Abstract
Objective
To assess the use, side effects and discontinuation rates of iron preparations during pregnancy.
Design
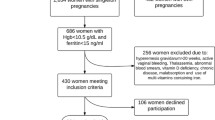
Six hundred and twelve randomly selected postpartum women completed a questionnaire on iron supplement use in the second and third trimesters.
Results
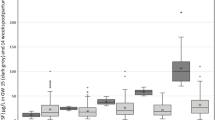
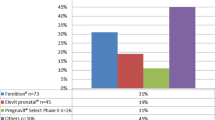
Of the 517 women (84.5%) reported using iron supplements, 453 were eligible for the study. The most common preparation was ferrous fumarate (46.8%, P < 0.01), followed by ferrous sulfate (31.8%), ferric polymaltose (12.4%), and ferric bisglycinate (7.3%). Almost half the participants (45%) reported at least one adverse effect, especially constipation (27.4%, P < 0.01), nausea (10.8%). Multivitamin preparations and ferric bisglycinate were associated with the fewest side effects (23.7, 21.2% respectively, P < 0.01), and ferrous fumarate and immediate-release ferrous sulfate with the most (56.3, 53.7% respectively). Eighty-three women discontinued their originally prescribed iron preparation, mainly (89%) due to side effects. Discontinuation rates were lowest for the multivitamin and ferric bisglycinate (10.5, 9.1%, respectively). In most cases, the specific preparation was recommended by the women’s physician (76%).
Conclusion
Ferrous fumarate-containing multivitamin preparations and ferric bisglycinate, although infrequently recommended as the first-line of iron supplementation, may be associated with less side effects and better compliance.
Similar content being viewed by others
References
World Health Organization (1992) The prevalence of anaemia in women: a tabulation of available information. WHO, Geneva
Macgregor M (1963) Maternal anaemia as a factor in prematurity and perinatal mortality. Scott Med J 8:134
Scholl TO, Hediger ML (1994) Anemia and iron-deficiency anemia: compilation of data on pregnancy outcome (discussion 500S–501S). Am J Clin Nutr 59(2 Suppl):492S–500S
Levy A, Fraser D, Katz M, Mazor M, Sheiner E (2005) Maternal anemia during pregnancy is an independent risk factor for low birthweight and preterm delivery. Eur J Obstet Gynecol Reprod Biol 122(2):182–186
Stoltzfus RD, Dreyfuss ML (2000) Guidelines for the use of iron supplements to prevent and treat iron deficiency anemia. International Nutritional Anemia Consultative Group (INACG)
Pena-Rosas JP, Viteri FE (2006) Effects of routine oral iron supplementation with or without folic acid for women during pregnancy. Cochrane Database Syst Rev 3:CD004736
Casanueva E, Viteri FE, Mares-Galindo M, Meza-Camacho C, Loria A, Schnaas L et al (2006) Weekly iron as a safe alternative to daily supplementation for nonanemic pregnant women. Arch Med Res 37(5):674–682
Hemminki E, Merilainen J (1995) Long-term follow-up of mothers and their infants in a randomized trial on iron prophylaxis during pregnancy. Am J Obstet Gynecol 173(1):205–209
Schultink W, van der Ree M, Matulessi P, Gross R (1993) Low compliance with an iron-supplementation program: a study among pregnant women in Jakarta, Indonesia. Am J Clin Nutr 57(2):135–139
Nordeng H, Eskild A, Nesheim BI, Aursnes I, Jacobsen G (2003) Guidelines for iron supplementation in pregnancy: compliance among 431 parous Scandinavian women. Eur J Clin Pharmacol 59(2):163–168
Yu SM, Keppel KG, Singh GK, Kessel W (1996) Preconceptional and prenatal multivitamin-mineral supplement use in the 1988 National Maternal and Infant Health Survey. Am J Public Health 86(2):240–242
DeMaeyer EM, Dallman P, Gumey JM, Hallberg L, Sood SK, Srikantia SG (1989) Preventing and controlling iron deficiency anaemia through primary health care. WHO, Geneva
Hansen CM (1994) Oral iron supplements. Am Pharm NS34(3):66–71
Rybo G, Solvell L (1971) Side-effect studies on a new sustained release iron preparation. Scand J Haematol 8(4):257–264
Brock C, Curry H, Hanna C, Knipfer M, Taylor L (1985) Adverse effects of iron supplementation: a comparative trial of a wax-matrix iron preparation and conventional ferrous sulfate tablets. Clin Ther 7(5):568–573
Jacobs P, Wood L, Bird AR (2000) Erythrocytes: better tolerance of iron polymaltose complex compared with ferrous sulphate in the treatment of anaemia. Hematol 5(1):77–83
Pineda O, Ashmead HD (2001) Effectiveness of treatment of iron-deficiency anemia in infants and young children with ferrous bis-glycinate chelate. Nutrition 17(5):381–384
Jacobs P, Fransman D, Coghlan P (1993) Comparative bioavailability of ferric polymaltose and ferrous sulphate in iron-deficient blood donors. J Clin Apher 8(2):89–95
Bezkorovainy A (1989) Biochemistry of nonheme iron in man. II. Absorption of iron. Clin Physiol Biochem 7(2):53–69
Everson GT (1992) Gastrointestinal motility in pregnancy. Gastroenterol Clin North Am 21(4):751–76
Greenhalf JO, Leonard HS (1973) Laxatives in the treatment of constipation in pregnant and breast-feeding mothers. Practitioner 210(256):259–263
Klebanoff MA, Koslowe PA, Kaslow R, Rhoads GG (1985) Epidemiology of vomiting in early pregnancy. Obstet Gynecol 66(5):612–616
FitzGerald CM (1984) Nausea and vomiting in pregnancy. Br J Med Psychol 57(Pt 2):159–165
Tierson FD, Olsen CL, Hook EB (1986) Nausea and vomiting of pregnancy and association with pregnancy outcome. Am J Obstet Gynecol 155(5):1017–1022
Hyder SM, Persson LA, Chowdhury AM, Ekstrom EC (2002) Do side-effects reduce compliance to iron supplementation? A study of daily- and weekly-dose regimens in pregnancy. J Health Popul Nutr 20(2):175–179
Makrides M, Crowther CA, Gibson RA, Gibson RS, Skeaff CM (2003) Efficacy and tolerability of low-dose iron supplements during pregnancy: a randomized controlled trial. Am J Clin Nutr 78(1):145–153
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Melamed, N., Ben-Haroush, A., Kaplan, B. et al. Iron supplementation in pregnancy—does the preparation matter?. Arch Gynecol Obstet 276, 601–604 (2007). https://doi.org/10.1007/s00404-007-0388-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00404-007-0388-3