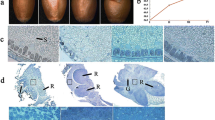
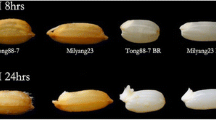
Abstract
Although embryo development is a major subject in plant growth and development research, a number of aspects of the mechanism of this development process remain unknown. Rice (Oryza sativa) is an excellent monocot plant model for studying embryogenesis with a known genome sequence. Here, we conducted proteomic analysis of embryo development in rice (O. sativa L. ssp. indica cv. 9311). The aim was to investigate and characterize the changes in the protein expression profile during embryo development. For this purpose, the proteome of developing embryos was characterized by two-dimensional gel electrophoresis and nano liquid chromatography/mass spectrometry/mass spectrometry. Proteomic analyses identified 275 differentially expressed proteins throughout the 5 sequential developmental stages from 5 to 30 days after pollination. Most of these proteins were classified into eight functional categories: metabolism, protein synthesis/destination, disease and defense, transporter, transcription, signal transduction, cell growth/division, and storage proteins, which were involved in different cellular and metabolic processes. Hierarchical clustering analyses of protein expression profiles showed that highly expressed proteins in early stages were involved in metabolism, protein synthesis/destination, and most of the other cellular functions, whereas the proteins highly expressed in later stages functioned in the desiccation and dormancy of the embryo.






Similar content being viewed by others
Abbreviations
- DAP:
-
Days after pollination
- FBA:
-
Fructose-bisphospate aldolase
- GAPDH:
-
Glyceraldehyde 3-P dehydrogenase
- PGAM:
-
2, 3-bisphosphoglycerate-independent phosphoglycerate mutase
- LEA:
-
Late embryogenesis abundant
- PFK:
-
Pyrophosphate-dependent phosphofructokinase
- PFP:
-
Pyrophosphate-fructose 6-phosphate 1-phosphotransferase
- TCA:
-
Tricarboxylic acid
References
Abbasi FM, Komatsu S (2004) A proteomic approach to analyze salt responsive proteins in rice leaf sheath. Proteomics 4:2072–2081
Agrawal GK, Rakwal R, Yonekura M, Kubo A, Saji H (2002) Proteome analysis of differentially displayed proteins as a tool for investigating ozone stress in rice (Oryza sativa L.) seedlings. Proteomics 2:947–959
Ahsan N, Lee DG, Lee SH, Kang KY, Lee JJ, Kim PJ, Yoon HS, Kim JS, Lee BH (2007) Excess copper induced physiological and proteomic changes in germinating rice seeds. Chemosphere 67:1182–1193
Ali N, Probert R, Hay F, Davies H, Stuppy W (2007) Post-dispersal embryo growth and acquisition of desiccation tolerance in Anemone nemorosa L. seeds. Seed Sci Res 17:155–163
Anderson NL, Anderson NG (1998) Proteome and proteomics: new technologies, new concepts, and new words. Electrophoresis 19:1853–1861
Andriotis VME, Kruger NJ, Pike MJ, Smith AM (2010) Plastidial glycolysis in developing Arabidopsis embryos. New Phytol 185:649–662
Balbuena TS, Silveira V, Junqueira M, Dias LL, Santa-Catarina C, Shevchenko A, Floh EI (2009) Changes in the 2-DE protein profile during zygotic embryogenesis in the Brazilian pine (Araucaria angustifolia). Proteomics 72:337–352
Battaglia M, Olvera-Carrillo Y, Garciarrubio A, Campos F, Covarrubias AA (2008) The enigmatic LEA proteins and other hydrophilins. Plant Physiol 148:6–24
Bechtel DB, Pomeranz Y (1978) Ultrastructure of the mature ungerminated rice (Oryza sativa) caryopsis. The starchy endosperm. Am J Bot 65:684–691
Bies-Ethève N, Gaubier-Comella P, Debures A, Lasserre E, Jobet E, Raynal M, Cooke R, Delseny M (2008) Inventory, evolution and expression profiling diversity of the LEA (late embryogenesis abundant) protein gene family in Arabidopsis thaliana. Plant Mol Biol 67:107–124
Bosch M, Mayer CD, Cookson A, Donnison IS (2011) Identification of genes involved in cell wall biogenesis in grasses by differential gene expression profiling of elongating and non-elongating maize internodes. J Exp Bot 62:3545–3561
Bradford MM (1976) A rapid and sensitive method for quantitation of microgram quantities of protein utilizing the principle of protein-dye-binding. Anal Biochem 72:248–254
Breviario D (2008) Plant tubulin genes: regulatory and evolutionary aspects. Plant Cell Monogr 11:207–232
Candiano G, Bruschi M, Musante L, Santucci L, Ghiggeri GM, Carnemolla B, Orecchia P, Zardi L, Righetti PG (2004) Blue silver: a very sensitive colloidal coomassie G-250 staining for proteome analysis. Electrophoresis 25:1327–1333
Carrari F, Fernie AR (2006) Metabolic regulation underlying tomato fruit development. J Exp Bot 57:1883–1897
Chen Q, Yang L, Ahmad P, Wan X, Hu X (2011) Proteomic profiling and redox status alteration of recalcitrant tea (Camellia sinensis) seed in response to desiccation. Planta 233:583–592
Chen S, Harmon AC (2006) Advances in plant proteomics. Proteomics 6:5504–5516
Chugh A, Khurana P (2002) Gene expression during somatic embryogenesis—recent advances. Curr Sci 86:715–730
Cordova-Tellez L, Burris JS (2002) Embryo drying rates during the acquisition of desiccation tolerance in maize seed. Crop Sci 42:1989–1995
Dai WG, Dong LC, Song YQ (2007) Nanosizing of a drug/carrageenan complex to increase solubility and dissolution rate. Int J Pharm 342:201–207
De Smet I, Lau S, Mayer U, Jürgens G (2010) Embryogenesis-the humble beginnings of plant life. Plant J 61:959–970
Egerton-Warburton LM, Balsamo RA, Close TJ (1997) Temporal accumulation and ultrastructural localization of dehydrins in Zea mays. Physiol Plant 101:545–555
Fait A, Angelovici R, Less H, Ohad I, Urbanczyk-Wochniak E, Fernie AR, Galili G (2006) Arabidopsis seed development and germination is associated with temporally distinct metabolic switches. Plant Physiol 142:839–854
Farré EM, Geigenberger P, Willmitzer L, Trethewey RN (2000) A possible role for pyrophosphate in the coordination of cytosolic and plastidial carbon metabolism within the potato tuber. Plant Physiol 123:681–688
Farré EM, Tiessen A, Roessner U, Geigenberger P, Trethewey RN, Willmitzer L (2001) Analysis of the compartmentation of glycolytic intermediates, nucleotides, sugars, organic acids, amino acids, and sugar alcohols in potato tubers using a nonaqueous fractionation method. Plant Physiol 127:685–700
Fernie AR, Carrari F, Sweetlove LJ (2004) Respiratory metabolism: glycolysis, the TCA cycle and mitochondrial electron transport. Curr Opin Plant Biol 7:254–261
Fiehn O, Kopka J, Dörmann P, Altmann T, Trethewey RN, Willmitzer L (2000) Metabolite profiling for plant functional genomics. Nat Biotechnol 18:1157–1161
Franco OL, Pelegrini PB, Gomes CP, Souza A, Costa FT, Domont G, Quirino BF, Eira MT, Mehta A (2009) Proteomic evaluation of coffee zygotic embryos in two different stages of seed development. Plant Physiol Biochem 47:1046–1050
Gallardo K, Firnhaber C, Zuber H, Héricher D, Belghazi M, Henry C, Küster H, Thompson R (2007) A combined proteome and transcriptome analysis of developing Medicago truncatula seeds: evidence for metabolic specialization of maternal and filial tissues. Mol Cell Proteomics 6:2165–2179
Garay-Arroyo A, Colmenero-Flores JM, Garciarrubio A, Covarrubias AA (2000) Highly hydrophilic proteins in prokaryotes and eukaryotes are common during conditions of water deficit. J Biol Chem 275:5668–5674
Gaubier P, Raynal M, Hull G, Huestis GM, Grellet F, Arenas C, Pagès M, Delseny M (1993) Two different Em-like genes are expressed in Arabidopsis thaliana seeds during maturation. Mol Gen Genet 238:409–418
Guillaumie S, San-Clemente H, Deswarte C, Martinez Y, Lapierre C, Murigneux A, Barriere Y, Pichon M, Goffner D (2007) Maizewall. Database and developmental gene expression profiling of cell wall biosynthesis and assembly in maize. Plant Physiol 143:339–363
Gygi SP, Rochon Y, Franza BR, Aebersold R (1999) Correlation between protein and mRNA abundance in yeast. Mol Cell Biol 19:1720–1730
Hajduch M, Casteel JE, Hurrelmeyer KE, Song Z, Agrawal GK, Thelen JJ (2006) Proteomic analysis of seed filling in Brassica napus: developmental characterization of metabolic isozymes using high-resolution two-dimensional gel electrophoresis. Plant Physiol 141:32–46
Harada JJ, DeLisle AJ, Baden CS, Crouch ML (1989) Unusual sequence of an abscisic acid inducible mRNA which accumulates late in Brassica napus seed development. Plant Mol Biol 12:395–401
Hewezi T, Petitprez M, Gentzbittel L (2006) Primary metabolic pathways and signal transduction in sunflower (Helianthus annuus L.): comparison of transcriptional profiling in leaves and immature embryos using cDNA microarrays. Planta 223:948–964
Hoekstra FA, Golovina EA, Tetteroo FA, Wolkers WF (2001) Induction of desiccation tolerance in plant somatic embryos: how exclusive is the protective role of sugars? Cryobiology 43:140–150
Hollung K, Espelund M, Jakobsen KS (1994) Another Lea B19 gene (group 1 Lea) from barley containing a single 20 amino acid hydrophilic motif. Plant Mol Biol 25:559–564
Hsing YC, Chen ZY, Shih MD, Hsieh JS, Chow TY (1995) Unusual sequences of group 3 LEA mRNA inducible by maturation or drying in soybean seeds. Plant Mol Biol 29:863–868
Ichihara K, Kobayashi N, Saito K (2003) Lipid synthesis and acyl-CoA synthetase in developing rice seeds. Lipids 38:881–884
Itoh J, Nonomura K, Ikeda K, Yamaki S, Inukai Y, Yamagishi H, Kitano H, Nagato Y (2005) Rice plant development: from zygote to spikelet. Plant Cell Physiol 46:23–47
Joosen R, Cordewener J, Supena ED, Vorst O, Lammers M, Maliepaard C, Zeilmaker T, Miki B, America T, Custers J, Boutilier K (2007) Combined transcriptome and proteome analysis identifies pathways and markers associated with the establishment of rapeseed microspore-derived embryo development. Plant Physiol 144:155–172
Katz E, Fon M, Lee YJ, Phinney BS, Sadka A, Blumwald E (2007) The citrus fruit proteome: insights into citrus fruit metabolism. Planta 226:989–1005
Kaufmann K, Smaczniak C, de Vries S, Angenent GC, Karlova R (2011) Proteomics insights into plant signaling and development. Proteomics 11:744–755
Kersten B, Bürkle L, Kuhn EJ, Giavalisco P, Konthur Z, Lueking A, Walter G, Eickhoff H, Schneider U (2002) Large-scale plant proteomics. Plant Mol Biol 48:133–141
Kim HS, Lee JH, Kim JJ, Kim CH, Jun SS, Hong YN (2005) Molecular and functional characterization of CaLEA6, the gene for a hydrophobic LEA protein from Capsicum annuum. Gene 344:115–123
Koga K, Minohata T (2011) An approach for identification of phosphoproteins using the G-electrode-loading method in two-dimensional gel electrophoresis. Proteomics 11:1545–1549
Komatsu S, Abbasi F, Kobori E, Fujisawa Y, Kato H, Iwasaki Y (2005) Proteomic analysis of rice embryo: an approach for investigating Galpha protein-regulated proteins. Proteomics 5:3932–3941
Kopczak SD, Haas NA, Hussey PJ, Silflow CD, Snustad DP (1992) The small genome of Arabidopsis contains at least six expressed alpha-tubulin genes. Plant Cell 4:539–547
Laux T, Würschum T, Breuninger H (2004) Genetic regulation of embryonic pattern formation. Plant Cell 16:S190–S202
Lee JM, Williams ME, Tingey SV, Rafalski JA (2002) DNA array profiling of gene expression changes during maize embryo development. Funct Integr Genomics 2:13–27
Lu TC, Meng LB, Yang CP, Liu GF, Liu GJ, Ma W, Wang BC (2008) A shotgun phosphoproteomics analysis of embryos in germinated maize seeds. Planta 228:1029–1041
Lyngved R, Renaut J, Hausman JF, Iversen TH, Hvoslef-Eide AK (2008) Embryo-specific proteins in Cyclamen persicum analyzed with 2-D DIGE. J Plant Growth Regul 27:353–369
Martin SW, Glover BJ, Davies JM (2005) Lipid microdomains—plant membranes get organized. Trends Plant Sci 10:263–265
Mayer U, Torres-Ruiz RA, Berleth T, Miseéra S, Jürgens G (1991) Mutations affecting body organization in the Arabidopsis embryo. Nature 353:402–407
Minic Z, Jamet E, San-Clemente H, Pelletier S, Renou JP, Rihouey C, Okinyo DP, Proux C, Lerouge P, Jouanin L (2009) Transcriptomic analysis of Arabidopsis developing stems: a close-up on cell wall genes. BMC Plant Biol 9:6
Parker KC, Garrels JI, Hines W, Butler EM, McKee AH, Patterson D, Martin S (1998) Identification of yeast proteins from two-dimensional gels: working out spot cross-contamination. Electrophoresis 19:1920–1932
Sengupta S, Majumder AL (2009) Insight into the salt tolerance factors of a wild halophytic rice, Porteresia coarctata: a physiological and proteomic approach. Planta 229:911–929
Sghaier-Hammami B, Drira N, Jorrín-Novo JV (2009) Comparative 2-DE proteomic analysis of date palm (Phoenix dactylifera L.) somatic and zygotic embryos. Proteomics 73:161–177
Spencer MW, Casson SA, Lindsey K (2007) Transcriptional profiling of the Arabidopsis embryo. Plant Physiol 143:924–940
Sulpice R, Trenkamp S, Steinfath M, Usadel B, Gibon Y, Witucka-Wall H, Pyl ET, Tschoep H, Steinhauser MC, Guenther M, Hoehne M, Rohwer JM, Altmann T, Fernie AR, Stitt M (2010) Network analysis of enzyme activities and metabolite levels and their relationship to biomass in a large panel of Arabidopsis accessions. Plant Cell 22:2872–2893
Takáč T, Pechan T, Samaj J (2011) Differential proteomics of plant development. J Proteomics 74:577–588
Tunnacliffe A, Wise MJ (2007) The continuing conundrum of the LEA proteins. Naturwissenschaften 94:791–812
Tzafrir I, Pena-Muralla R, Dickerman A, Berg M, Rogers R, Hutchens S, Sweeney TC, McElver J, Aux G, Patton D, Meinke D (2004) Identification of genes required for embryo development in Arabidopsis. Plant Physiol 135:1206–1220
Vicient CM, Gruber V, Delseny M (2001) The Arabidopsis AtEm1 promoter is active in Brassica napus L. and is temporally and spatially regulated. J Exp Bot 52:1587–1591
Vierstra RD (2003) The ubiquitin/26S proteasome pathway, the complex last chapter in the life of many plant proteins. Trends Plant Sci 8:135–142
West M, Harada JJ (1993) Embryogenesis in higher plants: an overview. Plant Cell 5:1361–1369
Williams BA, Tsang A (1994) Analysis of multiple classes of abscisic acid responsive genes during embryogenesis in Zea mays. Dev Genet 15:415–424
Winkelmann T, Heintz D, Van Dorsselaer A, Serek M, Braun HP (2006) Proteomic analyses of somatic and zygotic embryos of Cyclamen persicum Mill. reveal new insights into seed and germination physiology. Planta 224:508–519
Wise MJ, Tunnacliffe A (2004) POPP the question: what do LEA proteins do? Trends Plant Sci 9:13–17
Xu SB, Li T, Deng ZY, Chong K, Xue Y, Wang T (2008) Dynamic proteomic analysis reveals a switch between central carbon metabolism and alcoholic fermentation in rice filling grains. Plant Physiol 148:908–925
Yang P, Li X, Wang X, Chen H, Chen F, Shen S (2007a) Proteomic analysis of rice (Oryza sativa) seeds during germination. Proteomics 7:3358–3368
Yang Q, Wang Y, Zhang J, Shi W, Qian C, Peng X (2007b) Identification of aluminum-responsive proteins in rice roots by a proteomic approach: cysteine synthase as a key player in Al response. Proteomics 7:737–749
Zamboni A, Di Carli M, Guzzo F, Stocchero M, Zenoni S, Ferrarini A, Tononi P, Toffali K, Desiderio A, Lilley KS, Pè ME, Benvenuto E, Delledonne M, Pezzotti M (2010) Identification of putative stage-specific grapevine berry biomarkers and omics data integration into networks. Plant Physiol 154:1439–1459
Zegzouti H, Jones B, Marty C, Lelièvre JM, Latché A, Pech JC, Bouzayen M (1997) ER5, a tomato cDNA encoding an ethylene-responsive LEA-like protein: characterization and expression in response to drought, ABA and wounding. Plant Mol Biol 35:847–854
Acknowledgments
We thank Dr. Tai Wang (Institute of Botany, Chinese Academy of Sciences, China) for his help in 2-DE technique. This work was supported by the State Key Basic Research and Development Plan of China (2007CB108700), and the National Natural Science Foundation of China (30821064).
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
425_2011_1535_MOESM1_ESM.xls
Supplementary Table S1 Data obtained by nano-LC-MS/MS and protein database search for 242 differentially expressed protein spots. Sequence information obtained from nano-LC/MS/MS of a single excised protein spot was taken and submitted to MASCOT (Matrix Science Ltd.) search engine for protein identification using the Institute of Genomic Research (TIGR) protein database (XLS 314 kb)
425_2011_1535_MOESM2_ESM.doc
Supplementary Table S2 A catalog of identified proteins differentially expressed during embryo development in rice (DOC 599 kb)
425_2011_1535_MOESM3_ESM.xls
Supplementary Table S3 The proteins distribution in hierarchical clusters and the expression level of the differentially expressed proteins within each cluster. The following hierarchical clusters were created by GeneCluster 2.0. The proteins of unknown function were not listed (XLS 67 kb)
Rights and permissions
About this article
Cite this article
Xu, H., Zhang, W., Gao, Y. et al. Proteomic analysis of embryo development in rice (Oryza sativa). Planta 235, 687–701 (2012). https://doi.org/10.1007/s00425-011-1535-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-011-1535-4