Abstract
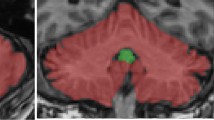
Neurological soft signs (NSS) are frequently found in psychiatric disorders of significant neurodevelopmental origin, e.g., in patients with schizophrenia and autism. Yet NSS are also present in healthy individuals suggesting a neurodevelopmental signature of motor function, probably as a continuum between health and disease. So far, little is known about the neural mechanisms underlying these motor phenomena in healthy persons, and it is even less known whether the cerebellum contributes to NSS expression. Thirty-seven healthy young adults (mean age = 23 years) were studied using high-resolution structural magnetic resonance imaging (MRI) and “resting-state” functional MRI at three Tesla. NSS levels were measured using the “Heidelberg Scale.” Cerebellar gray matter volume was investigated using cerebellum-optimized voxel-based analysis methods. Cerebellar function was assessed using regional homogeneity (ReHo), a measure of local network strength. The relationship between cerebellar structure and function and NSS was analyzed using regression models. There was no significant relationship between cerebellar volume and NSS (p < 0.005, uncorrected for height, p < 0.05 corrected for spatial extent). Positive associations with cerebellar lobule VI activity were found for the “motor coordination” and “hard signs” NSS domains. A negative relationship was found between lobule VI activity and “complex motor task” domain (p < 0.005, uncorrected for height, p < 0.05 corrected for spatial extent). The data indicate that in healthy young adults, distinct NSS domains are related to cerebellar activity, specifically with activity of cerebellar subregions with known cortical somatomotor projections. In contrast, cerebellar volume is not predictive of NSS in healthy persons.

Similar content being viewed by others
References
Schroder J, Niethammer R, Geider FJ, Reitz C, Binkert M, Jauss M et al (1991) Neurological soft signs in schizophrenia. Schizophr Res 6(1):25–30
Bombin I, Arango C, Buchanan RW (2005) Significance and meaning of neurological signs in schizophrenia: two decades later. Schizophr Bull 31(4):962–977
Chan RC, Gottesman II (2008) Neurological soft signs as candidate endophenotypes for schizophrenia: a shooting star or a Northern star? Neurosci Biobehav Rev 32(5):957–971
Hirjak D, Wolf RC, Stieltjes B, Hauser T, Seidl U, Schröder J, Thomann PA (2014) Cortical signature of neurological soft signs in recent onset schizophrenia. Brain Topogr 27(2):296–306. doi:10.1007/s10548-013-0292-z
Hirjak D, Wolf RC, Stieltjes B, Seidl U, Schröder J, Thomann PA (2012) Neurological soft signs and subcortical brain morphology in recent onset schizophrenia. J Psychiatr Res 46(4):533–539. doi:10.1016/j.jpsychires.2012.01.015
Mayoral M, Merchan-Naranjo J, Rapado M, Leiva M, Moreno C, Giraldez M et al (2010) Neurological soft signs in juvenile patients with Asperger syndrome, early onset psychosis, and healthy controls. Early Interv Psychiatry 4(4):283–290
De la Fuente JM, Bobes J, Vizuete C, Bascaran MT, Morlan I, Mendlewicz J (2006) Neurologic soft signs in borderline personality disorder. J Clin Psychiatry 67(4):541–546
Dazzan P, Morgan KD, Chitnis X, Suckling J, Morgan C, Fearon P et al (2006) The structural brain correlates of neurological soft signs in healthy individuals. Cereb Cortex 16(8):1225–1231
Zhao Q, Li Z, Huang J, Yan C, Dazzan P, Pantelis C, et al (2014) Neurological soft signs are not “soft” in brain structure and functional networks: evidence from ale meta-analysis. Schizophr Bull 40(3):626–641. doi:10.1093/schbul/sbt063
Hirjak D, Wolf RC, Kubera KM, Stieltjes B, Thomann PA (2014) Multiparametric mapping of neurological soft signs in healthy adults. Brain Struct Funct [Epub ahead of print]
Chan RC, Rao H, Chen EE, Ye B, Zhang C (2006) The neural basis of motor sequencing: an fMRI study of healthy subjects. Neurosci Lett 398(3):189–194
Schroder J, Wenz F, Schad LR, Baudendistel K, Knopp MV (1995) Sensorimotor cortex and supplementary motor area changes in schizophrenia. A study with functional magnetic resonance imaging. Br J Psychiatry 167(2):197–201
Schroder J, Essig M, Baudendistel K, Jahn T, Gerdsen I, Stockert A et al (1999) Motor dysfunction and sensorimotor cortex activation changes in schizophrenia: a study with functional magnetic resonance imaging. Neuroimage 9(1):81–87
Thomann PA, Hirjak D, Kubera KM, Stieltjes B, Wolf RC (2014) Neural network activity and neurological soft signs in healthy adults. Behav Brain Res 278C:514–519
Hirjak D, Wolf RC, Stieltjes B, Seidl U, Schroder J, Thomann PA (2012) Neurological soft signs and subcortical brain morphology in recent onset schizophrenia. J Psychiatr Res 46(4):533–539
Hirjak D, Wolf RC, Wilder-Smith EP, Kubera KM, Thomann PA (2015) Motor abnormalities and basal ganglia in schizophrenia: evidence from structural magnetic resonance imaging. Brain Topogr 28(1):135–152. doi:10.1007/s10548-014-0377-3
Thomann PA, Roebel M, Dos Santos V, Bachmann S, Essig M, Schroder J (2009) Cerebellar substructures and neurological soft signs in first-episode schizophrenia. Psychiatry Res 173(2):83–87
Hirjak D, Wolf RC, Kubera KM, Stieltjes B, Maier-Hein KH, Thomann PA (2015) Neurological soft signs in recent-onset schizophrenia: Focus on the cerebellum. Prog Neuropsychopharmacol Biol Psychiatry. doi:10.1016/j.pnpbp.2015.01.011
Schmahmann JD, Macmore J, Vangel M (2009) Cerebellar stroke without motor deficit: clinical evidence for motor and non-motor domains within the human cerebellum. Neuroscience 162(3):852–861
Stoodley CJ, Schmahmann JD (2009) Functional topography in the human cerebellum: a meta-analysis of neuroimaging studies. Neuroimage 44(2):489–501
Chan RC, Huang J, Di X (2009) Dexterous movement complexity and cerebellar activation: a meta-analysis. Brain Res Rev 59(2):316–323
Hardwick RM, Rottschy C, Miall RC, Eickhoff SB (2013) A quantitative meta-analysis and review of motor learning in the human brain. Neuroimage 67:283–297
Biswal B, Yetkin FZ, Haughton VM, Hyde JS (1995) Functional connectivity in the motor cortex of resting human brain using echo-planar MRI. Magn Reson Med 34(4):537–541
Fox MD, Corbetta M, Snyder AZ, Vincent JL, Raichle ME (2006) Spontaneous neuronal activity distinguishes human dorsal and ventral attention systems. Proc Natl Acad Sci USA 103(26):10046–10051
Fox MD, Snyder AZ, Vincent JL, Corbetta M, Van Essen DC, Raichle ME (2005) The human brain is intrinsically organized into dynamic, anticorrelated functional networks. Proc Natl Acad Sci USA 102(27):9673–9678
Bachmann S, Bottmer C, Schroder J (2005) Neurological soft signs in first-episode schizophrenia: a follow-up study. Am J Psychiatry 162(12):2337–2343
Diedrichsen J (2006) A spatially unbiased atlas template of the human cerebellum. Neuroimage 33(1):127–138
Kuhn S, Romanowski A, Schubert F, Gallinat J (2011) Reduction of cerebellar grey matter in Crus I and II in schizophrenia. Brain Struct Funct 217(2):523–529
Ashburner J, Friston KJ (2005) Unified segmentation. Neuroimage 26(3):839–851
Kuhn S, Romanowski A, Schilling C, Banaschewski T, Barbot A, Barker GJ et al (2012) Manual dexterity correlating with right lobule VI volume in right-handed 14-year-olds. Neuroimage 59(2):1615–1621
D’Agata F, Caroppo P, Boghi A, Coriasco M, Caglio M, Baudino B et al (2011) Linking coordinative and executive dysfunctions to atrophy in spinocerebellar ataxia 2 patients. Brain Struct Funct 216(3):275–288
Fan L, Tang Y, Sun B, Gong G, Chen ZJ, Lin X et al (2010) Sexual dimorphism and asymmetry in human cerebellum: an MRI-based morphometric study. Brain Res 1353:60–73
Zang Y, Jiang T, Lu Y, He Y, Tian L (2004) Regional homogeneity approach to fMRI data analysis. Neuroimage 22(1):394–400
Zou Q, Wu CW, Stein EA, Zang Y, Yang Y (2009) Static and dynamic characteristics of cerebral blood flow during the resting state. Neuroimage 48(3):515–524
Wu T, Long X, Zang Y, Wang L, Hallett M, Li K et al (2009) Regional homogeneity changes in patients with Parkinson’s disease. Hum Brain Mapp 30(5):1502–1510
Yao Z, Wang L, Lu Q, Liu H, Teng G (2009) Regional homogeneity in depression and its relationship with separate depressive symptom clusters: a resting-state fMRI study. J Affect Disord 115(3):430–438
Paakki JJ, Rahko J, Long X, Moilanen I, Tervonen O, Nikkinen J et al (2010) Alterations in regional homogeneity of resting-state brain activity in autism spectrum disorders. Brain Res 1321:169–179
Lui S, Li T, Deng W, Jiang L, Wu Q, Tang H et al (2010) Short-term effects of antipsychotic treatment on cerebral function in drug-naive first-episode schizophrenia revealed by “resting state” functional magnetic resonance imaging. Arch Gen Psychiatry 67(8):783–792
Diedrichsen J, Balsters JH, Flavell J, Cussans E, Ramnani N (2009) A probabilistic MR atlas of the human cerebellum. Neuroimage 46(1):39–46
Power JD, Barnes KA, Snyder AZ, Schlaggar BL, Petersen SE (2012) Spurious but systematic correlations in functional connectivity MRI networks arise from subject motion. Neuroimage 59(3):2142–2154
Visintin E, De Panfilis C, Antonucci C, Capecci C, Marchesi C, Sambataro F (2014) Parsing the intrinsic networks underlying attention: a resting state study. Behav Brain Res 278C:315–322
Hirjak D, Wolf RC, Koch SC, Mehl L, Kelbel JK, Kubera KM et al (2014) Neurological abnormalities in recent-onset schizophrenia and asperger-syndrome. Front Psychiatry 5:91
Thomann PA, Wustenberg T, Santos VD, Bachmann S, Essig M, Schroder J (2009) Neurological soft signs and brain morphology in first-episode schizophrenia. Psychol Med 39(3):371–379
Kong L, Bachmann S, Thomann PA, Essig M, Schroder J (2012) Neurological soft signs and gray matter changes: a longitudinal analysis in first-episode schizophrenia. Schizophr Res 134(1):27–32
Bottmer C, Bachmann S, Pantel J, Essig M, Amann M, Schad LR et al (2005) Reduced cerebellar volume and neurological soft signs in first-episode schizophrenia. Psychiatry Res 140(3):239–250
Fan LZ, Tang YC, Sun B, Gong GL, Chen ZJ, Lin XT et al (2010) Sexual dimorphism and asymmetry in human cerebellum: an MRI-based morphometric study. Brain Res 1353:60–73
Bernard JA, Seidler RD (2013) Relationships between regional cerebellar volume and sensorimotor and cognitive function in young and older adults. Cerebellum 12(5):721–737
Bernard JA, Seidler RD (2013) Cerebellar contributions to visuomotor adaptation and motor sequence learning: an ALE meta-analysis. Front Hum Neurosci 7:27
Stoodley CJ, Valera EM, Schmahmann JD (2010) An fMRI study of intra-individual functional topography in the human cerebellum. Behav Neurol 23(1–2):65–79
Buckner RL (2013) The cerebellum and cognitive function: 25 years of insight from anatomy and neuroimaging. Neuron 80(3):807–815
Krienen FM, Buckner RL (2009) Segregated fronto-cerebellar circuits revealed by intrinsic functional connectivity. Cereb Cortex 19(10):2485–2497
Grodd W, Hulsmann E, Lotze M, Wildgruber D, Erb M (2001) Sensorimotor mapping of the human cerebellum: fMRI evidence of somatotopic organization. Hum Brain Mapp 13(2):55–73
Bushara KO, Wheat JM, Khan A, Mock BJ, Turski PA, Sorenson J et al (2001) Multiple tactile maps in the human cerebellum. NeuroReport 12(11):2483–2486
Baillieux H, De Smet HJ, Paquier PF, De Deyn PP, Marien P (2008) Cerebellar neurocognition: insights into the bottom of the brain. Clin Neurol Neurosurg 110(8):763–773
Schmahmann JD (1991) An emerging concept. The cerebellar contribution to higher function. Arch Neurol 48(11):1178–1187
Schmahmann JD (1996) From movement to thought: anatomic substrates of the cerebellar contribution to cognitive processing. Hum Brain Mapp 4(3):174–198
Wisner KM, Atluri G, Lim KO, Macdonald AW 3rd (2013) Neurometrics of intrinsic connectivity networks at rest using fMRI: retest reliability and cross-validation using a meta-level method. Neuroimage 76:236–251
Acknowledgments
The authors would like to thank all participants for their time and interest in this study. We are grateful to Fabio Sambataro for insightful suggestions and comments on a previous version of this manuscript, as these comments led us to improve our work.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Additional information
Dusan Hirjak and Philipp A. Thomann have contributed equally to this article.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Hirjak, D., Thomann, P.A., Kubera, K.M. et al. Cerebellar contributions to neurological soft signs in healthy young adults. Eur Arch Psychiatry Clin Neurosci 266, 35–41 (2016). https://doi.org/10.1007/s00406-015-0582-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00406-015-0582-4