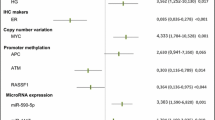
Abstract
We analysed the promoter methylation status of five genes, involved in adhesion (EPB41L3, TSLC-1), apoptosis (RASSF1, RASSF2) or angiogenesis (TSP-1), in intraoperative sentinel lymph node (SLN) biopsy samples from patients with breast cancer, that had been processed by the one-step nucleic acid amplification (OSNA) technique. SLN resection is performed to estimate the risk of tumour cells in the clinically negative axilla, to avoid unnecessary axillary lymph node dissection. OSNA is currently one of the eligible molecular methods for detecting tumour cells in SLNs. It is based on the quantitative evaluation of cytokeratin 19 mRNA which allows distinguishing between macrometastasis, micrometastasis and isolated tumour cells, on the basis of the quantity of tumour cells present. There have been no prior studies on the question whether or not samples processed by OSNA can be used for further molecular studies, including epigenetic abnormalities which are some of the most important molecular alterations in breast cancer. Genomic DNA was extracted from samples obtained from 50 patients diagnosed with primary breast cancer. The content of tumour cells in SLNs was evaluated by OSNA, and the promoter methylation status of the selected genes was analysed by methylation-specific PCR. All were found to be hypermethylated to a variable degree, and RASSF1 hypermethylation was significantly associated with macrometastasis, micrometastasis and isolated tumour cells (p = 0.002). We show that samples used for OSNA are suitable for molecular studies, including gene promoter methylation. These samples provide a new source of material for the identification of additional biomarkers.


Similar content being viewed by others
References
Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D (2011) Global cancer statistics. CA Cancer J Clin 61:69–90. doi:10.3322/caac.20107
Merlo DF, Ceppi M, Filiberti R, Bocchini V, Znaor A, Gamulin M, et al. (2012) Breast cancer incidence trends in European women aged 20-39 years at diagnosis. Breast Cancer Res Treat 134:363–370. doi:10.1007/s10549-012-2031-7
Youlden DR, Cramb SM, Dunn NA, Muller JM, Pyke CM, Baade PD (2012) The descriptive epidemiology of female breast cancer: an international comparison of screening, incidence, survival and mortality. Cancer Epidemiol 36:237–248. doi:10.1016/j.canep.2012.02.007
Sorlie T, Perou CM, Tibshirani R, Aas T, Geisler S, Johnsen H, et al. (2001) Gene expression patterns of breast carcinomas distinguish tumour subclasses with clinical implications. Proc Natl Acad Sci U S A 98:10869–10874. doi:10.1073/pnas.191367098
Network CGA (2012) Comprehensive molecular portraits of human breast tumours. Nature 490(7418):61–70. doi:10.1038/nature11412
Goldhirsch A, Winer EP, Coates AS, Gelber RD, Piccart-Gebhart M, Thürlimann B, et al. (2013) Personalizing the treatment of women with early breast cancer: highlights of the St Gallen International Expert Consensus on the Primary Therapy of Early Breast Cancer 2013. Ann Oncol 24:2206–2223. doi:10.1093/annonc/mdt303
Illyes I, Tokes AM, Kovacs A, Szasz AM, Molnar BA, Molnar IA, et al. (2014) In breast cancer patients sentinel lymph node metastasis characteristics predict further axillary involvement. Virchows Arch 465(1):15–24. doi:10.1007/s00428-014-1579-5
Reintgen D, Giuliano R, Cox CE (2000) Sentinel lymph node biopsy in breast cancer: an overview. Breast J 6:299–305. doi:10.1007/BF02967507
Breslin TM, Cohen L, Sahin A, Fleming JB, Kuerer HM, Newman LA et al (2000) Sentinel lymph node biopsy is accurate after neoadjuvant chemotherapy for breast cancer. J Clin Oncol 18:3480–3486. doi: 10.1016/S1470–2045(13)70166–9. doi: 10.1001/jama.2013.278932.
Veronesi U, Galimberti V, Zurrida S, Pigatto F, Veronesi P, Robertson C, et al. (2001) Sentinel lymph node biopsy as an indicator for axillary dissection in early breast cancer. Eur J Cancer 37:454–458. doi:10.1016/j.amjsurg.2007.08.068
Veronesi U, Paganelli G, Viale G, Galimberti V, Luini A, Zurrida S, et al. (1999) Sentinel lymph node biopsy and dissection in breast cancer: results in large series. J Natl Cancer Inst 91:368–373
Tsujimoto M, Nakabayashi K, Yoshidome K, Kaneko T, Iwase T, Akiyama F, et al. (2007) One-step nucleic acid amplification for intraoperative detection of lymph node metastasis in breast cancer patients. Clin Cancer Res 13:4807–4816. doi:10.1016/j.ejso.2013.12.007
Babar M, Madani R, Thwaites L, Jackson PA, Devalia HL, Chakravorty A, et al. (2014) A differential intra-operative molecular biological test for the detection of sentinel lymph node metastases in breast carcinoma. An extended experience from the first UK centre routinely offering the service in clinical practice. Eur J Surg Oncol 40(3):282–288. doi:10.1016/j.ejso.2013.10.030
Cserni G (2012) Intraoperative analysis of sentinel lymph nodes in breast cancer by one-step nucleic acid amplification. J Clin Pathol 65(3):193–199. doi:10.1136/jclinpath-2011-200301
Snook KL, Layer GT, Jackson PA, de Vries CS, Shousha S, Sinnett HD, et al. (2011) Multicentre evaluation of intraoperative molecular analysis of sentinel lymph nodes in breast carcinoma. Br J Surg 98:527–535. doi:10.1002/bjs.7347
Schem C, Maass N, Bauerschlag DO, Carstensen MH, Löning T, Roder C, et al. (2009) One-step nucleic acid amplification-a molecular method for the detection of lymph node metastases in breast cancer patients; results of the German study group. Virchows Arch 454:203–210. doi:10.1007/s00428-008-0703-9
Esteller M (2002) CpG island hypermethylation and tumor suppressor genes: a booming present, a brighter future. Oncogene 21:5427–5440. doi:10.1038/sj.onc.1205600
Wu Y, Sarkissyan M, Vadgama JV (2015) Epigenetics in breast and prostate cancer. Methods Mol Biol 1238:425–466. doi:10.1007/978-1-4939-1804-1_23
Liu C, Li P, Gao D, Zhou P, Shao Y, Zhang Q, et al. (2014) Abnormal promoter methylation of multiple tumor suppressor genes in human bronchial epithelial malignant cells. Biomed Rep 2:525–528. doi:10.3892/br.2014.268
Taback B, Giuliano AE, Lai R, Hansen N, Singer FR, Pantel K, et al. (2006) Epigenetic analysis of body fluids and tumor tissues: application of a comprehensive molecular assessment for early-stage breast cancer patients. Ann N Y Acad Sci 1075:211–221. doi:10.1196/annals.1368.029
Barekati Z, Radpour R, Lu Q, Bitzer J, Zheng H, Toniolo P, et al. (2012) Methylation signature of lymph node metastases in breast cancer patients. BMC Cancer 12:244. doi:10.1186/1471-2407-12-244
Cavalli LR, Urban CA, Dai D, de Assis S, Tavares DC, Rone JD, et al. (2003) Genetic and epigenetic alterations in sentinel lymph nodes metastatic lesions compared to their corresponding primary breast tumor. Cancer Genet Cytogenet 146:33–40. doi:10.1371/journal.pone.0103177
Shinozaki M, Hoon DS, Giuliano AE, Hansen NM, Wang HJ, Turner R, et al. (2005) Distinct hypermethylation profile of primary breast cancer is associated with sentinel lymph node metastasis. Clin Cancer Res 11:2156–2162. doi:10.1158/1078-0432.CCR-04
Guerrero-Setas D, Perez-Janices N, Blanco-Fernandez L, Ojer A, Cambra K, Berdasco M, et al. (2013) RASSF2 hypermethylation is present and related to shorter survival in squamous cervical cancer. Mod Pathol 26:1111–1122. doi:10.1038/modpathol.2013.32
Guerrero-Setas D, Perez-Janices N, Ojer A, Blanco-Fernandez L, Guarch-Troyas C, Guarch R (2013) Differential gene hypermethylation in genital lichen sclerosus and cancer: a comparative study. Histopathology 63:659–669. doi:10.1111/his.12204
Perez-Janices N, Blanco-Luquin I, Tuñón MT, Barba-Ramos E, Ibáñez B, Zazpe-Cenoz I et al (2015) EPB41L3, TSP-1 and RASSF2 as new clinically relevant prognostic biomarkers in diffuse gliomas. Oncotarget 6:368–380. doi: 10.18632/oncotarget.2745
Perez-Janices N, Blanco-Luquin I, Torrea N, Liechtenstein T, Escors D, Cordoba A et al (2015) Differential involvement of RASSF2 hypermethylation in breast cancer subtypes and their prognosis. Oncotarget 6(27):23944–23958. doi: 10.18632/oncotarget.4062
Blanco-Luquin I, Guarch R, Ojer A, Pérez-Janices N, Martín-Sánchez E, Maria-Ruiz S, et al. (2015) Differential role of gene hypermethylation in adenocarcinomas, squamous cell carcinomas and cervical intraepithelial lesions of the uterine cervix. Pathol Int 65(9):476–485. doi:10.1111/pin.12332
Yamada D, Kikuchi S, Williams YN, Sakurai-Yageta M, Masuda M, Maruyama T, et al. (2006) Promoter hypermethylation of the potential tumor suppressor DAL-1/4.1B gene in renal clear cell carcinoma. Int J Cancer 118:916–923. doi:10.1002/ijc.21450
Takahashi Y, Iwai M, Kawai T, Arakawa A, Ito T, Sakurai-Yageta M et al (2012) Aberrant expression of tumor suppressors CADM1 and 4.1B in invasive lesions of primary breast cancer. Breast Cancer 19(3):242–252. doi: 10.1007/s12282–011–0272-7. doi: 10.1007/s12282-011-0272-7
Watabe K, Ito A, Koma YI, Kitamura Y (2003) IGSF4: a new intercellular adhesion molecule that is called by three names, TSLC1, SgIGSF and SynCAM, by virtue of its diverse function. Histol Histopathol 18:1321–1329. doi:10.1007/s11033-013-2645-9
Chen KM, Stephen JK, Havard S, Mahan M, Divine G, Worsham MJ (2015) IGSF4 methylation as an independent marker of human papillomavirus-positive oropharyngeal squamous cell carcinoma. JAMA Otolaryngol Head Neck Surg 141:257–263. doi:10.1001/jamaoto.2014.3393
Richter AM, Pfeifer GP, Dammann RH (2009) The RASSF proteins in cancer; from epigenetic silencing to functional characterization. Biochim Biophys Acta 1796:114–128. doi:10.1016/j.bbcan.2009.03.004
Inda MM, Castresana JS (2007) RASSF1A promoter is highly methylated in primitive neuroectodermal tumours of the central nervous system. Neuropathology 27:341–346. doi:10.1111/j.1440-1789.2007.00788.x
Hagrass HA, Pasha HF, Shaheen MA, Abdel Bary EH, Kassem R (2014) Methylation status and protein expression of RASSF1A in breast cancer patients. Mol Biol Rep 41(1):57–65. doi:10.1007/s11033-013-2837-3
Majchrzak-Celinska A, Paluszczak J, Szalata M, Barciszewska AM, Nowak S, Kleszcz R, et al. (2015) The methylation of a panel of genes differentiates low-grade from high-grade gliomas. Tumour Biol 36(5):3831–3841. doi:10.1007/s13277-014-3025-3
Guerrero D, Guarch R, Ojer A, Casas JM, Méndez-Meca C, Esteller M, et al. (2011) Differential hypermethylation of genes in vulvar cancer and lichen sclerosus coexisting or not with vulvar cancer. Int J Cancer 128:2853–2864. doi:10.1002/ijc.25629
Guerrero D, Guarch R, Ojer A, Casas JM, Ropero S, Mancha A, et al. (2008) Hypermethylation of the thrombospondin-1 gene is associated with poor prognosis in penile squamous cell carcinoma. BJU Int 102(6):747–755. doi:10.1111/j.1464-410X.2008.07603.x
Vilardell F, Novell A, Martin J, Santacana M, Velasco A, Díez-Castro MJ, et al. (2012) Importance of assessing CK19 immunostaining in core biopsies in patients subjected to sentinel node study by OSNA. Virchows Arch 460(6):569–575. doi:10.1007/s00428-012-1241-z
Bernet L, Cano R, Martínez M, Dueñas B, Matias-Guiu X, Morell L, et al. (2011) Diagnosis of the sentinel lymph node in breast cancer: a reproducible molecular method. A multicentric Spanish study. Histopathology 58:863–869. doi:10.1111/j.1365-2559.2011.03836.x
Bernet L, Martínez M, Cano R, Sevilla F, Torro J, Ballester B, et al. (2011) Molecular diagnosis of sentinel lymph nodes for breast cancer: one step ahead for standardisation. Diagn Mol Pathol 20:18–21. doi:10.1097/PDM.0b013e3181eb9b30
Piñero A, Canteras M, Moreno A, Vicente F, Gimenez J, Tocino A, et al. (2013) Multicenter validation of two nomograms to predict non-sentinel node involvement in breast cancer. Clin Transl Oncol 15(2):117–123. doi:10.1007/s12094-012-0887-y
Giuliano AE, Hunt KK, Ballman KV, Beitsch PD, Whitworth PW, Blumencranz PW, et al. (2011) Axillary dissection vs no axillary dissection in women with invasive breast cancer and sentinel node metastasis: a randomized clinical trial. JAMA 305(6):569–575. doi:10.1001/jama.2011.90
Elston CW, Ellis IO (1991) Pathological prognostic factors in breast cancer. The value of histological grade in breast cancer: experience from a large study with long-term follow-up. Histopathology 19:403–410. doi:10.1111/j.1365-2559.1991.tb00229.x
Edge S, Byrd DR, Compton CC, Fritz AG, Greene FL, Trotti A (2010) AJCC cancer staging manual. Edited by Cancer AJ Co, Springer-Verlag New York
Ellis IO, Schnitt SJ, Sastre-Garau X, Bussolati G, Tavassoli FA, Eusebi V et al (2003) Tumours of breast: invasive breast carcinoma In: Tavasolli FA DPe, (ed). World Health Organization Classification of Tumours Pathology and Genetics of Tumours of the Breast and Female Genital Organs. IARC Press: Lyon; 13–59.
Layeeque R, Kepple J, Henry-Tillman RS, Adkins L, Kass R, Colvert M, et al. (2004) Intraoperative subareolar radioisotope injection for immediate sentinel lymph node biopsy. Ann Surg 239(6):841–848. doi:10.1097/01.sla.0000128304.13522.00
de Boer M, van Dijck JA, Bult P, Borm GF, Tjan-Heijnen VC (2010) Breast cancer prognosis and occult lymph node metastases, isolated tumor cells, and micrometastases. J Natl Cancer Inst 102:410–425. doi:10.3233/BD-2010-0302
Huxley N, Jones-Hughes T, Coelho H, Snowsill T, Cooper C, Meng Y et al (2015) A systematic review and economic evaluation of intraoperative tests [RD-100i one-step nucleic acid amplification (OSNA) system and Metasin test] for detecting sentinel lymph node metastases in breast cancer. Health Technol Assess 19: v-xxv, 1–215. doi: 10.3310/hta19020
Jara-Lazaro AR, Hussain IH, Thike AA, Wong CY, Ho GH, Yong WS, et al. (2014) Assessment of suitability of the one step nucleic acid amplification (OSNA) assay as an intraoperative procedure for detection of metastasis in sentinel lymph nodes of breast cancer. J Clin Pathol 67(12):1032–1037. doi:10.1136/jclinpath-2014-202361
Wong J, Yong WS, Thike AA, Iqbal J, Salahuddin AS, Ho GH, et al. (2015) False negative rate for intraoperative sentinel lymph node frozen section in patients with breast cancer: a retrospective analysis of patients in a single Asian institution. J Clin Pathol 68(7):536–540. doi:10.1136/jclinpath-2014-202799
Brambilla T, Fiamengo B, Tinterri C, Testori A, Grassi MM, Sciarra A, et al. (2015) One-step nucleic acid amplification in breast cancer sentinel lymph node: a single institutional experience and a short review. Front Med (Lausanne) 2:37. doi:10.3389/fmed.2015.00037
Kloten V, Becker B, Winner K, Schrauder MG, Fasching PA, Anzeneder T, et al. (2013) Promoter hypermethylation of the tumor-suppressor genes ITIH5, DKK3, and RASSF1A as novel biomarkers for blood-based breast cancer screening. Breast Cancer Res 15(1):R4. doi:10.1186/bcr3375
Wu Y, Zhang X, Lin L, Ma XP, Ma YC, Liu PS (2014) Aberrant methylation of RASSF2A in tumours and plasma of patients with epithelial ovarian cancer. Asian Pac J Cancer Prev 15:1171–1176. doi:10.1007/s13402-012-0106-4
Lee BB, Lee EJ, Jung EH, Chun HK, Chang DK, Song SY, et al. (2009) Aberrant methylation of APC, MGMT, RASSF2A, and Wif-1 genes in plasma as a biomarker for early detection of colorectal cancer. Clin Cancer Res 15:6185–6191. doi:10.1158/1078-0432.CCR-09-0111
Agostini M, Enzo MV, Bedin C, Belardinelli V, Goldin E, Del Bianco P, et al. (2012) Circulating cell-free DNA: a promising marker of regional lymphonode metastasis in breast cancer patients. Cancer Biomark 11(2–3):89–98. doi:10.3233/CBM-2012-0263
Boers A, Bosgraaf RP, van Leeuwen RW, Schuuring E, Heideman DA, Massuger LF, et al. (2014) DNA methylation analysis in self-sampled brush material as a triage test in hrHPV-positive women. Br J Cancer 111:1095–1101. doi:10.1038/bjc.2014.392
Bakkum-Gamez JN, Wentzensen N, Maurer MJ, Hawthorne KM, Voss JS, Kroneman TN, et al. (2015) Detection of endometrial cancer via molecular analysis of DNA collected with vaginal tampons. Gynecol Oncol 137:14–22. doi:10.1016/j.ygyno.2015.01.552
Hubers AJ, van der Drift MA, Prinsen CF, Witte BI, Wang Y, Shivapurkar N, et al. (2014) Methylation analysis in spontaneous sputum for lung cancer diagnosis. Lung Cancer 84:127–133. doi:10.1016/j.lungcan.2014.01.019
Nagasaka T, Tanaka N, Cullings HM, Sun DS, Sasamoto H, Uchida T, et al. (2009) Analysis of fecal DNA methylation to detect gastrointestinal neoplasia. J Natl Cancer Inst 101:1244–1258. doi:10.1093/jnci/djp265
Kioulafa M, Kaklamanis L, Mavroudis D, Georgoulias V, Lianidou ES (2009) Prognostic significance of RASSF1A promoter methylation in operable breast cancer. Clin Biochem 42(10–11):970–975. doi:10.1016/j.clinbiochem.2009.04.003
Sturgeon SR, Balasubramanian R, Schairer C, Muss HB, Ziegler RG, Arcaro KF (2012) Detection of promoter methylation of tumor suppressor genes in serum DNA of breast cancer cases and benign breast disease controls. Epigenetics 7(11):1258–1267. doi:10.4161/epi.22220
Xu J, Shetty PB, Feng W, Chenault C, Bast RC Jr, Issa JP, et al. (2012) Methylation of HIN-1, RASSF1A, RIL and CDH13 in breast cancer is associated with clinical characteristics, but only RASSF1A methylation is associated with outcome. BMC Cancer 12:243. doi:10.1186/1471-2407-12-243
Hinshelwood RA, Huschtscha LI, Melki J, Stirzaker C, Abdipranoto A, Vissel B, et al. (2007) Concordant epigenetic silencing of transforming growth factor-beta signalling pathway genes occurs early in breast carcinogenesis. Cancer Res 67(24):11517–11527. doi:10.1007/s11033-012-1810-x
Ahmed SS, Thike AA, Iqbal J, Yong WS, Tan B, Madhukumar P, et al. (2014) Sentinel lymph nodes with isolated tumour cells and micrometastases in breast cancer: clinical relevance and prognostic significance. J Clin Pathol 67:243–250. doi:10.1136/jclinpath-2013-201771
Weaver DL, Ashikaga T, Krag DN, Skelly JM, Anderson SJ, Harlow SP, et al. (2011) Effect of occult metastases on survival in node-negative breast cancer. N Engl J Med 364:412–421. doi:10.1056/NEJMoa1008108
Ram R, Singh J, McCaig E (2014) Sentinel node biopsy alone versus completion axillary node dissection in node positive breast cancer: systematic review and meta-analysis. Int J Breast Cancer 2014:513780. doi:10.1155/2014/513780
Sebova K, Zmetakova I, Bella V, Kajo K, Stankovicova I, Kajabova V, et al. (2011-2012) RASSF1A and CDH1 hypermethylation as potential epimarkers in breast cancer. Cancer Biomark 10(1):13–26. doi:10.3233/CBM-2012-0230
Barault L, Amatu A, Bleeker FE, Moutinho C, Falcomatà C, Fiano V, et al. (2015) Digital PCR quantification of MGMT methylation refines prediction of clinical benefit from alkylating agents in glioblastoma and metastatic colorectal cancer. Ann Oncol 26(9):1994–1999. doi:10.1093/annonc/mdv272
Acknowledgments
We are grateful to all the patients who participated in the study. We also thank staff of the Hospital Complex of Navarra (Navarra Health Service, Pamplona, Spain) for their invaluable support: E Barba, N Díaz, E Gochi, S Maria, ML Gomez-Dorronsoro, JM Martínez-Peñuela and A Panizo from the Department of Pathology; C Miranda and JM Lera from the Department of Surgery and S De La Cruz, JJ Illarramendi, E Salgado and R Vera from the Department of Medical Oncology. We also wish to thank A Aramendia, M Arraiza and E Reta from Navarrabiomed (Pamplona, Spain) for their technical assistance. We also appreciate the support from SARAY (Asociación Navarra de Cancer de Mama). This work was funded by a research grant from the Department of Health from the Government of Navarra (70/09). EMS is recipient of a postdoctoral grant from the SARAY (Asociación Navarra de Cancer de Mama); NPJ and IBL were recipients of an APICS predoctoral Fellowship from the Department of Health of the Government of Navarra and a postdoctoral grant from BIOEF, respectively.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding sources
This work was funded by a competitive research grant from the Department of Health from the Government of Navarra (70/09).
Conflict of interest
The authors declare that they have no conflict of interests.
Electronic Supplementary Material
Supplementary Fig. 1
Methylation-specific PCR (MSP) for methylation analysis of EPB41L3, TSLC-1 and TSP-1 genes in samples derived by the OSNA technique. The images show methylated and unmethylated cases, as indicated by the presence and absence of a band in the methylation reaction (M), respectively. The bands of interest and their size are indicated (bp: base pair; IVD: in vitro-methylated DNA; M: Methylation reaction; NL: DNA from normal lymphocytes; L: 100 bp DNA ladder; U: Unmethylation reaction). (TIFF 9619 kb)
Supplementary Table 1
(DOC 152 kb)
Supplementary Table 2
(DOC 36 kb)
Supplementary Table 3
(DOC 39 kb)
Supplementary Table 4
(DOC 36 kb)
Rights and permissions
About this article
Cite this article
Martín-Sánchez, E., Pernaut-Leza, E., Mendaza, S. et al. Gene promoter hypermethylation is found in sentinel lymph nodes of breast cancer patients, in samples identified as positive by one-step nucleic acid amplification of cytokeratin 19 mRNA. Virchows Arch 469, 51–59 (2016). https://doi.org/10.1007/s00428-016-1941-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00428-016-1941-x