Abstract
Purpose
There have been contradictory findings regarding the effects of sevoflurane on the oncogenic properties of cancer cells. This study was conducted to gain insights into the fundamental rules governing the differential effects of sevoflurane exposure on various cancer cells derived from multiple origins.
Methods
A series of cancer cell lines were exposed to 1% (v/v) sevoflurane for 2–8 h and then assessed for their proliferation, Matrigel invasion, and apoptotic cell death, in comparison with their untreated counterparts. Cell proliferation and Matrigel invasion assays were performed using Coulter counter and Boyden chamber techniques, respectively. Apoptosis was evaluated by staining cells with Annexin V and 7-AAD followed by fluorescence flow cytometry. In addition, the expression of cleaved caspase-3 protein, another marker of apoptosis, was assessed using immunoblotting.
Results
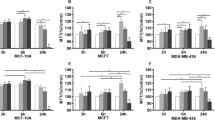
Proliferation was significantly enhanced after sevoflurane exposure in six of eight cancer cell lines (NCI-H1299, MDA-MB-231, HCT116, DLD-1, HT29, and RKO). In contrast, sevoflurane attenuated proliferation in the last two cancer cell lines, A549 and MCF-7, as well as in the non-cancerous MCF10A cell line. Cell biological assays using four cancer cell lines demonstrated that accelerated but not attenuated cancer cell proliferation after sevoflurane exposure is associated with enhanced Matrigel invasion and suppressed apoptosis.
Conclusion
Sevoflurane augmented or hampered cell proliferation and Matrigel invasion depending on the cancer cell line examined. Loss of sevoflurane-induced apoptosis occurring in cancer cell lines is likely to be correlated with their enhanced proliferation after sevoflurane exposure.







Similar content being viewed by others
Change history
03 December 2019
The article Differential effects of sevoflurane on the growth and apoptosis of human cancer cell lines, written by Takahiro Hirai, Yuko Konishi, Shoko Mizuno, Zhou Rui, Yao Sun and Kimitoshi Nishiwaki, was originally published electronically on the publisher’s internet portal (currently SpringerLink) on 31 October 2019 with open access. With the author(s)’ decision to step back from Open Choice, the copyright of the article changed on 5 December 2019 to © Japanese Society of Anesthesiologists 2019 and the article is forthwith distributed under the terms of copyright.
References
Robinson BJ, Uhrich TD, Ebert TJ. A review of recovery from sevoflurane anaesthesia: comparisons with isoflurane and propofol including meta-analysis. Acta Anaesthesiol Scand. 1999;43(2):185–90.
Tavare AN, Perry NJS, Benzonana LL, Takata M, Ma D. Cancer recurrence after surgery: Direct and indirect effects of anesthetic agents. Int J Cancer. 2012;130(6):1237–50.
Brioni JD, Varughese S, Ahmed R, Bein B. A clinical review of inhalation anesthesia with sevoflurane: from early research to emerging topics. J Anesth. 2017;31(5):764–78.
Ecimovic P, McHugh B, Murray D, Doran P, Buggy DJ. Effects of sevoflurane on breast cancer cell function in vitro. Anticancer Res. 2013;33(10):4255–60.
Jaura AI, Flood G, Gallagher HC, Buggy DJ. Differential effects of serum from patients administered distinct anaesthetic techniques on apoptosis in breast cancer cells in vitro: a pilot study. Br J Anaesth. 2014;113(Suppl 1):i63–i6767.
Nishiwada T, Kawaraguchi Y, Uemura K, Sugimoto H, Kawaguchi M. Effect of sevoflurane on human hepatocellular carcinoma HepG2 cells under conditions of high glucose and insulin. J Anesth. 2015;29(5):805–8.
Shi QY, Zhang SJ, Liu L, Chen QS, Yu LN, Zhang FJ, Yan M. Sevoflurane promotes the expansion of glioma stem cells through activation of hypoxia-inducible factors in vitro. Br J Anaesth. 2015;114(5):825–30.
Benzonana LL, Perry NJ, Watts HR, Yang B, Perry IA, Coombes C, Takata M, Ma D. Isoflurane, a commonly used volatile anesthetic, enhances renal cancer growth and malignant potential via the hypoxia-inducible factor cellular signaling pathway in vitro. Anesthesiology. 2013;119(3):593–605.
Kawaraguchi Y, Horikawa YT, Murphy AN, Murray F, Miyanohara A, Ali SS, Head BP, Patel PM, Roth DM, Patel HH. Volatile anesthetics protect cancer cells against tumor necrosis factor-related apoptosis-inducing ligand-induced apoptosis via caveolins. Anesthesiology. 2011;115(3):499–508.
Niwa H, Rowbotham DJ, Lambert DG, Buggy DJ. Can anesthetic techniques or drugs affect cancer recurrence in patients undergoing cancer surgery? J Anesth. 2013;27(5):731–41.
Kvolik S, Glavas-Obrovac L, Bares V, Karner I. Effects of inhalation anesthetics halothane, sevoflurane, and isoflurane on human cell lines. Life Sci. 2005;77(19):2369–83.
Kvolik S, Dobrosevic B, Marczi S, Prlic L, Glavas-Obrovac L. Different apoptosis ratios and gene expressions in two human cell lines after sevoflurane anaesthesia. Acta Anaesthesiol Scand. 2009;53(9):1192–9.
Muller-Edenborn B, Roth-Z'graggen B, Bartnicka K, Borgeat A, Hoos A, Borsig L, Beck-Schimmer B. Volatile anesthetics reduce invasion of colorectal cancer cells through down-regulation of matrix metalloproteinase-9. Anesthesiology. 2012;117(2):293–301.
Liang H, Gu M, Yang C, Wang H, Wen X, Zhou Q. Sevoflurane inhibits invasion and migration of lung cancer cells by inactivating the p38 MAPK signaling pathway. J Anesth. 2012;26(3):381–92.
Liu S, Fang F, Song R, Gao X, Jiang M, Cang J. Sevoflurane affects neurogenesis through cell cycle arrest via inhibiting wnt/beta-catenin signaling pathway in mouse neural stem cells. Life Sci. 2018;209:34–42.
Loop T, Dovi-Akue D, Frick M, Roesslein M, Egger L, Humar M, Hoetzel A, Schmidt R, Borner C, Pahl HL, Geiger KK, Pannen BH. Volatile anesthetics induce caspase-dependent, mitochondria-mediated apoptosis in human T lymphocytes in vitro. Anesthesiology. 2005;102(6):1147–57.
Dong Y, Zhang G, Zhang B, Moir RD, Xia W, Marcantonio ER, Culley DJ, Crosby G, Tanzi RE, Xie Z. The common inhalational anesthetic sevoflurane induces apoptosis and increases beta-amyloid protein levels. Arch Neurol. 2009;66(5):620–31.
Topouzova-Hristova T, Daza P, Garcia-Herdugo G, Stephanova E. Volatile anaesthetic halothane causes DNA damage in A549 lung cells. Toxicol In Vitro. 2006;20(5):585–93.
Yang Z, Lv J, Li X, Meng Q, Yang Q, Ma W, Li Y, Ke ZJ. Sevoflurane decreases self-renewal capacity and causes c-Jun N-terminal kinase–mediated damage of rat fetal neural stem cells. Sci Rep. 2017;7:46304.
Kuilman T, Michaloglou C, Vredeveld LC, Douma S, van Doorn R, Desmet CJ, Aarden LA, Mooi WJ, Peeper DS. Oncogene-induced senescence relayed by an interleukin-dependent inflammatory network. Cell. 2008;133(6):1019–31.
Dhanasekaran DN, Reddy EP. JNK signaling in apoptosis. Oncogene. 2008;27(48):6245–51.
Acknowledgements
We wish to thank Mr. Minoru Tanaka in the Division of Medical Research Engineering, Nagoya University Graduate School of Medicine, for assistance with flow cytometry. This study was supported by Grants-in-Aid for Scientific Research (KAKENHI) from the Japan Society for the Promotion of Science (JSPS) [15K15569 to TH, 16K10932 to YK, 15K20042 to SM], as well as the Program to supporting research activities of female researchers from the Ministry of Education, Culture, Sports, Science and Technology (MEXT), Japan, 2016–2018 (to YK). This study constitutes the doctoral thesis of T.H.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original version of this article was revised due to retrospective open access cancellation order.
About this article
Cite this article
Hirai, T., Konishi, Y., Mizuno, S. et al. Differential effects of sevoflurane on the growth and apoptosis of human cancer cell lines. J Anesth 34, 47–57 (2020). https://doi.org/10.1007/s00540-019-02701-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00540-019-02701-w