H, Jiang Y, Huang C, Han E, Yao B, He Y (2016) Optimized dendrimer-encapsulated gold nanoparticles and enhanced carbon nanotube nanoprobes for amplified electrochemical immunoassay of E. Coli in dairy product based on enzymatically induced deposition of polyaniline. Biosens Bioelectron 80:666–673
Peña RC, Silva VO, Quina FH, Bertotti M (2012) Hydrogen peroxide monitoring in Fenton reaction by using a ruthenium oxide hexacyanoferrate/multiwalled carbon nanotubes modified electrode. J Electroanal Chem 686:1–6
Zhang L, Wan M, Wei Y (2006) Nanoscaled polyaniline fibers prepared by ferric chloride as an oxidant. Macromol Rapid Commun 27:366–371
Jiang W, Wu L, Duan J, Yin H, Ai S (2018) Ultrasensitive electrochemiluminescence immunosensor for 5-hydroxymethylcytosine detection based on Fe3O4@SiO2 nanoparticles and PAMAM dendrimers. Biosens Bioelectron 99:660–666
Marcano DC, Kosynkin DV, Berlin JM, Sinitskii A, Sun Z, Slesarev A, Alemany LB, Lu W, Tour JM (2010) Improved synthesis of graphene oxide. ACS Nano 4:4806–4814
Tang J, Huang Y, Zhang C, Liu H, Tang D (2016) Amplified impedimetric immunosensor based on instant catalyst for sensitive determination of ochratoxin A. Biosens Bioelectron 86:386–392
Lai G, Zhang H, Tamanna T, Yu A (2014) Ultrasensitive immunoassay based on electrochemical measurement of enzymatically produced polyaniline. Anal Chem 86:1789–1793
Mei L, Zhang P, Chen J, Chen D, Quan Y, Gu N, Zhang G, Cui R (2016) Non-enzymatic sensing of glucose and hydrogen peroxide using a glassy carbon electrode modified with a nanocomposite consisting of nanoporous copper, carbon black and nafion. Microchim Acta 183:1359–1365
Yusoff N, Rameshkumar P, Mehmood MS, Pandikumar A, Lee HW, Huang NM (2017) Ternary nanohybrid of reduced graphene oxide-nafion@silver nanoparticles for boosting the sensor performance in non-enzymatic amperometric detection of hydrogen peroxide. Biosens Bioelectron 87:1020–1028
Zhang J, Tu L, Zhao S, Liu G, Wang Y, Wang Y, Yue Z (2015) Fluorescent gold nanoclusters based photoelectrochemical sensors for detection of H2O2 and glucose. Biosens Bioelectron 67:296–302
Sun X, Guo S, Chung C-S, Zhu W, Sun S (2013) A sensitive H2O2 assay based on dumbbell-like PtPd-Fe3O4 nanoparticles. Adv Mater 25:132–136
Ding L, Chen S, Zhang W, Zhang Y, Wang X-D (2018) Fully reversible optical sensor for hydrogen peroxide with fast response. Anal Chem 90:7544–7551
Abnous K, Danesh NM, Alibolandi M, Ramezani M, Taghdisi SM (2017) Amperometric aptasensor for ochratoxin A based on the use of a gold electrode modified with aptamer, complementary DNA, SWNTs and the redox marker methylene blue. Microchim Acta 184:1151–1159
Neal CJ, Gupta A, Barkam S, Saraf S, Das S, Cho HJ, Seal S (2017) Picomolar detection of hydrogen peroxide using enzyme-free inorganic nanoparticle-based sensor. Sci Rep 7:1324
Miao X, Yang C, Leung C-H, Ma D-L (2016) Application of iridium(iii) complex in label-free and non-enzymatic electrochemical detection of hydrogen peroxide based on a novel "on-off-on" switch platform. Sci Rep 6:25774–25774
Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (21864013, 21505060), the Nature Science Foundation of Jiangxi Province (20161BAB213073), the Foundation of Jiangxi Educational Committee (GJJ150327), the Natural Science Foundation of Fujian Province (2018 J01442), the Fujian Educational Committee (JAT170487), the Foundation for High-Level Talents in Quanzhou City (2017Z029), Scientific Research Foundation of Jiangxi Normal University, the Open Project Program of Key Laboratory of Functional Small Organic Molecule, Ministry of Education, Jiangxi Normal University (No. KLFS-KF-201710), Outstanding Youth Funds of Jiangxi Normal University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The author(s) declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOC 133 kb)
Rights and permissions
About this article
Cite this article
Tang, J., Huang, L., Cheng, Y. et al. Nonenzymatic sensing of hydrogen peroxide using a glassy carbon electrode modified with graphene oxide, a polyamidoamine dendrimer, and with polyaniline deposited by the Fenton reaction. Microchim Acta 185, 569 (2018). https://doi.org/10.1007/s00604-018-3089-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00604-018-3089-7