Abstract
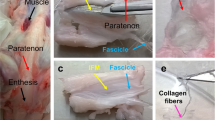
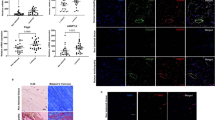
Tendons lack sufficient blood supply and represent a bradytroph tissue with prolonged healing time under pathological conditions. While the role of lymphatics in wound/defect healing in tissues with regular blood supply is well investigated, its involvement in tendon defects is not clear. We here try to identify the role of the lymphatic system in a tendon lesion model with morphological methods. A rat Achilles tendon lesion model (n = 5) was created via surgical intervention. Two weeks after surgery, animals were killed and lesioned site removed and prepared for polarization microscopy (picrosirius red) and immunohistochemistry using the lymphatic markers PROX1, VEGFR3, CCL21, LYVE-1, PDPN, and the vascular marker CD31. Additionally, DAPI was applied. Untreated tendons served as controls, confocal laser-scanning microscopy was used for documentation. At the lesion site, polarization microscopy revealed a structural reintegration while immunohistochemistry detected band-like profiles immunoreactive for PDPN, VEGFR3, CCL21, LYVE1, and CD31, surrounding DAPI-positive nuclei. PROX1-positive nuclei were detected within the lesion forming lines and opposed to each other. These PROX1-positive nuclei were surrounded by LYVE-1- or VEGFR3-positive surfaces. Few CD31-positve profiles contained PROX1-positive nuclei, while the majority of CD31-positive profiles lacked PROX1-positive nuclei. VEGFR3-, PDPN-, and LYVE-1-positive profiles were numerous within the lesion site, but absent in control tissue. Within 2 weeks, a structural rearrangement takes place in this lesion model, with dense lymphatic supply. The role of lymphatics in tendon wound healing is unclear, and proposed model represents a good possibility to study healing dynamics and lymphangiogenesis in a tissue almost completely lacking lymphatics in physiological conditions.




Similar content being viewed by others
References
Alitalo K (2011) The lymphatic vasculature in disease. Nat Med 17:1371–1380
Arya S, Kulig K (2010) Tendinopathy alters mechanical and material properties of the Achilles tendon. J Appl Physiol (1985) 108:670–675
Baluk P, McDonald DM (2008) Markers for microscopic imaging of lymphangiogenesis and angiogenesis. Ann N Y Acad Sci 1131:1–12
Banerji S, Ni J, Wang SX, Clasper S, Su J, Tammi R, Jones M, Jackson DG (1999) LYVE-1, a new homologue of the CD44 glycoprotein, is a lymph-specific receptor for hyaluronan. J Cell Biol 144:789–801
Breiteneder-Geleff S, Soleiman A, Kowalski H, Horvat R, Amann G, Kriehuber E, Diem K, Weninger W, Tschachler E, Alitalo K, Kerjaschki D (1999) Angiosarcomas express mixed endothelial phenotypes of blood and lymphatic capillaries: podoplanin as a specific marker for lymphatic endothelium. Am J Pathol 154:385–394
Bromley SK, Thomas SY, Luster AD (2005) Chemokine receptor CCR7 guides T cell exit from peripheral tissues and entry into afferent lymphatics. Nat Immunol 6:895–901
Carr AJ, Norris SH (1989) The blood supply of the calcaneal tendon. J Bone Joint Surg Br 71:100–101
Edwards DA (1946) The blood supply and lymphatic drainage of tendons. J Anat 80(147–152):142
Guermazi A, Brice P, Hennequin C, Sarfati E (2003) Lymphography: an old technique retains its usefulness. Radiographics 23:1541–1558 discussion 1559–1560
Heinemeier KM, Schjerling P, Heinemeier J, Magnusson SP, Kjaer M (2013) Lack of tissue renewal in human adult Achilles tendon is revealed by nuclear bomb (14)C. FASEB J 27:2074–2079
Huggenberger R, Detmar M (2011) The cutaneous vascular system in chronic skin inflammation. J Investig Dermatol Symp Proc 15:24–32
Jackson DG (2007) Lymphatic markers, tumour lymphangiogenesis and lymph node metastasis. Cancer Treat Res 135:39–53
Józsa LG, Kannus P (1997) Human tendons: anatomy, physiology, and pathology. Human Kinetics, Champaign, IL
Junghans BM, Collin HB (1989) Limbal lymphangiogenesis after corneal injury: an autoradiographic study. Curr Eye Res 8:91–100
Junqueira LC, Montes GS, Sanchez EM (1982) The influence of tissue section thickness on the study of collagen by the Picrosirius-polarization method. Histochemistry 74:153–156
Kaipainen A, Korhonen J, Mustonen T, van Hinsbergh VW, Fang GH, Dumont D, Breitman M, Alitalo K (1995) Expression of the fms-like tyrosine kinase 4 gene becomes restricted to lymphatic endothelium during development. Proc Natl Acad Sci U S A 92:3566–3570
Kerjaschki D (2014) The lymphatic vasculature revisited. J Clin Invest 124:874–877
Kesler CT, Liao S, Munn LL, Padera TP (2013) Lymphatic vessels in health and disease. Wiley Interdiscip Rev Syst Biol Med 5:111–124
Langberg H, Bulow J, Kjaer M (1998) Blood flow in the peritendinous space of the human Achilles tendon during exercise. Acta Physiol Scand 163:149–153
Lattouf R, Younes R, Lutomski D, Naaman N, Godeau G, Senni K, Changotade S (2014) Picrosirius red staining: a useful tool to appraise collagen networks in normal and pathological tissues. J Histochem Cytochem 62(10):751–758
Müller SA, Todorov A, Heisterbach PE, Martin I, Majewski M (2013) Tendon healing: an overview of physiology, biology, and pathology of tendon healing and systematic review of state of the art in tendon bioengineering. Knee Surg Sports Traumatol Arthrosc. doi:10.1007/s00167-013-2680-z
Nakao S, Maruyama K, Zandi S, Melhorn MI, Taher M, Noda K, Nusayr E, Doetschman T, Hafezi-Moghadam A (2010) Lymphangiogenesis and angiogenesis: concurrence and/or dependence? Studies in inbred mouse strains. FASEB J 24:504–513
Onuta G, Westerweel PE, Zandvoort A, van Riezen M, Rozing J, Hillebrands JL, Verhaar MC (2008) Angiogenic sprouting from the aortic vascular wall is impaired in the BB rat model of autoimmune diabetes. Microvasc Res 75:420–425
Podgrabinska S, Braun P, Velasco P, Kloos B, Pepper MS, Skobe M (2002) Molecular characterization of lymphatic endothelial cells. Proc Natl Acad Sci U S A 99:16069–16074
Puchtler H, Waldrop FS, Valentine LS (1973) Polarization microscopic studies of connective tissue stained with picro-sirius red FBA. Beitr Pathol 150:174–187
Pufe T, Petersen WJ, Mentlein R, Tillmann BN (2005) The role of vasculature and angiogenesis for the pathogenesis of degenerative tendons disease. Scand J Med Sci Sports 15:211–222
Pullinger BD, Florey HW (1937) Proliferation of lymphatics in inflammation. J Pathol Bacteriol 45:157–170
Regenfuss B, Bock F, Parthasarathy A, Cursiefen C (2008) Corneal (lymph)angiogenesis–from bedside to bench and back: a tribute to Judah Folkman. Lymphat Res Biol 6:191–201
Reis RM, Reis-Filho JS, Longatto Filho A, Tomarev S, Silva P, Lopes JM (2005) Differential Prox-1 and CD 31 expression in mucousae, cutaneous and soft tissue vascular lesions and tumors. Pathol Res Pract 201:771–776
Rich L, Whittaker P (2005) Collagen and Picrosirius Red staining: a polarized light assessment of fibrillar hue and spatial distribution. Braz J morphol Sci 22:97–104
Sahin H, Tholema N, Petersen W, Raschke MJ, Stange R (2012) Impaired biomechanical properties correlate with neoangiogenesis as well as VEGF and MMP-3 expression during rat patellar tendon healing. J Orthop Res 30:1952–1957
Scavelli C, Weber E, Agliano M, Cirulli T, Nico B, Vacca A, Ribatti D (2004) Lymphatics at the crossroads of angiogenesis and lymphangiogenesis. J Anat 204:433–449
Schrödl F, Trost A, Strohmaier C, Bogner B, Runge C, Kaser-Eichberger A, Eberhard N, Santic R, Kofler B, Reitsamer HA (2013) Distribution of the regulatory peptide alarin in the eye of various species. Exp Eye Res 106:74–81
Setti GC, Tazzi A (1972) Microscopical anatomy of the lymphatic circulation of the tendons. Ateneo Parmense 1(43):115–131
Sharma P, Maffulli N (2005) Basic biology of tendon injury and healing. Surgeon 3:309–316
Sharma P, Maffulli N (2006) Biology of tendon injury: healing, modeling and remodeling. J Musculoskelet Neuronal Interact 6:181–190
Skobe M, Detmar M (2000) Structure, function, and molecular control of the skin lymphatic system. J Investig Dermatol Symp Proc 5:14–19
Sleeman JP, Krishnan J, Kirkin V, Baumann P (2001) Markers for the lymphatic endothelium: in search of the holy grail? Microsc Res Tech 55:61–69
Sweat F, Puchtler H, Rosenthal SI (1964) Sirius red F3ba as a stain for connective tissue. Arch Pathol 78:69–72
Tempfer H, Wagner A, Gehwolf R, Lehner C, Tauber M, Resch H, Bauer HC (2009) Perivascular cells of the supraspinatus tendon express both tendon- and stem cell-related markers. Histochem Cell Biol 131:733–741
Thorpe CT, Streeter I, Pinchbeck GL, Goodship AE, Clegg PD, Birch HL (2010) Aspartic acid racemization and collagen degradation markers reveal an accumulation of damage in tendon collagen that is enhanced with aging. J Biol Chem 285:15674–15681
Woodfin A, Voisin MB, Nourshargh S (2007) PECAM-1: a multi-functional molecule in inflammation and vascular biology. Arterioscler Thromb Vasc Biol 27:2514–2523
Yang G, Rothrauff BB, Tuan RS (2013) Tendon and ligament regeneration and repair: clinical relevance and developmental paradigm. Birth Defects Res C Embryo Today 99:203–222
Acknowledgments
The Institute of Tendon and Bone Regeneration is part of the Austrian Cluster for Tissue Regeneration. This work was supported by the Hermann and Marianne Straniak Foundation (Sarnen, Switzerland), by the Lorenz Boehler Fund (Vienna, Austria), and by grants Nr. E09/09/051-BAH and R-13/02/047-TEM (HT) and R13/01/42-KAS (AK) from the Paracelsus Medical University Research Fund (Salzburg, Austria).
Conflict of interest
All authors disclose any actual or potential conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Herbert Tempfer and Alexandra Kaser-Eichberger have contributed equally to this work.
Rights and permissions
About this article
Cite this article
Tempfer, H., Kaser-Eichberger, A., Korntner, S. et al. Presence of lymphatics in a rat tendon lesion model. Histochem Cell Biol 143, 411–419 (2015). https://doi.org/10.1007/s00418-014-1287-x
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00418-014-1287-x