Abstract
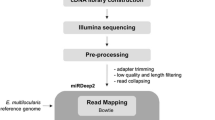
Ascaris nematodes, which cause ascariasis in humans and pigs, are among the most important nematodes from both health and economic perspectives. microRNA (miRNA) is now recognized as key regulator of gene expression at posttranscription level. The public availability of the genome and transcripts of Ascaris suum provides powerful resources for the research of miRNA profiles of the parasite. Therefore, we investigated and compared the miRNA profiles of male and female adult A. suum using Solexa deep sequencing combined with bioinformatic analysis and stem-loop reverse transcription polymerase chain reaction. Deep sequencing of small RNAs yielded 11.71 and 11.72 million raw reads from male and female adults of A. suum, respectively. Analysis showed that the noncoding RNA of the two genders, including tRNA, rRNA, snRNA, and snoRNA, were similar. By mapping to the A. suum genome, we obtained 494 and 505 miRNA candidates from the female and male parasite, respectively, and 87 and 82 of miRNA candidates were consistent with A. suum miRNAs deposited in the miRBase database. Among the miRNA candidates, 154 were shared by the two genders, and 340 and 351 were female and male specific with their target numbers ranged from one to thousands, respectively. Functional prediction revealed a set of elongation factors, heat shock proteins, and growth factors from the targets of gender-specific miRNAs, which were essential for the development of the parasite. Moreover, major sperm protein and nematode sperm cell motility protein were found in targets of the male-specific miRNAs. Ovarian message protein was found in targets of the female-specific miRNAs. Enrichment analysis revealed significant differences among Gene Ontology terms of miRNA targets of the two genders, such as electron carrier and biological adhesion process. The regulating functions of gender-specific miRNAs was therefore not only related to the fundamental functions of cells but also were essential to the germ development of the parasite. The present study provides a framework for further research of Ascaris miRNAs, and consequently leads to the development of potential nucleotide vaccines against Ascaris of human and animal health significance.


Similar content being viewed by others
References
Altschul SF, Gish W, Miller W, Myers EW, Lipman DJ (1990) Basic local alignment search tool. J Mol Biol 215:403–410
Antebi A (2012) Regulation of longevity by the reproductive system. Exp Gerontol. doi:10.1016/j.exger.2012.09.009
Ai L, Xu MJ, Chen MX, Zhang YN, Chen SH, Guo J, Cai YC, Zhou XN, Zhu XQ, Chen JX (2011) Characterization of microRNAs in Taenia saginata of zoonotic significance by solexa deep sequencing and bioinformatics analysis. Parasitol Res 110:2373–2378
Bartel DP (2004) microRNAs genomics, biogenesis, mechanism, and function. Cell 116:281–297
Cantacessi C, Zou FC, Hall RS, Zhong W, Jex AR, Campbell BE, Ranganathan S, Sternberg PW, Zhu XQ, Gasser RB (2009) Bioinformatic analysis of abundant, gender-enriched transcripts of adult Ascaris suum (Nematoda) using a semi-automated workflow platform. Mol Cell Probes 23:205–217
Chen C, Ridzon DA, Broomer AJ, Zhou Z, Lee DH, Nguyen JT, Barbisin M, Xu NL, Mahuvakar VR, Andersen MR, Lao KQ, Livak KJ, Guegler KJ (2005) Real-time quantification of microRNAs by stem-loop RT-PCR. Nucleic Acids Res 33:e179
Chiba Y, Hijikata T (2010) MicroRNAs and their therapeutic potential for human diseases: preface. J Pharmacol Sci 114:262–263
Crompton DW (2001) Ascaris and ascariasis. Adv Parasitol 48:285–375
de Silva NR, Brooker S, Hotez PJ, Montresor A, Engels D, Savioli L (2003) Soil-transmitted helminth infections: updating the global picture. Trends Parasitol 19:547–551
Dold C, Holland CV (2011) Ascaris and ascariasis. Microbes Infect 13:632–637
Goździk K, Engström A, Höglund J (2012) Optimization of in-house ELISA based on recombinant major sperm protein (rMSP) of Dictyocaulus viviparus for the detection of lungworm infection in cattle. Res Vet Sci 93:813–818
Du T, Zamore PD (2007) Beginning to understand microRNA function. Cell Res 17:661–663
Fonseca CT, Braz Figueiredo Carvalho G, Carvalho Alves C, de Melo TT (2012) Schistosoma tegument proteins in vaccine and diagnosis development: an update. J Parasitol Res 2012:541268
Huang CQ, Gasser RB, Cantacessi C, Nisbet AJ, Zhong W, Sternberg PW, Loukas A, Mulvenna J, Lin RQ, Chen N, Zhu XQ (2008) Genomic-bioinformatic analysis of transcripts enriched in the third-stage larva of the parasitic nematode Ascaris suum. PLoS Negl Trop Dis 2:e246
Höglund J, Engström A, Morrison DA, Mineur A, Mattsson JG (2008) Limited sequence variation in the major sperm protein 1 (MSP) gene within populations and species of the genus Dictyocaulus (Nematoda). Parasitol Res 103:11–20
Jex AR, Liu S, Li B, Young ND, Hall RS, Li Y, Yang L, Zeng N, Xu X, Xiong Z, Chen F, Wu X, Zhang G, Fang X, Kang Y, Anderson GA, Harris TW, Campbell BE, Vlaminck J, Wang T, Cantacessi C, Schwarz EM, Ranganathan S, Geldhof P, Nejsum P, Sternberg PW, Yang H, Wang J, Wang J, Gasser RB (2011) Ascaris suum draft genome. Nature 479:529–533
Joachim A, Dulmer N, Daugschies A, Roepstorff A (2001) Occurrence of helminths in pig fattening units with different management systems in Northern Germany. Vet Parasitol 96:135–146
Kruger J, Rehmsmeier M (2006) RNAhybrid: microRNA target prediction easy, fast and flexible. Nucleic Acids Res 34:W451–W454
Laabs EM, Schnieder T, Strube C (2012) In vitro studies on the sexual maturation of the bovine lungworm Dictyocaulus viviparus during the development of preadult larvae to adult worms. Parasitol Res 110:1249–1259
Leles D, Gardner SL, Reinhard K, Iniguez A, Araujo A (2012) Are Ascaris lumbricoides and Ascaris suum a single species? Parasit Vectors 5:42
Lewis R, Behnke JM, Stafford P, Holland CV (2009) Dose-dependent impact of larval Ascaris suum on host body weight in the mouse model. J Helminthol 83:1–5
Li BW, Rush AC, Crosby SD, Warren WC, Williams SA, Mitreva M, Weil GJ (2005) Profiling of gender-regulated gene transcripts in the filarial nematode Brugia malayi by cDNA oligonucleotide array analysis. Mol Biochem Parasitol 143:49–57
Li R, Yu C, Li Y, Lam TW, Yiu SM, Kristiansen K, Wang J (2009) SOAP2: an improved ultrafast tool for short read alignment. Bioinformatics 25:1966–1967
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(−delta delta C(T)) method. Methods 25:402–408
Myers EM (2012) Gαo and Gαq regulate the expression of daf-7, a TGFβ-like gene, in Caenorhabditis elegans. PLoS One 7:e40368
Nisbet AJ, Gasser RB (2004) Profiling of gender-specific gene expression for Trichostrongylus vitrinus (Nematoda: Strongylida) by microarray analysis of expressed sequence tag libraries constructed by suppressive–subtractive hybridisation. Int J Parasitol 34:633–643
Park J, Dickerson TJ, Janda KD (2008) Major sperm protein as a diagnostic antigen for onchocerciasis. Bioorg Med Chem 16:7206–7209
Roberts TM, Stewart M (2012) Role of major sperm protein (MSP) in the protrusion and retraction of Ascaris sperm. Int Rev Cell Mol Biol 297:265–293
Schunn AM, Forbes A, Schnieder T, Strube C (2012) Validation of a Dictyocaulus viviparus MSP-ELISA and cut-off adjustment in a one-year longitudinal field study in dairy cattle herds. Vet Parasitol 189:291–298
Strube C, Buschbaum S, Schnieder T (2009) Molecular characterization and real-time PCR transcriptional analysis of Dictyocaulus viviparus major sperm proteins. Parasitol Res 104:543–551
Thorsen SB, Obad S, Jensen NF, Stenvang J, Kauppinen S (2012) The therapeutic potential of microRNAs in cancer. Cancer J 18:275–284
Uysal HK, Boral O, Metiner K, Ilgaz A (2009) Investigation of intestinal parasites in pig feces that are also human pathogens. Turkiye Parazitol Derg 33:218–221
Wang J, Czech B, Crunk A, Wallace A, Mitreva M, Hannon GJ, Davis RE (2011) Deep small RNA sequencing from the nematode Ascaris reveals conservation, functional diversification, and novel developmental profiles. Genome Res 21:1462–1477
Xu MJ, Liu Q, Nisbet AJ, Cai XQ, Yan C, Lin RQ, Yuan ZG, Song HQ, He XH, Zhu XQ (2010) Identification and characterization of microRNAs in Clonorchis sinensis of human health significance. BMC Genomics 11:521
Acknowledgments
This work was supported by The Science Fund for Creative Research Groups of Gansu Province (grant No. 1210RJIA006), the National S & T Major Program (grant No. 2012ZX10004220), the Open Funds of the State Key Laboratory of Veterinary Etiological Biology, Lanzhou Veterinary Research Institute, Chinese Academy of Agricultural Sciences (grant No. SKLVEB2011KFKT004 and SKLVEB2011KFKT010), the China Postdoctoral Science Foundation (grant No. 201104363 and 20090460064), and the Guangdong Provincial Program for Excellent Ph.D. theses (grant No. sybzzxm201037). The experiments comply with the current laws of the country in which the experiments were performed.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Additional file 1
Conserved gender-specific miRNAs of A. suum. (DOCX 18 kb)
Additional file 2
Annotated functions of conserved gender-specific miRNAs of A. suum. (DOCX 17 kb)
Additional file 3
Predicated female targets of female-specific miRNAs by using mRNA and EST deposited in the GenBank. (DOCX 52 kb)
Additional file 4
Predicated male targets of male-specific miRNAs by using mRNA and EST deposited in the GenBank. (DOCX 50 kb)
Rights and permissions
About this article
Cite this article
Xu, MJ., Fu, JH., Nisbet, A.J. et al. Comparative profiling of microRNAs in male and female adults of Ascaris suum . Parasitol Res 112, 1189–1195 (2013). https://doi.org/10.1007/s00436-012-3250-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-012-3250-x