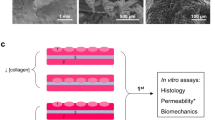
Abstract
The urinary tract is exposed to a variety of possible injures that may lead to organ damage or loss, and thus, the establishment of valid in vitro urothelial models to study the mechanism of drug candidates is necessary. This study is the first to investigate the effect of chitosan on urothelia in vitro and to evaluate whether chitosan-treated urothelial models can regenerate in vitro and reestablish a functional urothelium. Biomimetic hyperplastic and normoplastic urothelial models were used to test the effect of chitosan (0.05 %) on partially and highly differentiated urothelial cells (UCs) by monitoring their molecular, ultrastructural, and physiological changes for 3 weeks. Chitosan caused an immediate and complete loss of transepithelial resistance (TER), tight junction disruption, cytopathological changes of UCs, and consequently enhanced the permeability of partially and highly differentiated urothelial models. However, 3 weeks after chitosan treatment, TER was reestablished, tight junctions resealed, permeability decreased, and progressive differentiation stages of newly exposed superficial UCs expressing uroplakins and tight junction protein claudin-8 were found. The in vitro models regenerated and reestablished urothelia with a tight barrier. The biomimetic urothelial models represent appropriate in vitro models for studying urothelial drug candidates as well as evaluating drug permeabilities and their intracellular function. Understanding the possible intracellular function of chitosan could significantly advance approaches to treating urothelial-specific diseases.







Similar content being viewed by others
References
Blango MG, Ott EM, Erman A, Veranic P, Mulvey MA (2014) Forced resurgence and targeting of intracellular uropathogenic Escherichia coli reservoirs. PLoS ONE 9(3):e93327. doi:10.1371/journal.pone.0093327
Canali MM, Pedrotti LP, Balsinde J, Ibarra C, Correa SG (2012) Chitosan enhances transcellular permeability in human and rat intestine epithelium. Eur J Pharm Biopharm 80(2):418–425. doi:10.1016/j.ejpb.2011.11.007
Carr SF, Papp E, Wu JC, Natsumeda Y (1993) Characterization of human type I and type II IMP dehydrogenases. J Biol Chem 268(36):27286–27290
Dai T, Tanaka M, Huang YY, Hamblin MR (2011) Chitosan preparations for wounds and burns: antimicrobial and wound-healing effects. Expert Rev Anti Infect Ther 9(7):857–879. doi:10.1586/eri.11.59
Deli MA (2009) Potential use of tight junction modulators to reversibly open membranous barriers and improve drug delivery. Biochim Biophys Acta 1788(4):892–910. doi:10.1016/j.bbamem.2008.09.016
Dodane V, Khan MA, Merwin JR (1999) Effect of chitosan on epithelial permeability and structure. Int J Pharm 182(1):21–32
Dodou D, Breedveld P, Wieringa PA (2005) Mucoadhesives in the gastrointestinal tract: revisiting the literature for novel applications. Eur J Pharm Biopharm 60(1):1–16. doi:10.1016/j.ejpb.2005.01.007
Erman A, Kos MK, Zakelj S, Resnik N, Romih R, Veranič P (2013) Correlative study of functional and structural regeneration of urothelium after chitosan-induced injury. Histochem Cell Biol. doi:10.1007/s00418-013-1088-7
Hejazi R, Amiji M (2003) Chitosan-based gastrointestinal delivery systems. J Control Release 89(2):151–165
Hicks RM (1965) The fine structure of the transitional epithelium of rat ureter. J Cell Biol 26(1):25–48
Hicks RM (1975) The mammalian urinary bladder: an accommodating organ. Biol Rev Camb Philos Soc 50(2):215–246
Hsu LW, Lee PL, Chen CT, Mi FL, Juang JH, Hwang SM, Ho YC, Sung HW (2012) Elucidating the signaling mechanism of an epithelial tight-junction opening induced by chitosan. Biomaterials 33(26):6254–6263. doi:10.1016/j.biomaterials.2012.05.013
Hsu LW, Ho YC, Chuang EY, Chen CT, Juang JH, Su FY, Hwang SM, Sung HW (2013) Effects of pH on molecular mechanisms of chitosan-integrin interactions and resulting tight-junction disruptions. Biomaterials 34(3):784–793. doi:10.1016/j.biomaterials.2012.09.082
Jerman UD, Veranič P, Kreft ME (2013) Amniotic membrane scaffolds enable the development of tissue-engineered urothelium with molecular and ultrastructural properties comparable to that of native urothelium. Tissue Eng Part C Methods. doi:10.1089/ten.TEC.2013.0298
Kerec M, Bogataj M, Veranic P, Mrhar A (2005) Permeability of pig urinary bladder wall: the effect of chitosan and the role of calcium. Eur J Pharm Sci 25(1):113–121. doi:10.1016/j.ejps.2005.02.003
Kos MK, Bogataj M, Veranic P, Mrhar A (2006) Permeability of pig urinary bladder wall: time and concentration dependent effect of chitosan. Biol Pharm Bull 29(8):1685–1691
Kos MK, Bogataj M, Mrhar A (2008) Heparin decreases permeability of pig urinary bladder wall preliminarily enhanced by chitosan. Drug Dev Ind Pharm 34(2):215–220. doi:10.1080/03639040701542440
Kotze AF, Luessen HL, de Leeuw BJ, de Boer AG, Verhoef JC, Junginger HE (1998) Comparison of the effect of different chitosan salts and N-trimethyl chitosan chloride on the permeability of intestinal epithelial cells (Caco-2). J Control Release 51(1):35–46
Kotze AF, Luessen HL, de Boer AG, Verhoef JC, Junginger HE (1999) Chitosan for enhanced intestinal permeability: prospects for derivatives soluble in neutral and basic environments. Eur J Pharm Sci 7(2):145–151
Kreft ME, Hudoklin S, Sterle M (2005) Establishment and characterization of primary and subsequent subcultures of normal mouse urothelial cells. Folia Biol (Praha) 51(5):126–132
Kreft ME, Sterle M, Jezernik K (2006) Distribution of junction- and differentiation-related proteins in urothelial cells at the leading edge of primary explant outgrowths. Histochem Cell Biol 125(5):475–485. doi:10.1007/s00418-005-0104-y
Kreft ME, Romih R, Kreft M, Jezernik K (2009) Endocytotic activity of bladder superficial urothelial cells is inversely related to their differentiation stage. Differentiation 77(1):48–59. doi:10.1016/j.diff.2008.09.011
Kreft ME, Di Giandomenico D, Beznoussenko GV, Resnik N, Mironov AA, Jezernik K (2010) Golgi apparatus fragmentation as a mechanism responsible for uniform delivery of uroplakins to the apical plasma membrane of uroepithelial cells. Biol Cell 102(11):593–607. doi:10.1042/BC20100024
Lavelle J, Meyers S, Ramage R, Bastacky S, Doty D, Apodaca G, Zeidel ML (2002) Bladder permeability barrier: recovery from selective injury of surface epithelial cells. Am J Physiol Renal Physiol 283(2):F242–F253. doi:10.1152/ajprenal.00307.2001
Lilly JD, Parsons CL (1990) Bladder surface glycosaminoglycans is a human epithelial permeability barrier. Surg Gynecol Obstet 171(6):493–496
Min G, Zhou G, Schapira M, Sun TT, Kong XP (2003) Structural basis of urothelial permeability barrier function as revealed by Cryo-EM studies of the 16 nm uroplakin particle. J Cell Sci 116(Pt 20):4087–4094. doi:10.1242/jcs.00811
Negrete HO, Lavelle JP, Berg J, Lewis SA, Zeidel ML (1996) Permeability properties of the intact mammalian bladder epithelium. Am J Physiol 271(4 Pt 2):F886–F894
Parsons CL, Boychuk D, Jones S, Hurst R, Callahan H (1990) Bladder surface glycosaminoglycans: an epithelial permeability barrier. J Urol 143(1):139–142
Pick DL, Shelkovnikov S, Canvasser N, Louie MK, Tongson-Ignacio J, McDougall EM, Clayman RV (2011) First prize: chitosan and the urothelial barrier: effects on ureteral intraluminal drug penetration and peristalsis. J Endourol 25(3):385–390. doi:10.1089/end.2010.0205
Ranaldi G, Marigliano I, Vespignani I, Perozzi G, Sambuy Y (2002) The effect of chitosan and other polycations on tight junction permeability in the human intestinal Caco-2 cell line(1). J Nutr Biochem 13(3):157–167
Rosenthal R, Gunzel D, Finger C, Krug SM, Richter JF, Schulzke JD, Fromm M, Amasheh S (2012) The effect of chitosan on transcellular and paracellular mechanisms in the intestinal epithelial barrier. Biomaterials 33(9):2791–2800. doi:10.1016/j.biomaterials.2011.12.034
Stewart FA (1986) Mechanism of bladder damage and repair after treatment with radiation and cytostatic drugs. Br J Cancer Suppl 7:280–291
Truschel ST, Ruiz WG, Shulman T, Pilewski J, Sun TT, Zeidel ML, Apodaca G (1999) Primary uroepithelial cultures. A model system to analyze umbrella cell barrier function. J Biol Chem 274(21):15020–15029
Veranic P, Erman A, Kerec-Kos M, Bogataj M, Mrhar A, Jezernik K (2009) Rapid differentiation of superficial urothelial cells after chitosan-induced desquamation. Histochem Cell Biol 131(1):129–139. doi:10.1007/s00418-008-0492-x
Višnjar T, Kreft ME (2013) Air–liquid and liquid–liquid interfaces influence the formation of the urothelial permeability barrier in vitro. In Vitro Cell Dev Biol Anim 49(3):196–204. doi:10.1007/s11626-013-9585-5
Visnjar T, Kocbek P, Kreft ME (2012) Hyperplasia as a mechanism for rapid resealing urothelial injuries and maintaining high transepithelial resistance. Histochem Cell Biol 137(2):177–186. doi:10.1007/s00418-011-0893-0
Vllasaliu D, Casettari L, Fowler R, Exposito-Harris R, Garnett M, Illum L, Stolnik S (2012) Absorption-promoting effects of chitosan in airway and intestinal cell lines: a comparative study. Int J Pharm 430(1–2):151–160. doi:10.1016/j.ijpharm.2012.04.012
Walker DC, Hill G, Wood SM, Smallwood RH, Southgate J (2004) Agent-based computational modeling of wounded epithelial cell monolayers. IEEE Trans Nanobiosci 3(3):153–163
Yeh TH, Hsu LW, Tseng MT, Lee PL, Sonjae K, Ho YC, Sung HW (2011) Mechanism and consequence of chitosan-mediated reversible epithelial tight junction opening. Biomaterials 32(26):6164–6173. doi:10.1016/j.biomaterials.2011.03.056
Acknowledgments
The authors thank Professor Dr. Tung-Tien Sun (New York University Medical School) for his generous gift of uroplakin antibodies, Professor Dr. Peter Veranič for his critical reading of the manuscript, Eva Lasič for proofreading the manuscript, and Sanja Čabraja, Sabina Železnik, Nada Pavlica, and Linda Štrus for technical assistance. The study was supported by the Slovenian Research Agency (Grant No. P3-0108).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Višnjar, T., Kreft, M.E. The complete functional recovery of chitosan-treated biomimetic hyperplastic and normoplastic urothelial models. Histochem Cell Biol 143, 95–107 (2015). https://doi.org/10.1007/s00418-014-1265-3
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00418-014-1265-3