Abstract
Purpose
Transglutaminase 2 (TGase 2), a cross-linking enzyme, plays an important role in both pro-survival and anti-apoptosis during oncogenesis. For instance, TGase 2 induces NF-κB activation through I-κBα polymerization, which leads to the increase of pro-survival factors such as BCl-2. TGase 2 also suppresses apoptosis via depletion of caspase 3 and cathepsin D. Therefore, a specific TGase 2 inhibitor may become a very useful treatment for cancer showing high levels of TGase 2 expression.
Methods
By small-molecule library screening, we managed to locate a competitive TGase 2 inhibiting quinoxaline compound (GK13) from 50 other quinoxaline derivatives. The 50 compounds that were screened represent a thousand structurally diverse, potentially pharmaceutical heterocyclic compound libraries, including benzopyrans, oxadiazoles, thiadiazoles, and quinoxalines. By measuring GI50, TGI, and LC50 using SRB assay, GK13 was selected.
Results
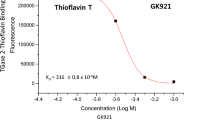
In vitro enzyme kinetics using guinea pig liver TGase 2 showed that IC50 value was about 16.4 E−6 M. GK13 inhibits TGase 2-mediated I-κBα polymerization in a dose-dependent manner. LC50 of GK13 showed greater efficacy as 4.3E−4 M than LC50 of doxorubicin that showed efficacy as 3.87E−3 M in NCC72 composing 11 tissue origins and 72 cancer cell lines.
Conclusion
GK13 showed a possibility that quinoxaline derivatives may be effective for anti-cancer activity via TGase 2 inhibition.












Similar content being viewed by others
References
Caccamo D, Curro M, Ientile R (2010) Potential of transglutaminase 2 as a therapeutic target. Expert Opin Ther Targets 14(9):989–1003. doi:10.1517/14728222.2010.510134
Choi K, Siegel M, Piper JL, Yuan L, Cho E, Strnad P, Omary B, Rich KM, Khosla C (2005) Chemistry and biology of dihydroisoxazole derivatives: selective inhibitors of human transglutaminase 2. Chem Biol 12(4):469–475. doi:10.1016/j.chembiol.2005.02.007
Delhase M, Kim SY, Lee H, Naiki-Ito A, Chen Y, Ahn ER, Murata K, Kim SJ, Lautsch N, Kobayashi KS, Shirai T, Karin M, Nakanishi M (2012) TANK-binding kinase 1 (TBK1) controls cell survival through PAI-2/serpinB2 and transglutaminase 2. Proc Natl Acad Sci USA 109(4):E177–E186. doi:10.1073/pnas.1119296109
Folk JE (1980) Transglutaminases. Annu Rev Biochem 49:517–531. doi:10.1146/annurev.bi.49.070180.002505
Folk JE, Park MH, Chung SI, Schrode J, Lester EP, Cooper HL (1980) Polyamines as physiological substrates for transglutaminases. J Biol Chem 255(8):3695–3700
Frese-Schaper M, Schardt JA, Sakai T, Carboni GL, Schmid RA, Frese S (2010) Inhibition of tissue transglutaminase sensitizes TRAIL-resistant lung cancer cells through upregulation of death receptor 5. FEBS Lett 584(13):2867–2871. doi:10.1016/j.febslet.2010.04.072
Gong YD, Lee T (2010) Combinatorial syntheses of five-membered ring heterocycles using carbon disulfide and a solid support. J Comb Chem 12(4):393–409. doi:10.1021/cc100049u
Gong YD, Dong MS, Lee SB, Kim N, Bae MS, Kang NS (2011) A novel 3-arylethynyl-substituted pyrido[2,3,-b]pyrazine derivatives and pharmacophore model as Wnt2/beta-catenin pathway inhibitors in non-small-cell lung cancer cell lines. Bioorg Med Chem 19(18):5639–5647. doi:10.1016/j.bmc.2011.07.028
Griffin M, Mongeot A, Collighan R, Saint RE, Jones RA, Coutts IG, Rathbone DL (2008) Synthesis of potent water-soluble tissue transglutaminase inhibitors. Bioorg Med Chem Lett 18(20):5559–5562. doi:10.1016/j.bmcl.2008.09.006
Gupta R, Srinivasan R, Nijhawan R, Suri V (2010) Tissue transglutaminase 2 as a biomarker of cervical intraepithelial neoplasia (CIN) and its relationship to p16INK4A and nuclear factor kappaB expression. Virchows Arch 456(1):45–51. doi:10.1007/s00428-009-0860-5
Hollingshead MG, Alley MC, Camalier RF, Abbott BJ, Mayo JG, Malspeis L, Grever MR (1995) In vivo cultivation of tumor cells in hollow fibers. Life Sci 57(2):131–141
Iismaa SE, Mearns BM, Lorand L, Graham RM (2009) Transglutaminases and disease: lessons from genetically engineered mouse models and inherited disorders. Physiol Rev 89(3):991–1023. doi:10.1152/physrev.00044.2008
Kim SY (2006) Transglutaminase 2 in inflammation. Front Biosci 11:3026–3035. doi:10.1007/s00432-009-0681-6
Kim SY (2010) A new paradigm for cancer therapeutics development. BMB Rep 43(6):383–388
Kim SY (2011) Transglutaminase 2: a new paradigm for NF-kappaB involvement in disease. Adv Enzymol Relat Areas Mol Biol 78:161–195
Kim DS, Park SS, Nam BH, Kim IH, Kim SY (2006) Reversal of drug resistance in breast cancer cells by transglutaminase 2 inhibition and nuclear factor-kappaB inactivation. Cancer Res 66(22):10936–10943. doi:10.1158/0008-5472.CAN-06-1521
Kim DS, Park KS, Jeong KC, Lee BI, Lee CH, Kim SY (2009a) Glucosamine is an effective chemo-sensitizer via transglutaminase 2 inhibition. Cancer Lett 273(2):243–249. doi:10.1016/j.canlet.2008.08.015
Kim DS, Park KS, Kim SY (2009b) Silencing of TGase 2 sensitizes breast cancer cells to apoptosis by regulation of survival factors. Front Biosci 14:2514–2521. doi:10.2741/3394
Kim SJ, Kim KH, Ahn ER, Yoo BC, Kim SY (2011) Depletion of cathepsin D by transglutaminase 2 through protein cross-linking promotes cell survival. Amino Acids. doi:10.1007/s00726-011-1089-6
Ku JL, Park JG (2005) Biology of SNU cell lines. Cancer Res Treat 37(1):1–19. doi:10.4143/crt.2005.37.1.1
Lai TS, Liu Y, Tucker T, Daniel KR, Sane DC, Toone E, Burke JR, Strittmatter WJ, Greenberg CS (2008) Identification of chemical inhibitors to human tissue transglutaminase by screening existing drug libraries. Chem Biol 15(9):969–978. doi:10.1016/j.chembiol.2008.07.015
Lee T, Gong YD (2012) Solid-phase parallel synthesis of drug-like artificial 2H-benzopyran libraries. Molecules 17(5):5467–5496. doi:10.3390/molecules17055467
Lee J, Kim YS, Choi DH, Bang MS, Han TR, Joh TH, Kim SY (2004) Transglutaminase 2 induces nuclear factor-kappaB activation via a novel pathway in BV-2 microglia. J Biol Chem 279(51):53725–53735. doi:10.1074/jbc.M407627200
Lin CY, Tsai PH, Kandaswami CC, Chang GD, Cheng CH, Huang CJ, Lee PP, Hwang JJ, Lee MT (2011) Role of tissue transglutaminase 2 in the acquisition of a mesenchymal-like phenotype in highly invasive A431 tumor cells. Mol Cancer 10:87. doi:10.1186/1476-4598-10-87
Lorand L, Graham RM (2003) Transglutaminases: crosslinking enzymes with pleiotropic functions. Nat Rev Mol Cell Biol 4(2):140–156. doi:10.1038/nrm1014
Mann AP, Verma A, Sethi G, Manavathi B, Wang H, Fok JY, Kunnumakkara AB, Kumar R, Aggarwal BB, Mehta K (2006) Overexpression of tissue transglutaminase leads to constitutive activation of nuclear factor-kappaB in cancer cells: delineation of a novel pathway. Cancer Res 66(17):8788–8795. doi:10.1158/0008-5472.CAN-06-1457
Molnar Z, Bekesi JG (1972) Cytotoxic effects of D-glucosamine on the ultrastructures of normal and neoplastic tissues in vivo. Cancer Res 32(4):756–765
Pardin C, Pelletier JN, Lubell WD, Keillor JW (2008) Cinnamoyl inhibitors of tissue transglutaminase. J Org Chem 73(15):5766–5775. doi:10.1021/jo8004843
Park SS, Kim JM, Kim DS, Kim IH, Kim SY (2006) Transglutaminase 2 mediates polymer formation of I-kappaBalpha through C-terminal glutamine cluster. J Biol Chem 281(46):34965–34972. doi:10.1074/jbc.M604150200
Park KS, Kim DS, Ko C, Lee SJ, Oh SH, Kim SY (2011) TNF-alpha mediated NF-kappaB activation is constantly extended by transglutaminase 2. Front Biosci (Elite Ed) 3:341–354
Shao M, Cao L, Shen C, Satpathy M, Chelladurai B, Bigsby RM, Nakshatri H, Matei D (2009) Epithelial-to-mesenchymal transition and ovarian tumor progression induced by tissue transglutaminase. Cancer Res 69(24):9192–9201. doi:10.1158/0008-5472.CAN-09-1257
Shoemaker RH (2006) The NCI60 human tumour cell line anticancer drug screen. Nat Rev Cancer 6(10):813–823. doi:10.1038/nrc1951
Siegel M, Khosla C (2007) Transglutaminase 2 inhibitors and their therapeutic role in disease states. Pharmacol Ther 115(2):232–245. doi:10.1016/j.pharmthera.2007.05.003
Siegel M, Xia J, Khosla C (2007) Structure-based design of alpha-amido aldehyde containing gluten peptide analogues as modulators of HLA-DQ2 and transglutaminase 2. Bioorg Med Chem 15(18):6253–6261. doi:10.1016/j.bmc.2007.06.020
Sohn J, Kim TI, Yoon YH, Kim JY, Kim SY (2003) Novel transglutaminase inhibitors reverse the inflammation of allergic conjunctivitis. J Clin Invest 111(1):121–128. doi:10.1172/JCI15937
Verhaar R, Jongenelen CA, Gerard M, Baekelandt V, Van Dam AM, Wilhelmus MM, Drukarch B (2011) Blockade of enzyme activity inhibits tissue transglutaminase-mediated transamidation of alpha-synuclein in a cellular model of Parkinson’s disease. Neurochem Int 58(7):785–793. doi:10.1016/j.neuint.2011.03.004
Verma A, Mehta K (2007) Transglutaminase-mediated activation of nuclear transcription factor-kappaB in cancer cells: a new therapeutic opportunity. Curr Cancer Drug Targets 7(6):559–565
Yoo H, Ahn ER, Kim SJ, Lee SH, Oh SH, Kim SY (2013) Divergent results induced by different types of septic shock in transglutaminase 2 knockout mice. Amino Acids 44(1):189–197. doi:10.1007/s00726-012-1412-x
Yuan L, Siegel M, Choi K, Khosla C, Miller CR, Jackson EN, Piwnica-Worms D, Rich KM (2007) Transglutaminase 2 inhibitor, KCC009, disrupts fibronectin assembly in the extracellular matrix and sensitizes orthotopic glioblastomas to chemotherapy. Oncogene 26(18):2563–2573. doi:10.1038/sj.onc.1210048
Acknowledgments
This work was supported by a research grant (NCC1110011-2) from the National Cancer Center in Korea to S.Y.K. and National R&D Program for Cancer Control (No. 1020050) in Korea to Y.D.G. We declare that none of the authors have a financial interest related to this work, and none of the authors have any financial support beyond the research grant mentioned above.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Lee, SH., Kim, N., Kim, SJ. et al. Anti-cancer effect of a quinoxaline derivative GK13 as a transglutaminase 2 inhibitor. J Cancer Res Clin Oncol 139, 1279–1294 (2013). https://doi.org/10.1007/s00432-013-1433-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-013-1433-1